Positively selected germinal center B cells (GCBC) can either resume proliferation and somatic hypermutation or differentiate. The mechanisms dictating these alternative cell fates are incompletely understood. We show that the protein arginine methyltransferase 1 (Prmt1) is upregulated in murine GCBC by Myc and mTORC-dependent signaling after positive selection. Deleting Prmt1 in activated B cells compromises antibody affinity maturation by hampering proliferation and GCBC light zone to dark zone cycling. Prmt1 deficiency also results in enhanced memory B cell generation and plasma cell differentiation, albeit the quality of these cells is compromised by the GCBC defects. We further demonstrate that Prmt1 intrinsically limits plasma cell differentiation, a function co-opted by B cell lymphoma (BCL) cells. Consistently, PRMT1 expression in BCL correlates with poor disease outcome, depends on MYC and mTORC1 activity, is required for cell proliferation, and prevents differentiation. Collectively, these data identify PRMT1 as a determinant of normal and cancerous mature B cell proliferation and differentiation balance.© 2023 Litzler et al.
In The Journal of Experimental Medicine on 4 September 2023 by Litzler, L. C., Zahn, A., et al.
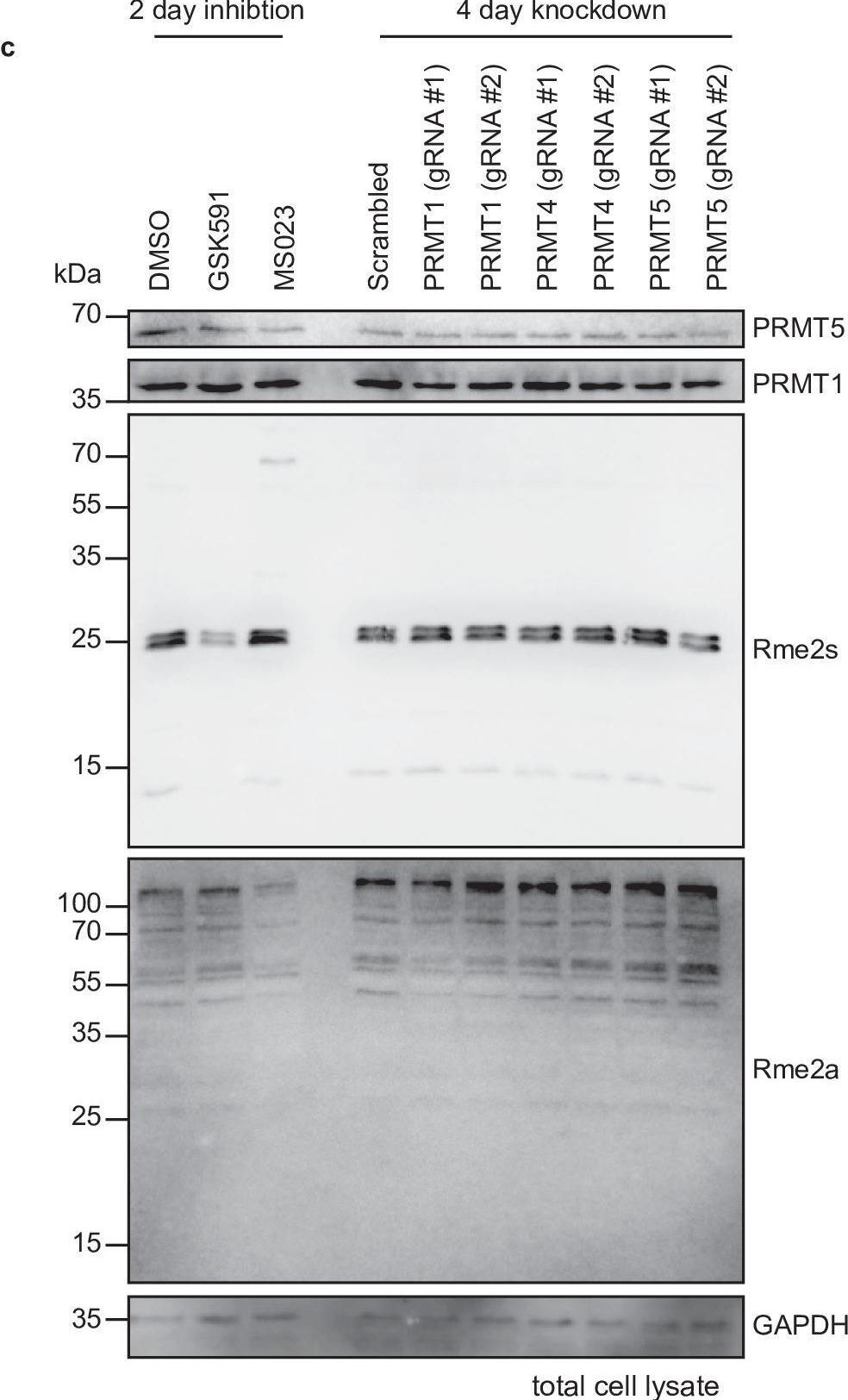
In eLife on 5 January 2022 by Maron, M. I., Casill, A. D., et al.
Protein arginine methyltransferases (PRMTs) are required for the regulation of RNA processing factors. Type I PRMT enzymes catalyze mono- and asymmetric dimethylation; Type II enzymes catalyze mono- and symmetric dimethylation. To understand the specific mechanisms of PRMT activity in splicing regulation, we inhibited Type I and II PRMTs and probed their transcriptomic consequences. Using the newly developed Splicing Kinetics and Transcript Elongation Rates by Sequencing (SKaTER-seq) method, analysis of co-transcriptional splicing demonstrated that PRMT inhibition resulted in altered splicing rates. Surprisingly, co-transcriptional splicing kinetics did not correlate with final changes in splicing of polyadenylated RNA. This was particularly true for retained introns (RI). By using actinomycin D to inhibit ongoing transcription, we determined that PRMTs post-transcriptionally regulate RI. Subsequent proteomic analysis of both PRMT-inhibited chromatin and chromatin-associated polyadenylated RNA identified altered binding of many proteins, including the Type I substrate, CHTOP, and the Type II substrate, SmB. Targeted mutagenesis of all methylarginine sites in SmD3, SmB, and SmD1 recapitulated splicing changes seen with Type II PRMT inhibition, without disrupting snRNP assembly. Similarly, mutagenesis of all methylarginine sites in CHTOP recapitulated the splicing changes seen with Type I PRMT inhibition. Examination of subcellular fractions further revealed that RI were enriched in the nucleoplasm and chromatin. Taken together, these data demonstrate that, through Sm and CHTOP arginine methylation, PRMTs regulate the post-transcriptional processing of nuclear, detained introns.
© 2022, Maron et al.
In IScience on 24 September 2021 by Maron, M. I., Lehman, S. M., et al.
Protein arginine methyltransferases (PRMTs) catalyze the post-translational monomethylation (Rme1), asymmetric (Rme2a), or symmetric (Rme2s) dimethylation of arginine. To determine the cellular consequences of type I (Rme2a) and II (Rme2s) PRMTs, we developed and integrated multiple approaches. First, we determined total cellular dimethylarginine levels, revealing that Rme2s was ∼3% of total Rme2 and that this percentage was dependent upon cell type and PRMT inhibition status. Second, we quantitatively characterized in vitro substrates of the major enzymes and expanded upon PRMT substrate recognition motifs. We also compiled our data with publicly available methylarginine-modified residues into a comprehensive database. Third, we inhibited type I and II PRMTs and performed proteomic and transcriptomic analyses to reveal their phenotypic consequences. These experiments revealed both overlapping and independent PRMT substrates and cellular functions. Overall, this study expands upon PRMT substrate diversity, the arginine methylome, and the complex interplay of type I and II PRMTs.
© 2021 The Author(s).
In The EMBO Journal on 1 July 2021 by Repenning, A., Happel, D., et al.
The p14ARF protein is a well-known regulator of p53-dependent and p53-independent tumor-suppressive activities. In unstressed cells, p14ARF is predominantly sequestered in the nucleoli, bound to its nucleolar interaction partner NPM. Upon genotoxic stress, p14ARF undergoes an immediate redistribution to the nucleo- and cytoplasm, where it promotes activation of cell cycle arrest and apoptosis. Here, we identify p14ARF as a novel interaction partner and substrate of PRMT1 (protein arginine methyltransferase 1). PRMT1 methylates several arginine residues in the C-terminal nuclear/nucleolar localization sequence (NLS/NoLS) of p14ARF . In the absence of cellular stress, these arginines are crucial for nucleolar localization of p14ARF . Genotoxic stress causes augmented interaction between PRMT1 and p14ARF , accompanied by arginine methylation of p14ARF . PRMT1-dependent NLS/NoLS methylation promotes the release of p14ARF from NPM and nucleolar sequestration, subsequently leading to p53-independent apoptosis. This PRMT1-p14ARF cooperation is cancer-relevant and indicative for PDAC (pancreatic ductal adenocarcinoma) prognosis and chemotherapy response of pancreatic tumor cells. Our data reveal that PRMT1-mediated arginine methylation is an important trigger for p14ARF 's stress-induced tumor-suppressive function.
© 2021 The Authors. Published under the terms of the CC BY NC ND 4.0 license.
In FEBS Letters on 1 September 2020 by Park, M. J., Liao, J., et al.
We previously reported the involvement of protein arginine methyltransferase 1 (PRMT1) in adipocyte thermogenesis. Here, we investigate the effects of PRMT1 inhibitors on thermogenesis. Unexpectedly, we find that the PRMT1 inhibitor TC-E 5003 (TC-E) induces the thermogenic properties of primary murine and human subcutaneous adipocytes. TC-E treatment upregulates the expression of Ucp1 and Fgf21 significantly and activates protein kinase A signaling and lipolysis in primary subcutaneous adipocytes from both mouse and humans. We further find that the thermogenic effects of TC-E are independent of PRMT1 and beta-adrenergic receptors. Our data indicate that TC-E exerts strong effects on murine and human subcutaneous adipocytes by activating beige adipocytes via PKA signaling.
© 2020 Federation of European Biochemical Societies.
In Elife on 5 January 2022 by Maron, M. I., Casill, A. D., et al.
Fig.1.C
-
WB
-
Collected and cropped from eLife by CiteAb, provided under a CC-BY license
Image 1 of 1
