ABSTRACT High expression of the myeloid master ETS transcription factor PU.1 drives the development of monocyte/macrophage (Mono/MΦ), a crucial cellular component of the innate immune system. Disruptions in normal expression patterns of PU.1 are linked to a variety myeloid malignancy and immune diseases. It is evidenced that PU.1 binds to and modulates enhancers of several myeloid genes. While noncoding RNAs transcribed from noncoding genes at the enhancers are increasingly reported to be involved in enhancer regulation, the crosstalk between PU.1 and noncoding RNAs in enhancer-mediated myeloid gene regulation in Mono/MΦ differentiation and immune response has not been systematically investigated. In this study, we interrogated the PU.1-mediated transcriptome and cistrome with our comprehensive collection of putative and verified enhancers. Among a repertoire of noncoding genes present at PU.1-bound enhancers, we discovered that PU.1 acts as a potent transcription factor inducer of the noncoding RNA LOUP , which we previously identified as an RNA inducer of PU.1. The genomic region within the LOUP locus occupied by PU.1 is characterized by the epigenetic features of a myeloid-specific super-enhancer. Targeted disruption of the PU.1-binding motifs resulted in the downregulation of LOUP promoter activity. Depletion of LOUP reduced the expression of Mono/MΦ cell markers as well as the transcriptional program associated with Mono/MΦ differentiation Mono/MΦ innate defense mechanisms, including phagocytosis, antimicrobial activity, and chemoattractant cytokine production. LOUP induces Mono/MΦ phagocytic activities. Collectively, our findings indicate that PU.1 and enhancer RNA LOUP are biomolecular components of an unidentified feed-forward loop that promotes their mutual expression, contributing to Mono/MΦ differentiation and innate immune functions. The identification of the PU.1/ LOUP regulatory circuit provides valuable insights into the mechanisms underlying cell-type and gene-specific enhancer activity and Mono/MΦ biology, as well as significant implications for advancing our understanding of immune diseases and myeloid malignancies.
Product Citations: 13
Preprint on BioRxiv : the Preprint Server for Biology on 21 February 2025 by Waliullah, A. S. M., Qiu, K., et al.
-
Genetics
-
Immunology and Microbiology
In IScience on 19 July 2024 by AlSaeed, H., Haider, M. J. A., et al.
The interplay between lipid metabolism and immune response in macrophages plays a pivotal role in various infectious diseases, notably tuberculosis (TB). Herein, we illuminate the modulatory effect of heat-killed Mycobacterium tuberculosis (HKMT) on macrophage lipid metabolism and its implications on the inflammatory cascade. Our findings demonstrate that HKMT potently activates the lipid scavenger receptor, CD36, instigating lipid accumulation. While CD36 inhibition mitigated lipid increase, it unexpectedly exacerbated the inflammatory response. Intriguingly, this paradoxical effect was linked to an upregulation of PPARδ. Functional analyses employing PPARδ modulation revealed its central role in regulating both lipid dynamics and inflammation, suggesting it as a potential therapeutic target. Moreover, primary monocytic cells from diabetic individuals, a demographic at amplified risk of TB, exhibited heightened PPARδ expression and inflammation, further underscoring its pathological relevance. Targeting PPARδ in these cells effectively dampened the inflammatory response, offering a promising therapeutic avenue against TB.
© 2024 The Author(s).
-
Immunology and Microbiology
Reduced monocyte proportions and responsiveness in convalescent COVID-19 patients.
In Frontiers in Immunology on 22 January 2024 by Ravkov, E. V., Williams, E. S. C. P., et al.
The clinical manifestations of acute severe acute respiratory syndrome coronavirus type 2 (SARS-CoV-2) infection and coronavirus disease 2019 (COVID-19) suggest a dysregulation of the host immune response that leads to inflammation, thrombosis, and organ dysfunction. It is less clear whether these dysregulated processes persist during the convalescent phase of disease or during long COVID. We sought to examine the effects of SARS-CoV-2 infection on the proportions of classical, intermediate, and nonclassical monocytes, their activation status, and their functional properties in convalescent COVID-19 patients.
Peripheral blood mononuclear cells (PBMCs) from convalescent COVID-19 patients and uninfected controls were analyzed by multiparameter flow cytometry to determine relative percentages of total monocytes and monocyte subsets. The expression of activation markers and proinflammatory cytokines in response to LPS treatment were measured by flow cytometry and ELISA, respectively.
We found that the percentage of total monocytes was decreased in convalescent COVID-19 patients compared to uninfected controls. This was due to decreased intermediate and non-classical monocytes. Classical monocytes from convalescent COVID-19 patients demonstrated a decrease in activation markers, such as CD56, in response to stimulation with bacterial lipopolysaccharide (LPS). In addition, classical monocytes from convalescent COVID-19 patients showed decreased expression of CD142 (tissue factor), which can initiate the extrinsic coagulation cascade, in response to LPS stimulation. Finally, we found that monocytes from convalescent COVID-19 patients produced less TNF-α and IL-6 in response to LPS stimulation, than those from uninfected controls.
SARS-CoV-2 infection exhibits a clear effect on the relative proportions of monocyte subsets, the activation status of classical monocytes, and proinflammatory cytokine production that persists during the convalescent phase of disease.
Copyright © 2024 Ravkov, Williams, Elgort, Barker, Planelles, Spivak, Delgado, Lin and Hanley.
-
FC/FACS
-
COVID-19
-
Immunology and Microbiology
In IScience on 21 July 2023 by Al-Rashed, F., Haddad, D., et al.
Foamy and inflammatory macrophages play pathogenic roles in metabolic disorders. However, the mechanisms that promote foamy and inflammatory macrophage phenotypes under acute-high-fat feeding (AHFF) remain elusive. Herein, we investigated the role of acyl-CoA synthetase-1 (ACSL1) in favoring the foamy/inflammatory phenotype of monocytes/macrophages upon short-term exposure to palmitate or AHFF. Palmitate exposure induced a foamy/inflammatory phenotype in macrophages which was associated with increased ACSL1 expression. Inhibition/knockdown of ACSL1 in macrophages suppressed the foamy/inflammatory phenotype through the inhibition of the CD36-FABP4-p38-PPARδ signaling axis. ACSL1 inhibition/knockdown suppressed macrophage foaming/inflammation after palmitate stimulation by downregulating the FABP4 expression. Similar results were obtained using primary human monocytes. As expected, oral administration of ACSL1 inhibitor triacsin-C in mice before AHFF normalized the inflammatory/foamy phenotype of the circulatory monocytes by suppressing FABP4 expression. Our results reveal that targeting ACSL1 leads to the attenuation of the CD36-FABP4-p38-PPARδ signaling axis, providing a therapeutic strategy to prevent the AHFF-induced macrophage foaming and inflammation.
© 2023 The Author(s).
-
Immunology and Microbiology
In Experimental and Therapeutic Medicine on 1 May 2023 by Ni, W., Deng, K., et al.
Acute promyelocytic leukemia (APL) during pregnancy is rare and difficult to treat. To the best of our knowledge, there is little precedent for successful treatment with combined chemotherapeutic agents without affecting delivery. The present study reported the case of a 31-year-old woman pregnant with twins who presented to the antenatal service at 13-week gestational age with complaints of vaginal bleeding, lower abdominal pain, bleeding gums and skin ecchymosis, and was eventually diagnosed with APL. After treatment with all-trans-retinoic acid (ATRA) and arsenic trioxide (ATO)-based induction regimen, the patient achieved a complete remission (CR) and delivered two healthy male infants at 34 weeks of gestation. The use of ATRA and ATO for the treatment of APL is controversial due to teratogenic effects and lethal retinoic acid syndrome. However, the patient demonstrated that the chemotherapy regimen with ATRA and ATO during the second and third trimesters can result in a sustainable remission and successful pregnancy outcome.
Copyright © 2020, Spandidos Publications.
-
Cancer Research
-
Endocrinology and Physiology
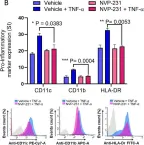
In Sci Rep on 15 April 2021 by Al-Rashed, F., Ahmad, Z., et al.
Fig.1.B

-
FC/FACS
-
Collected and cropped from Sci Rep by CiteAb, provided under a CC-BY license
Image 1 of 1