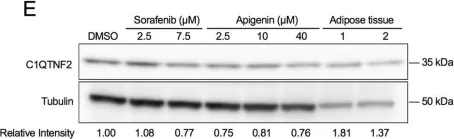
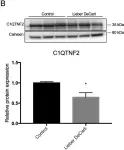
We have used a computational approach to identify anti-fibrotic therapies by querying a transcriptome. A transcriptome signature of activated hepatic stellate cells (HSCs), the primary collagen-secreting cell in liver, and queried against a transcriptomic database that quantifies changes in gene expression in response to 1,309 FDA-approved drugs and bioactives (CMap). The flavonoid apigenin was among 9 top-ranked compounds predicted to have anti-fibrotic activity; indeed, apigenin dose-dependently reduced collagen I in the human HSC line, TWNT-4. To identify proteins mediating apigenin's effect, we next overlapped a 122-gene signature unique to HSCs with a list of 160 genes encoding proteins that are known to interact with apigenin, which identified C1QTNF2, encoding for Complement C1q tumor necrosis factor-related protein 2, a secreted adipocytokine with metabolic effects in liver. To validate its disease relevance, C1QTNF2 expression is reduced during hepatic stellate cell activation in culture and in a mouse model of alcoholic liver injury in vivo, and its expression correlates with better clinical outcomes in patients with hepatitis C cirrhosis (n = 216), suggesting it may have a protective role in cirrhosis progression.These findings reinforce the value of computational approaches to drug discovery for hepatic fibrosis, and identify C1QTNF2 as a potential mediator of apigenin's anti-fibrotic activity.
Applications
Reactivity
Research Area
In Scientific Reports on 3 March 2017 by Hicks, D. F., Goossens, N., et al.
-
WB
-
Homo sapiens (Human)
CTRP2 overexpression improves insulin and lipid tolerance in diet-induced obese mice.
In PLoS ONE on 4 March 2014 by Peterson, J. M., Seldin, M. M., et al.
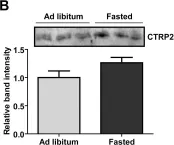
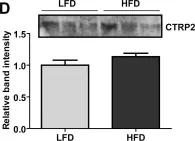
CTRP2 is a secreted plasma protein of the C1q family that enhances glycogen deposition and fat oxidation in cultured myotubes. Its in vivo metabolic function, however, has not been established. We show here that acute and chronic metabolic perturbations induced by fasting or high-fat feeding up-regulated the mRNA expression of Ctrp2 in white adipose tissue without affecting its circulating plasma levels. We generated a transgenic mouse model with elevated circulating levels of CTRP2 to determine its metabolic function in vivo. When fed a low-fat diet, wild-type and CTRP2 transgenic mice exhibited no metabolic phenotypes. When challenged with a high-fat diet to induce obesity, wild-type and CTRP2 transgenic mice had similar weight gain, adiposity, food intake, metabolic rate, and energy expenditure. Fasting serum lipid and adipokine profiles were also similar between the two groups of mice. However, while glucose and insulin levels in the fasted state were comparable between wild-type and CTRP2 transgenic mice, insulin levels in the fed state were consistently lower in transgenic mice. Notably, CTRP2 transgenic mice had improved insulin tolerance and a greater capacity to handle acute lipid challenge relative to littermate controls. Our results highlight, for the first time, the in vivo role of CTRP2 in modulating whole-body metabolism.
-
WB
-
Endocrinology and Physiology
In Sci Rep on 3 March 2017 by Hicks, D. F., Goossens, N., et al.
Fig.2.E
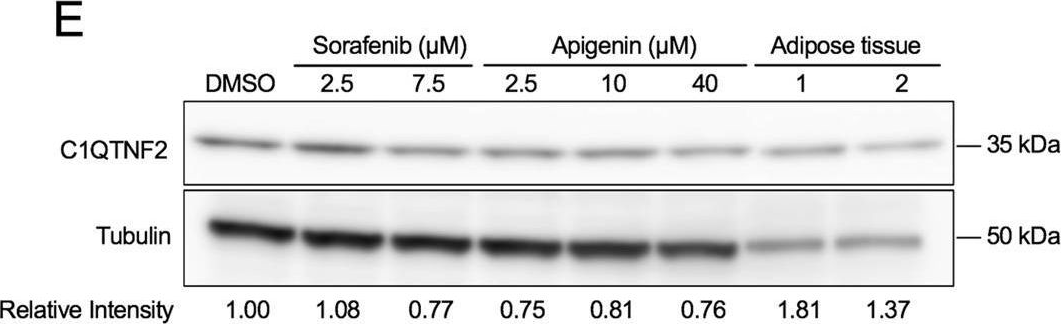
-
WB
-
Collected and cropped from Scientific Reports by CiteAb, provided under a CC-BY license
Image 1 of 4
In Sci Rep on 3 March 2017 by Hicks, D. F., Goossens, N., et al.
Fig.3.B
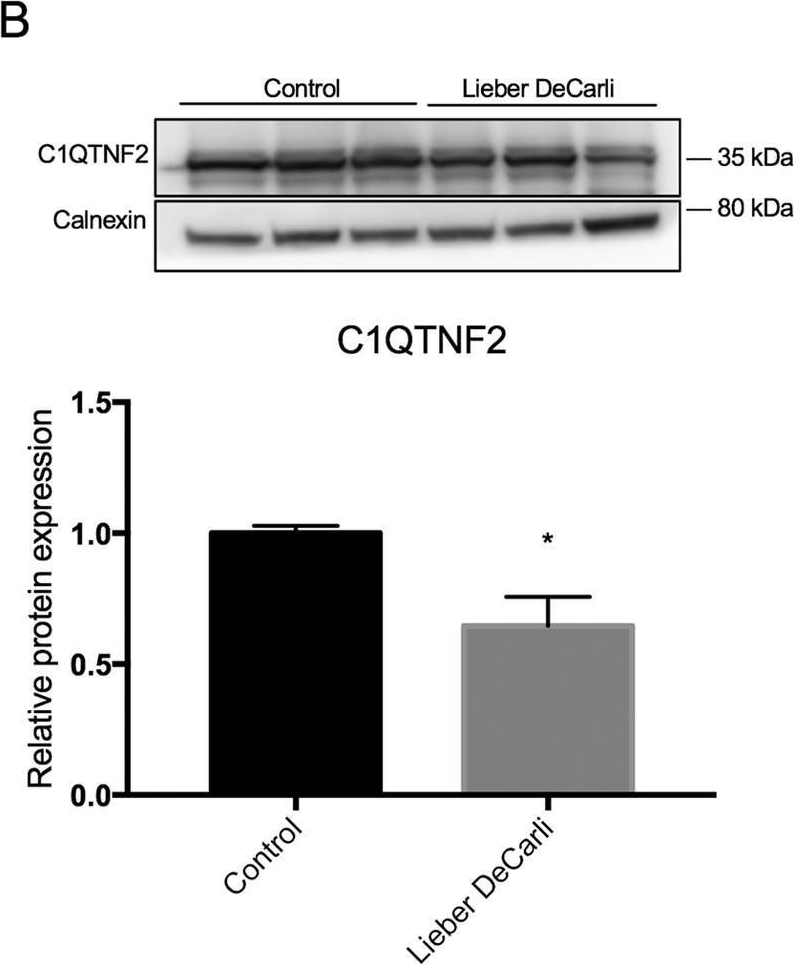
-
WB
-
Collected and cropped from Scientific Reports by CiteAb, provided under a CC-BY license
Image 1 of 4
In PLoS One on 4 March 2014 by Peterson, J. M., Seldin, M. M., et al.
Fig.1.B
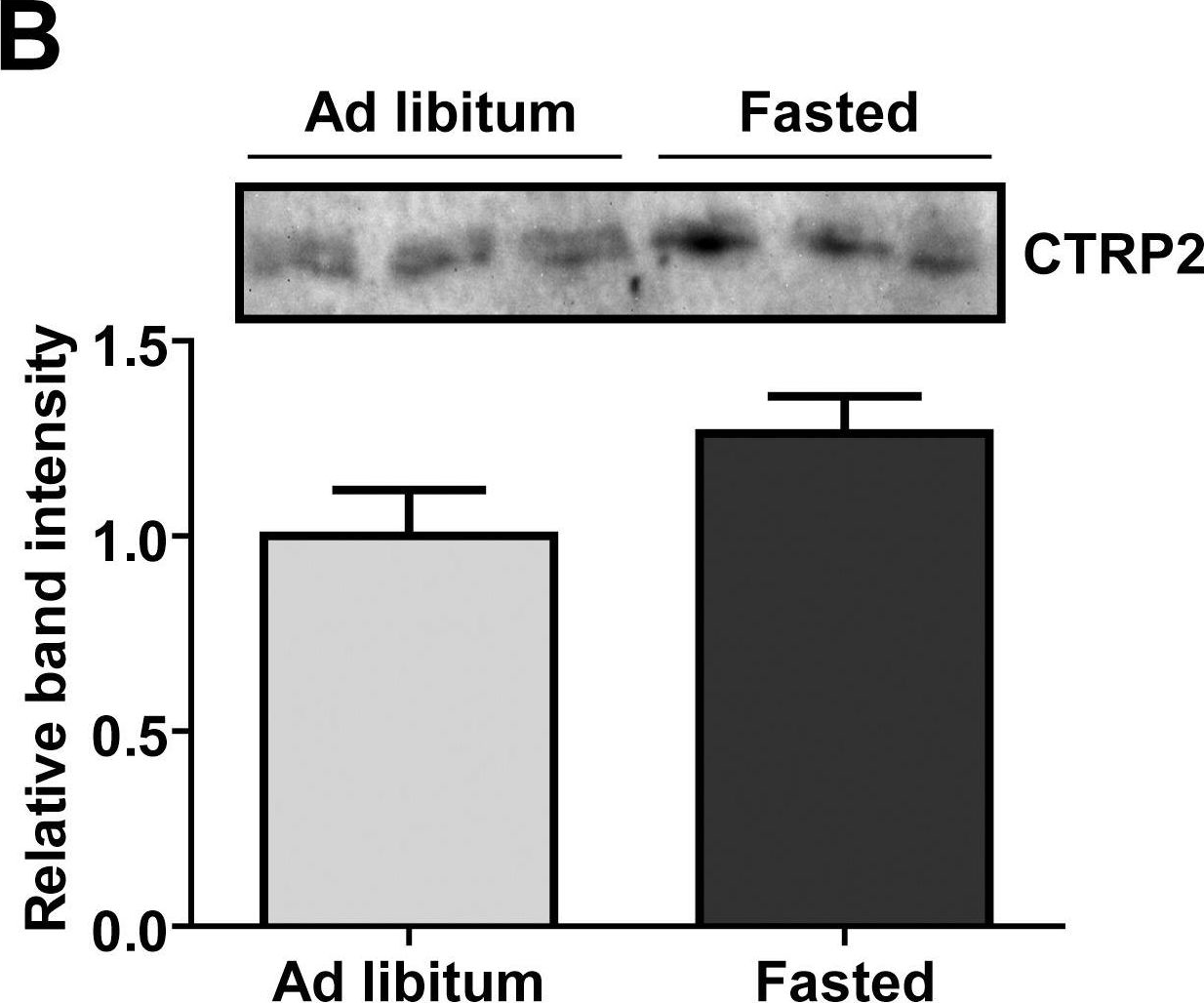
-
WB
-
Collected and cropped from PLoS ONE by CiteAb, provided under a CC-BY license
Image 1 of 4
In PLoS One on 4 March 2014 by Peterson, J. M., Seldin, M. M., et al.
Fig.1.D
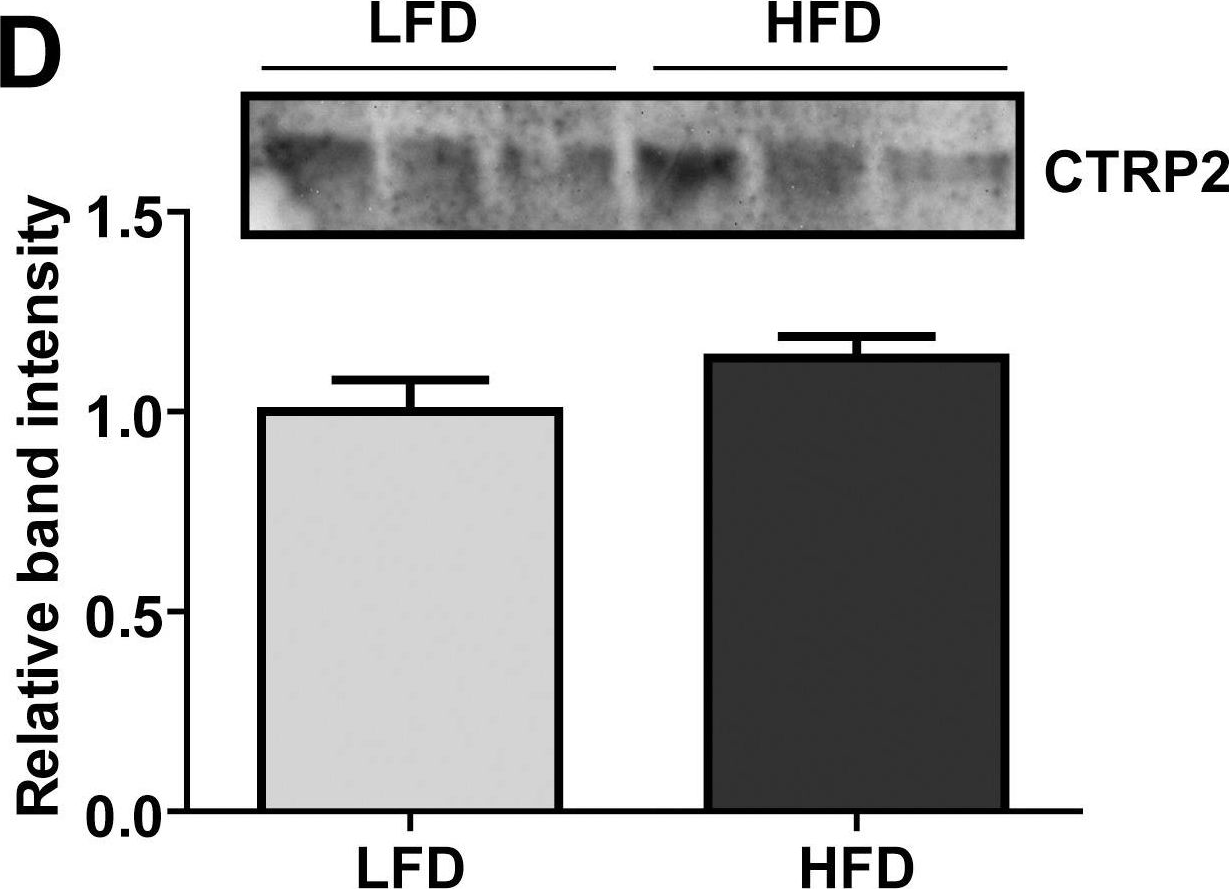
-
WB
-
Collected and cropped from PLoS ONE by CiteAb, provided under a CC-BY license
Image 1 of 4