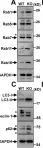
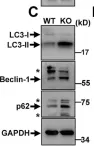
Genetic mutations of trappc9 cause intellectual disability with the atrophy of brain structures and variable obesity by poorly understood mechanisms. Trappc9-deficient mice develop phenotypes resembling pathological changes in humans and appear overweight shortly after weaning, and thus are useful for studying the pathogenesis of obesity. Here, we investigated the effects of trappc9 deficiency on the proliferation and differentiation capacity of adipose-derived stem cells (ASCs). We isolated ASCs from mice before overweight was developed and found that trappc9-null ASCs exhibited signs of premature senescence and cell death. While the lineage commitment was retained, trappc9-null ASCs preferred adipogenic differentiation. We observed a profound accumulation of lipid droplets in adipogenic cells derived from trappc9-deficient ASCs and marked differences in the distribution patterns and levels of calcium deposited in osteoblasts obtained from trappc9-null ASCs. Biochemical studies revealed that trappc9 deficiency resulted in an upregulated expression of rab1, rab11, and rab18, and agitated autophagy in ASCs. Moreover, we found that the content of neural stem cells in both the subventricular zone of the lateral ventricle and the subgranular zone of the dentate gyrus vastly declined in trappc9-null mice. Collectively, our results suggest that obesity, as well as brain structure hypoplasia induced by the deficiency of trappc9, involves an impairment in the plasticity of stem cells.
Applications
Reactivity
Research Area
Trappc9 Deficiency Impairs the Plasticity of Stem Cells.
In International Journal of Molecular Sciences on 28 April 2022 by Usman, M., Li, Y., et al.
-
WB
-
Mus musculus (House mouse)
-
Stem Cells and Developmental Biology
The cytoplasmic nuclear shuttling of Beclin 1 in neurons with Alzheimer's disease-like injury.
In Neuroscience Letters on 20 November 2017 by Wang, L., Xu, X. B., et al.
The abnormal expression of the autophagy-related protein Beclin 1 has been implicated in Alzheimer's disease (AD) brains, whereas the precise involvement of Caspase-mediated Beclin 1 cleavage in AD neurons has not yet been fully clarified. In this study, we investigated the distribution of Beclin 1 fragments in neurons with AD-like injury. Our results demonstrated that Beclin 1 was expressed in neurons but not in astrocytes in both neuron-glia co-cultures and in cortical tissue slices. The full length and C-terminal fragments of human Beclin 1 was mainly expressed in cytoplasm, while the N-terminal fragment of Beclin 1 was predominantly localized in nucleus. Compared to amyloid-β (Aβ)42-1 treatment control, exposure of PC12 cells or cortical neurons to Aβ1-42 resulted in cell injury, with the appearance of neuritic shortening, reduced nuclear diameter in PC12 cells, beading formation and fragmentation in cortical neurons. A partial nuclear translocation of Beclin 1 was detected in cells incubated with Aβ1-42, which could be inhibited by the administration of pan-Caspase inhibitor or Caspase 3 specific inhibitor. Moreover, Beclin 1 mutation at 146/149 sites was resistant to Aβ1-42-induced nuclear translocation. The nuclear translocation of Beclin 1 could also been detected in the brains of 12-month-old APPSwe/PS1dE9 transgenic mice. Our findings suggest that after Caspase 3-mediated Beclin 1 cleavage at 146/149 sites, the N-terminal fragments of Beclin 1 may partially translocate into nuclei in neurons subjected to AD-like injury.
Copyright © 2017 Elsevier B.V. All rights reserved.
-
Cell Biology
-
Neuroscience
In BMC Cancer on 9 May 2014 by Qiu, D. M., Wang, G. L., et al.
A role for autophagy, a conserved cellular response to stress, has recently been demonstrated in human cancers. Aberrant expression of Beclin-1, an important autophagic gene, has been reported in various human cancers. In the present study, we investigated the significance and relationship between Beclin-1 expression and cell proliferation, apoptosis, microvessel density (MVD) and clinical pathological changes or prognosis in human hepatocellular carcinoma (HCC).
A total of 103 primary HCC patients were involved in the study. Expression of Beclin-1, PCNA, NET-1, Bcl-2, Bax, Survivin in cancer cells and CD34 in stromal microvessels were evaluated immunohistochemically in tissue microarrays comprising 103 cases of HCC and 57 matched adjacent nontumor liver tissues. Correlations between clinicopathological characteristics and survival of HCC patients were explored.
The positive rate of Beclin-1 was significantly lower in HCC tissues than adjacent tissues (72.8 vs. 89.5%, χ2 = 6.085, P = 0.015). In HCC, Beclin-1 expression was negatively correlated with cirrhosis background (r = -0.216, P = 0.029), Edmondson grade (r = -0.249, P = 0.011), vascular invasion (r = -0.246, P = 0.012), PCNA (r = -0.242, P = 0.014), NET-1 (r = -0.245, P = 0.013), anti-apoptosis protein Bcl-2 (r = -0.245, P = 0.013) and MVD (r = -0.292, P = 0.003), and positively correlated with pro-apoptosis protein Bax (r = 0.242, P = 0.014).Significant differences in the 5-year survival rates were seen among patients with Beclin-1 strong positive (++) (59.1%, 13/22), moderate positive (+) (28.3%, 15/53) and weak negative expression (-) (14.6%, 7/28) (P = 0.043). Significant differences were detected between Beclin-1 (++) and either Beclin-1 (+) (P = 0.036) or Beclin-1 (-) groups (P = 0.008), but no significant difference between Beclin-1 (+) and Beclin-1 (-) groups (P = 0.281) was observed.Survival rates were positively related to high Beclin-1 co-expressed with low PCNA, NET-1, or Bcl-2, lower MVD, and high Bax. Univariate and multivariate Cox regression analysis revealed that Beclin-1 expression was an independent indicator for overall survival in HCC patients (P < 0.05).
The pathogenesis and progression of HCC are associated with reduced autophagy. The expression of Beclin-1 and Bax in HCC tissues may provide a synergistic effect towards inhibiting HCC proliferation, infiltration, metastasis and angiogenesis. Beclin-1 expression may be a valuable prognostic marker of HCC.
-
Cancer Research
In PLoS ONE on 13 August 2013 by Yu, Y., Shiou, S. R., et al.
Neonatal necrotizing enterocolitis (NEC) is a devastating gastrointestinal disease of preterm infants. Increased intestinal epithelium permeability is an early event in NEC pathogenesis. Autophagy and apoptosis are induced by multiple stress pathways which may impact the intestinal barrier, and they have been associated with pathogenesis of diverse gastrointestinal diseases including inflammatory bowel disease. Using both in vitro and in vivo models, this study investigates autophagy and apoptosis under experimental NEC stresses. Furthermore this study evaluates the effect of erythropoietin (Epo), a component of breast milk previously shown to decrease the incidence of NEC and to preserve intestinal barrier function, on intestinal autophagy and apoptosis. It was found that autophagy and apoptosis are both rapidly up regulated in NEC in vivo as indicated by increased expression of the autophagy markers Beclin 1 and LC3II, and by evidence of apoptosis by TUNEL and cleaved caspase-3 staining. In the rat NEC experimental model, autophagy preceded the onset of apoptosis in intestine. In vitro studies suggested that Epo supplementation significantly decreased both autophagy and apoptosis via the Akt/mTOR signaling pathway and the MAPK/ERK pathway respectively. These results suggest that Epo protects intestinal epithelium from excessive autophagy and apoptosis in experimental NEC.
-
Cell Biology
-
Endocrinology and Physiology
In Breast Cancer Research and Treatment on 1 November 2011 by Thomas, S., Thurn, K. T., et al.
Modulation of estrogen signaling is one of the most successful modalities for the treatment of estrogen receptor (ER)-positive breast cancer, yet de novo and acquired resistance are frequent. Recent data suggests that the induction of autophagy may play a considerable role in promoting tumor cell survival and resistance to anti-estrogen therapy. Hence, bypassing autophagy may offer a novel strategy to enhance the anti-tumor efficacy of anti-estrogens. Histone deacetylases (HDAC) are involved in the regulation of steroid hormone receptor mediated cell signaling and their inhibition potentiates the anti-tumor effects of anti-estrogens. However, the mechanism underlying this anti-tumor activity is poorly understood. In this report, we show that the addition of an HDAC inhibitor redirects the response of ER-positive breast cancer cells when treated with tamoxifen from growth arrest to apoptotic cell death. This redirection requires functional ER signaling and is mediated by a depletion of Bcl-2 and an induction of Bax and Bak, manifesting in cytochrome C release and PARP cleavage. With combined treatment, a subpopulation of cells is refractory to apoptosis and exhibit a strong induction of autophagy. Inhibition of autophagy in these cells, using siRNA directed against Beclin-1 or treatment with chloroquine, further promotes the induction of apoptosis. Thus, supporting prior reports that autophagy acts as a survival mechanism, our findings demonstrate that HDAC and autophagy inhibition directs autophagy-protected cells into apoptotic cell death, which may impair development of tamoxifen resistance.
-
Cancer Research
-
Cell Biology
-
Genetics
In Int J Mol Sci on 28 April 2022 by Usman, M., Li, Y., et al.
Fig.4.A
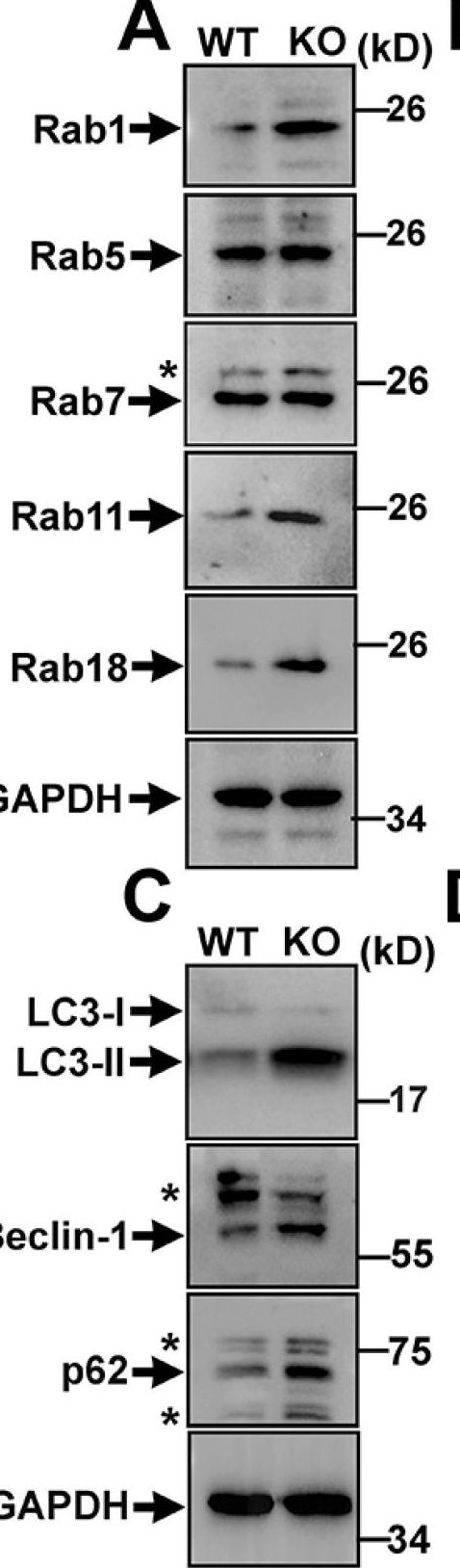
-
WB
-
Mus musculus (House mouse)
Collected and cropped from International Journal of Molecular Sciences by CiteAb, provided under a CC-BY license
Image 1 of 2
In Int J Mol Sci on 28 April 2022 by Usman, M., Li, Y., et al.
Fig.4.C
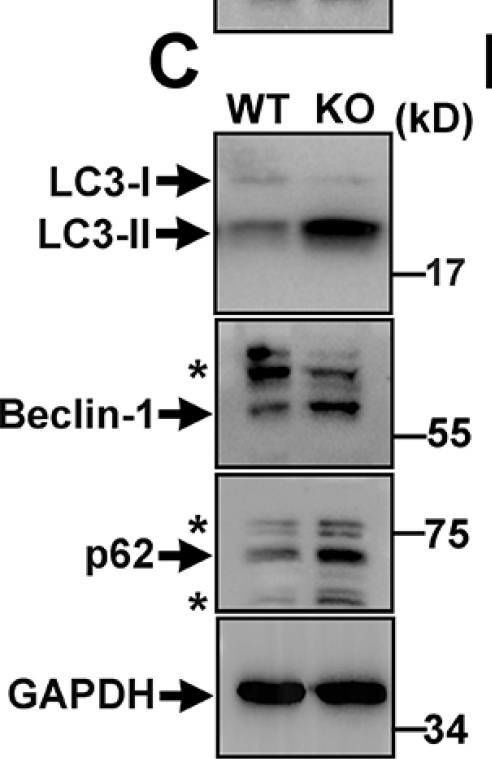
-
WB
-
Collected and cropped from International Journal of Molecular Sciences by CiteAb, provided under a CC-BY license
Image 1 of 2