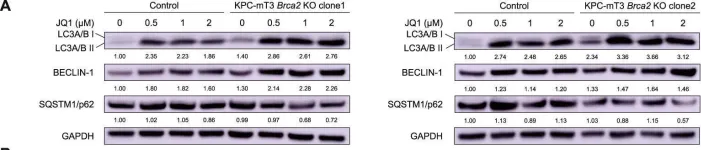
Pancreatic cancer is one of the deadliest diseases in human malignancies. Among total pancreatic cancer patients, ~10% of patients are categorized as familial pancreatic cancer (FPC) patients, carrying germline mutations of the genes involved in DNA repair pathways (e.g., BRCA2). Personalized medicine approaches tailored toward patients' mutations would improve patients' outcome. To identify novel vulnerabilities of BRCA2-deficient pancreatic cancer, we generated isogenic Brca2-deficient murine pancreatic cancer cell lines and performed high-throughput drug screens. High-throughput drug screening revealed that Brca2-deficient cells are sensitive to Bromodomain and Extraterminal Motif (BET) inhibitors, suggesting that BET inhibition might be a potential therapeutic approach. We found that BRCA2 deficiency increased autophagic flux, which was further enhanced by BET inhibition in Brca2-deficient pancreatic cancer cells, resulting in autophagy-dependent cell death. Our data suggests that BET inhibition can be a novel therapeutic strategy for BRCA2-deficient pancreatic cancer.
© 2023. The Author(s).
Applications
Reactivity
Research Area
In Cell Death & Disease on 21 September 2023 by Lee, E., Archasappawat, S., et al.
-
WB
-
Mus musculus (House mouse)
-
Cancer Research
-
Cell Biology
In Blood on 2 March 2023 by Yu, X., Ma, H., et al.
Intestinal epithelial cells (IECs) are implicated in the propagation of T-cell-mediated inflammatory diseases, including graft-versus-host disease (GVHD), but the underlying mechanism remains poorly defined. Here, we report that IECs require receptor-interacting protein kinase-3 (RIPK3) to drive both gastrointestinal (GI) tract and systemic GVHD after allogeneic hematopoietic stem cell transplantation. Selectively inhibiting RIPK3 in IECs markedly reduces GVHD in murine intestine and liver. IEC RIPK3 cooperates with RIPK1 to trigger mixed lineage kinase domain-like protein-independent production of T-cell-recruiting chemokines and major histocompatibility complex (MHC) class II molecules, which amplify and sustain alloreactive T-cell responses. Alloreactive T-cell-produced interferon gamma enhances this RIPK1/RIPK3 action in IECs through a JAK/STAT1-dependent mechanism, creating a feed-forward inflammatory cascade. RIPK1/RIPK3 forms a complex with JAK1 to promote STAT1 activation in IECs. The RIPK1/RIPK3-mediated inflammatory cascade of alloreactive T-cell responses results in intestinal tissue damage, converting the local inflammation into a systemic syndrome. Human patients with severe GVHD showed highly activated RIPK1 in the colon epithelium. Finally, we discover a selective and potent RIPK1 inhibitor (Zharp1-211) that significantly reduces JAK/STAT1-mediated expression of chemokines and MHC class II molecules in IECs, restores intestinal homeostasis, and arrests GVHD without compromising the graft-versus-leukemia (GVL) effect. Thus, targeting RIPK1/RIPK3 in IECs represents an effective nonimmunosuppressive strategy for GVHD treatment and potentially for other diseases involving GI tract inflammation.
© 2023 by The American Society of Hematology. Licensed under Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0), permitting only noncommercial, nonderivative use with attribution. All other rights reserved.
-
Cardiovascular biology
PEX11β and FIS1 cooperate in peroxisome division independently of mitochondrial fission factor.
In Journal of Cell Science on 1 July 2022 by Schrader, T. A., Carmichael, R. E., et al.
Peroxisome membrane dynamics and division are essential to adapt the peroxisomal compartment to cellular needs. The peroxisomal membrane protein PEX11β (also known as PEX11B) and the tail-anchored adaptor proteins FIS1 (mitochondrial fission protein 1) and MFF (mitochondrial fission factor), which recruit the fission GTPase DRP1 (dynamin-related protein 1, also known as DNML1) to both peroxisomes and mitochondria, are key factors of peroxisomal division. The current model suggests that MFF is essential for peroxisome division, whereas the role of FIS1 is unclear. Here, we reveal that PEX11β can promote peroxisome division in the absence of MFF in a DRP1- and FIS1-dependent manner. We also demonstrate that MFF permits peroxisome division independently of PEX11β and restores peroxisome morphology in PEX11β-deficient patient cells. Moreover, targeting of PEX11β to mitochondria induces mitochondrial division, indicating the potential for PEX11β to modulate mitochondrial dynamics. Our findings suggest the existence of an alternative, MFF-independent pathway in peroxisome division and report a function for FIS1 in the division of peroxisomes. This article has an associated First Person interview with the first authors of the paper.
© 2022. Published by The Company of Biologists Ltd.
-
Cell Biology
Degradation of Blos1 mRNA by IRE1 repositions lysosomes and protects cells from stress.
In The Journal of Cell Biology on 1 April 2019 by Bae, D., Moore, K. A., et al.
Cells respond to stress in the ER by initiating the widely conserved unfolded protein response. Activation of the ER transmembrane nuclease IRE1 leads to the degradation of specific mRNAs, but how this pathway affects the ability of cells to recover from stress is not known. Here, we show that degradation of the mRNA encoding biogenesis of lysosome-related organelles 1 subunit 1 (Blos1) leads to the repositioning of late endosomes (LEs)/lysosomes to the microtubule-organizing center in response to stress in mouse cells. Overriding Blos1 degradation led to ER stress sensitivity and the accumulation of ubiquitinated protein aggregates, whose efficient degradation required their independent trafficking to the cell center and the LE-associated endosomal sorting complexes required for transport. We propose that Blos1 regulation by IRE1 promotes LE-mediated microautophagy of protein aggregates and protects cells from their cytotoxic effects.
© 2019 Bae et al.
-
WB
-
Cell Biology
-
Genetics
Novel carbazole inhibits phospho-STAT3 through induction of protein-tyrosine phosphatase PTPN6.
In Journal of Medicinal Chemistry on 14 August 2014 by Hou, S., Yi, Y. W., et al.
The aberrant activation of STAT3 occurs in many human cancers and promotes tumor progression. Phosphorylation of a tyrosine at amino acid Y705 is essential for the function of STAT3. Synthesized carbazole derived with fluorophore compound 12 was discovered to target STAT3 phosphorylation. Compound 12 was found to inhibit STAT3-mediated transcription as well as to reduce IL-6 induced STAT3 phosphorylation in cancer cell lines expressing both elevated and low levels of phospho-STAT3 (Y705). Compound 12 potently induced apoptosis in a broad number of TNBC cancer cell lines in vitro and was effective at inhibiting the in vivo growth of human TNBC xenograft tumors (SUM149) without any observed toxicity. Compound 12 also effectively inhibited the growth of human lung tumor xenografts (A549) harboring aberrantly active STAT3. In vitro and in vivo studies showed that the inhibitory effects of 12 on phospho-STAT3 were through up-regulation of the protein-tyrosine phosphatase PTPN6. Our present studies strongly support the continued preclinical evaluation of compound 12 as a potential chemotherapeutic agent for TNBC and cancers with constitutive STAT3 signaling.
-
WB
-
Chemistry
In Cell Death Dis on 21 September 2023 by Lee, E., Archasappawat, S., et al.
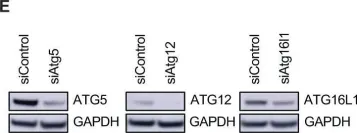
Fig.6.E
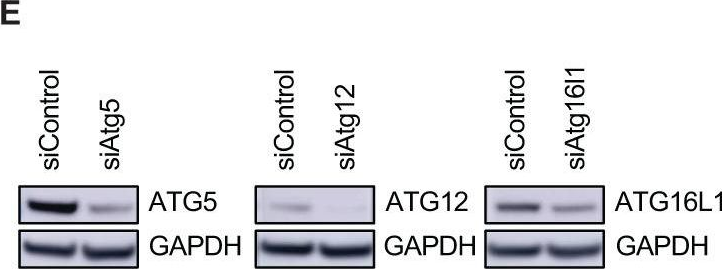
-
WB
-
Collected and cropped from Cell Death & Disease by CiteAb, provided under a CC-BY license
Image 1 of 2
In Cell Death Dis on 21 September 2023 by Lee, E., Archasappawat, S., et al.
Fig.5.A
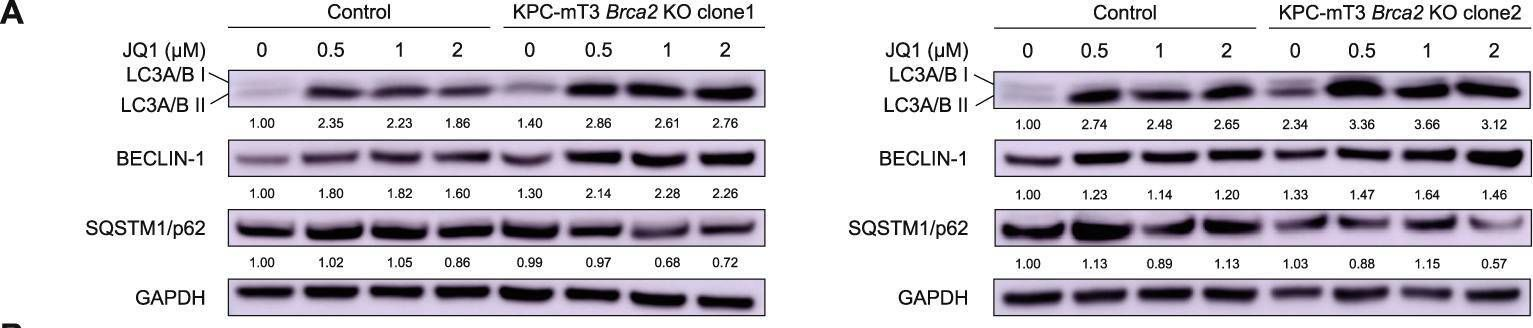
-
WB
-
Mus musculus (House mouse)
Collected and cropped from Cell Death & Disease by CiteAb, provided under a CC-BY license
Image 1 of 2