The oxidative stress, induced by both environmental and intrinsic stimuli, underlies the onset and persistency of chronic rhinosinusitis (CRS). Scavenger receptors (SRs) are a broad family of transmembrane receptors involved in a dysfunctional host-environment interaction through a reaction with reactive oxygen species (ROS) production.
We hypothesized possible roles of two major SRs in CRS pathology that can translate to clinical phenotypes or histological subtypes: lectin-like oxidized low-density lipoproteins (LDL) receptor-1 (LOX-1) and scavenger receptor class B type 1 (SR-B1).
We collected ethmoid sinus mucosa specimens and blood samples from patients with CRS with nasal polyps (CRSwNP; n = 31) or CRS without NP (CRSsNP; n = 13) and 19 control subjects. We performed an RT-PCR analysis, ELISA assay, and immunostaining to determine the expressions and distributions of LOX-1 and SR-B1.
The CRSwNP group showed a significant increase in LOX-1 mRNA expression compared to the control group. There was no significant difference in SR-B1 mRNA levels among the three groups. The LOX-1 mRNA levels were positively correlated with the sinus computed tomography (CT) scores. Sinus tissue, but not serum samples, showed elevated concentrations of LOX-1 protein in the CRSwNP group versus the control group. The LOX-1 protein distribution was localized in inflammatory cells and vascular endothelial cells.
LOX-1 is a major receptor for oxidized low-density lipoprotein produced by oxidative stress. This is the first study to report alterations in LOX-1 expression and production triggered by persistent inflammatory processes in CRSwNP patients. Our findings reveal complex but important roles for SRs that may contribute to the onset of different CRS phenotypes.
Applications
Reactivity
Research Area
None Available
In Diagnostics (Basel, Switzerland) on 23 April 2020 by Nishida, M., Takeno, S., et al.
-
IHC
-
Homo sapiens (Human)
Open-gate mutants of the mammalian proteasome show enhanced ubiquitin-conjugate degradation.
In Nature Communications on 9 March 2016 by Choi, W. H., de Poot, S. A., et al.
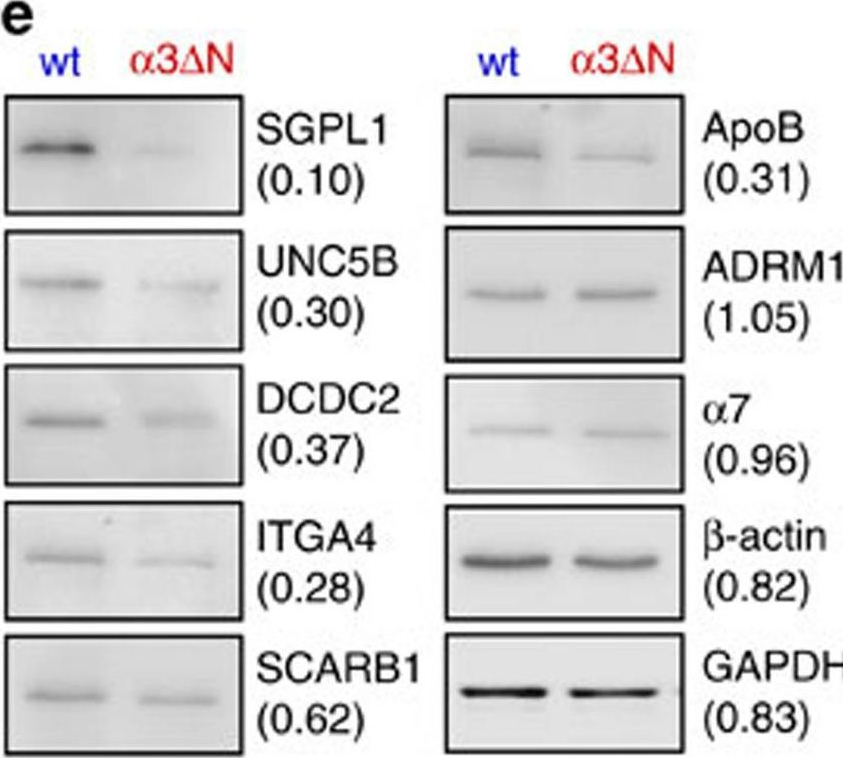
When in the closed form, the substrate translocation channel of the proteasome core particle (CP) is blocked by the convergent N termini of α-subunits. To probe the role of channel gating in mammalian proteasomes, we deleted the N-terminal tail of α3; the resulting α3ΔN proteasomes are intact but hyperactive in the hydrolysis of fluorogenic peptide substrates and the degradation of polyubiquitinated proteins. Cells expressing the hyperactive proteasomes show markedly elevated degradation of many established proteasome substrates and resistance to oxidative stress. Multiplexed quantitative proteomics revealed ∼ 200 proteins with reduced levels in the mutant cells. Potentially toxic proteins such as tau exhibit reduced accumulation and aggregate formation. These data demonstrate that the CP gate is a key negative regulator of proteasome function in mammals, and that opening the CP gate may be an effective strategy to increase proteasome activity and reduce levels of toxic proteins in cells.
-
WB
-
Homo sapiens (Human)
In Nat Commun on 9 March 2016 by Choi, W. H., de Poot, S. A., et al.
Fig.5.E
-
WB
-
Collected and cropped from Nature Communications by CiteAb, provided under a CC-BY license
Image 1 of 1