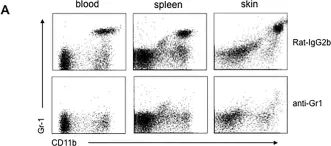
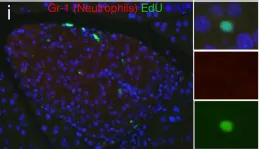
The severity of spinal cord injury (SCI) is closely tied to pulmonary function, especially in cases of higher SCI levels. Despite this connection, the underlying pathological mechanisms in the lungs post-SCI are not well understood. Previous research has established a connection between disrupted sympathetic regulation and splenocyte apoptosis in high thoracic SCI, leading to pulmonary dysfunction. The aim of this study was to investigate whether mice with low-level SCI exhibit increased susceptibility to acute lung injury by eliciting systemic inflammatory responses that operate independently of the sympathetic nervous system.
Here, we employed T9 contusion SCI and exposed mice to aerosolized lipopolysaccharide (LPS) to simulate lung inflammation associated with acute respiratory distress syndrome (ARDS). Twenty-four hours post-LPS exposure, lung tissues and bronchoalveolar lavage (BAL) fluid were analyzed.
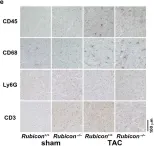
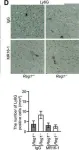
LPS markedly induced proinflammatory gene expression (SAA3, IRG1, NLRP3, IL-1beta, MCP-1) and cytokine release (IL-1beta, IL-6, MCP-1) in SCI mice compared to controls, indicating an exaggerated inflammatory response. Infiltration of Ly6G/C positive neutrophils and macrophages was significantly higher in SCI mice lungs post-LPS exposure. Interestingly, spleen size and weight did not differ between control and SCI mice, suggesting that T9 SCI alone does not cause spleen atrophy. Notably, bone-marrow-derived macrophages (BMDMs) from SCI mice exhibited hyper-responsiveness to LPS.
This study demonstrated an increase in lung inflammation and immune responses subsequent to low-level T9 SCI, underscoring the widespread influence of systemic inflammation post-SCI, especially pronounced in specific organs like the lungs.
Copyright © 2025 Berk, Pereira, Vizcarra, Pröschel and Hsu.
Product Citations: 368
In Frontiers in Immunology on 30 January 2025 by Berk, B. C., Pereira, A., et al.
-
IHC-IF
-
Mus musculus (House mouse)
-
Immunology and Microbiology
-
Neuroscience
Protective Role of MAVS Signaling for Murine Lipopolysaccharide-Induced Acute Kidney Injury.
In ImmunoHorizons on 1 January 2024 by Tran, T. A. T., Iwata, Y., et al.
Despite treatment advances, acute kidney injury (AKI)-related mortality rates are still high in hospitalized adults, often due to sepsis. Sepsis and AKI could synergistically worsen the outcomes of critically ill patients. TLR4 signaling and mitochondrial antiviral signaling protein (MAVS) signaling are innate immune responses essential in kidney diseases, but their involvement in sepsis-associated AKI (SA-AKI) remains unclear. We studied the role of MAVS in kidney injury related to the TLR4 signaling pathway using a murine LPS-induced AKI model in wild-type and MAVS-knockout mice. We confirmed the importance of M1 macrophage in SA-AKI through in vivo assessment of inflammatory responses. The TLR4 signaling pathway was upregulated in activated bone marrow-derived macrophages, in which MAVS helped maintain the LPS-suppressed TLR4 mRNA level. MAVS regulated redox homeostasis via NADPH oxidase Nox2 and mitochondrial reverse electron transport in macrophages to alleviate the TLR4 signaling response to LPS. Hypoxia-inducible factor 1α (HIF-1α) and AP-1 were key regulators of TLR4 transcription and connected MAVS-dependent reactive oxygen species signaling with the TLR4 pathway. Inhibition of succinate dehydrogenase could partly reduce inflammation in LPS-treated bone marrow-derived macrophages without MAVS. These findings highlight the renoprotective role of MAVS in LPS-induced AKI by regulating reactive oxygen species generation-related genes and maintaining redox balance. Controlling redox homeostasis through MAVS signaling may be a promising therapy for SA-AKI.
Copyright © 2024 The Authors.
-
Mus musculus (House mouse)
In Proceedings of the National Academy of Sciences of the United States of America on 26 December 2023 by Cheng, Q., Farbiak, L., et al.
Genetic medicines have the potential to treat various diseases; however, certain ailments including inflammatory diseases and cancer would benefit from control over extracellular localization of therapeutic proteins. A critical gap therefore remains the need to develop and incorporate methodologies that allow for posttranslational control over expression dynamics, localization, and stability of nucleic acid-generated protein therapeutics. To address this, we explored how the body's endogenous machinery controls protein localization through signal peptides (SPs), including how these motifs could be incorporated modularly into therapeutics. SPs serve as a virtual zip code for mRNA transcripts that direct the cell where to send completed proteins within the cell and the body. Utilizing this signaling biology, we incorporated secretory SP sequences upstream of mRNA transcripts coding for reporter, natural, and therapeutic proteins to induce secretion of the proteins into systemic circulation. SP sequences generated secretion of various engineered proteins into the bloodstream following intravenous, intramuscular, and subcutaneous SP mRNA delivery by lipid, polymer, and ionizable phospholipid delivery carriers. SP-engineered etanercept/TNF-α inhibitor proteins demonstrated therapeutic efficacy in an imiquimod-induced psoriasis model by reducing hyperkeratosis and inflammation. An SP-engineered anti-PD-L1 construct mediated mRNA encoded proteins with longer serum half-lives that reduced tumor burden and extended survival in MC38 and B16F10 cancer models. The modular nature of SP platform should enable intracellular and extracellular localization control of various functional proteins for diverse therapeutic applications.
-
Mus musculus (House mouse)
-
Cancer Research
Preprint on Research Square on 8 August 2023 by Albanesi, C., Mercurio, L., et al.
Psoriasis is a chronic immune-mediated skin disease in which upper epidermal keratinocytes exhibit a senescent-like phenotype. In psoriatic skin, a variety of inflammatory cytokines can activate intracellular pathways including phosphatidylinositol 3-kinase (PI3K)/AKT signaling and RAS effectors. AKT and RAS participate to cellular senescence, but currently their role in senescence responses occurring in psoriasis have not yet been investigated. Here, we found AKT hyperactivation associated to the upregulation of senescence markers in psoriatic keratinocyte cultures subjected to multiple passages to promote senescence in vitro , as well as in skin lesions of patients affected by psoriasis. AKT-induced senescence was sustained by constitutive RAS activation, and PI3K/AKT pharmacological inhibition contrasted senescence processes induced by cytokines in psoriatic keratinocyte cultures. Additionally, RAS overexpression in the suprabasal epidermis in a murine model of psoriasis was accompanied by AKT upregulation, increase of senescence marker expression and by skin inflammation. In this model, both senescence and inflammation were significantly reduced by selective AKT inhibition. Therefore, targeting RAS-AKT pathway could be a promising novel strategy to counteract multiple psoriasis symptoms.
-
IHC
-
Mus musculus (House mouse)
In Nature Communications on 31 July 2023 by Sugihara, R., Taneike, M., et al.
Heart failure is a leading cause of mortality in developed countries. Cell death is a key player in the development of heart failure. Calcium-independent phospholipase A2β (iPLA2β) produces lipid mediators by catalyzing lipids and induces nuclear shrinkage in caspase-independent cell death. Here, we show that lysophosphatidylserine generated by iPLA2β induces necrotic cardiomyocyte death, as well as contractile dysfunction mediated through its receptor, G protein-coupled receptor 34 (GPR34). Cardiomyocyte-specific iPLA2β-deficient male mice were subjected to pressure overload. While control mice showed left ventricular systolic dysfunction with necrotic cardiomyocyte death, iPLA2β-deficient mice preserved cardiac function. Lipidomic analysis revealed a reduction of 18:0 lysophosphatidylserine in iPLA2β-deficient hearts. Knockdown of Gpr34 attenuated 18:0 lysophosphatidylserine-induced necrosis in neonatal male rat cardiomyocytes, while the ablation of Gpr34 in male mice reduced the development of pressure overload-induced cardiac remodeling. Thus, the iPLA2β-lysophosphatidylserine-GPR34-necrosis signaling axis plays a detrimental role in the heart in response to pressure overload.
© 2023. The Author(s).
-
IHC
-
Mus musculus (House mouse)
In Sci Rep on 7 January 2022 by Akazawa, Y., Taneike, M., et al.
Fig.2.E

-
IHC
-
Rattus norvegicus (Rat)
Collected and cropped from Sci Rep by CiteAb, provided under a CC-BY license
Image 1 of 6
In Circulation on 25 February 2020 by Omiya, S., Omori, Y., et al.
Fig.5.D

-
IHC
-
Collected and cropped from Circulation by CiteAb, provided under a CC-BY license
Image 1 of 6
In Int J Mol Sci on 5 December 2019 by Liu, N., Jiang, Y., et al.
Fig.2.C

-
WB
-
Collected and cropped from Int J Mol Sci by CiteAb, provided under a CC-BY license
Image 1 of 6
In Front Immunol on 5 November 2019 by Zhang, F., Zhao, Q., et al.
Fig.2.A

-
IHC-IF
-
Mus musculus (House mouse)
Collected and cropped from Front Immunol by CiteAb, provided under a CC-BY license
Image 1 of 6
In PLoS One on 13 January 2017 by Jiang, X., Park, C. O., et al.
Fig.6.A

-
FC/FACS
-
Collected and cropped from PLoS One by CiteAb, provided under a CC-BY license
Image 1 of 6
In PLoS One on 15 July 2016 by Cox, A. R., Barrandon, O., et al.
Fig.9.I

-
WB
-
Mus musculus (House mouse)
Collected and cropped from PLoS One by CiteAb, provided under a CC-BY license
Image 1 of 6