While young blood can restore many aged tissues, its effects on the aged blood system itself and old hematopoietic stem cells (HSCs) have not been determined. Here, we used transplantation, parabiosis, plasma transfer, exercise, calorie restriction, and aging mutant mice to understand the effects of age-regulated systemic factors on HSCs and their bone marrow (BM) niche. We found that neither exposure to young blood, nor long-term residence in young niches after parabiont separation, nor direct heterochronic transplantation had any observable rejuvenating effects on old HSCs. Likewise, exercise and calorie restriction did not improve old HSC function, nor old BM niches. Conversely, young HSCs were not affected by systemic pro-aging conditions, and HSC function was not impacted by mutations influencing organismal aging in established long-lived or progeroid genetic models. Therefore, the blood system that carries factors with either rejuvenating or pro-aging properties for many other tissues is itself refractory to those factors.© 2021 Ho et al.
Product Citations: 14
Aged hematopoietic stem cells are refractory to bloodborne systemic rejuvenation interventions.
In The Journal of Experimental Medicine on 5 July 2021 by Ho, T. T., Dellorusso, P. V., et al.
-
FC/FACS
-
Mus musculus (House mouse)
-
Stem Cells and Developmental Biology
Rap1 and membrane lipids cooperatively recruit talin to trigger integrin activation.
In Journal of Cell Science on 1 November 2019 by Bromberger, T., Zhu, L., et al.
Recruitment and tethering of talin to the plasma membrane initiate the process of integrin activation. Multiple factors including the Rap1 proteins, RIAM (also known as APBB1IP) and PIP2 bind talin proteins and have been proposed to regulate these processes, but not systematically analyzed. By expressing specific talin mutants into talin-null fibroblasts, we show that binding of the talin F0 domain to Rap1 synergizes with membrane lipid binding of the talin F2 domain during talin membrane targeting and integrin activation, whereas the interaction of the talin rod with RIAM was dispensable. We also characterized a second Rap1-binding site within the talin F1 domain by detailed NMR analysis. Interestingly, while talin F1 exhibited significantly weaker Rap1-binding affinity than talin F0, expression of a talin F1 Rap1-binding mutant inhibited cell adhesion, spreading, talin recruitment and integrin activation similarly to the talin F0 Rap1-binding mutant. Moreover, the defects became significantly stronger when both Rap1-binding sites were mutated. In conclusion, our data suggest a model in which cooperative binding of Rap1 to the talin F0 and F1 domains synergizes with membrane PIP2 binding to spatiotemporally position and activate talins to regulate integrin activity.
© 2019. Published by The Company of Biologists Ltd.
-
Mus musculus (House mouse)
-
Cell Biology
The cis-Regulatory Atlas of the Mouse Immune System.
In Cell on 7 February 2019 by Yoshida, H., Lareau, C. A., et al.
A complete chart of cis-regulatory elements and their dynamic activity is necessary to understand the transcriptional basis of differentiation and function of an organ system. We generated matched epigenome and transcriptome measurements in 86 primary cell types that span the mouse immune system and its differentiation cascades. This breadth of data enable variance components analysis that suggests that genes fall into two distinct classes, controlled by either enhancer- or promoter-driven logic, and multiple regression that connects genes to the enhancers that regulate them. Relating transcription factor (TF) expression to the genome-wide accessibility of their binding motifs classifies them as predominantly openers or closers of local chromatin accessibility, pinpointing specific cis-regulatory elements where binding of given TFs is likely functionally relevant, validated by chromatin immunoprecipitation sequencing (ChIP-seq). Overall, this cis-regulatory atlas provides a trove of information on transcriptional regulation through immune differentiation and a foundational scaffold to define key regulatory events throughout the immunological genome.
Copyright © 2018 Elsevier Inc. All rights reserved.
-
FC/FACS
-
Mus musculus (House mouse)
-
Immunology and Microbiology
iRGD-guided Tumor-penetrating Nanocomplexes for Therapeutic siRNA Delivery to Pancreatic Cancer.
In Molecular Cancer Therapeutics on 1 November 2018 by Lo, J. H., Hao, L., et al.
Pancreatic cancer is one of the leading causes of cancer-related death, with 5-year survival of 8.5%. The lack of significant progress in improving therapy reflects our inability to overcome the desmoplastic stromal barrier in pancreatic ductal adenocarcinoma (PDAC) as well as a paucity of new approaches targeting its genetic underpinnings. RNA interference holds promise in targeting key mutations driving PDAC; however, a nucleic acid delivery vehicle that homes to PDAC and breaches the stroma does not yet exist. Noting that the cyclic peptide iRGD mediates tumor targeting and penetration through interactions with αvβ3/5 integrins and neuropilin-1, we hypothesized that "tandem" peptides combining a cell-penetrating peptide and iRGD can encapsulate siRNA to form tumor-penetrating nanocomplexes (TPN) capable of delivering siRNA to PDAC. The use of directly conjugated iRGD is justified by receptor expression patterns in human PDAC biopsies. In this work, we optimize iRGD TPNs with polyethylene glycol (PEG)-peptide conjugates for systemic delivery to sites of disease. We show that TPNs effectively knockdown siRNA targets in PDAC cell lines and in an immunocompetent genetically engineered mouse model of PDAC. Furthermore, we validate their tumor-penetrating ability in three-dimensional organoids and autochthonous tumors. In murine therapeutic trials, TPNs delivering anti-Kras siRNA significantly delay tumor growth. Thus, iRGD TPNs hold promise in treating PDAC by not only overcoming physical barriers to therapy, but by leveraging the stroma to achieve knockdown of the gold-standard genetic target. Moreover, the modular construction of this delivery platform allows for facile adaptation to future genetic target candidates in pancreatic cancer. Mol Cancer Ther; 17(11); 2377-88. ©2018 AACR.
©2018 American Association for Cancer Research.
-
FC/FACS
-
Mus musculus (House mouse)
-
Cancer Research
-
Genetics
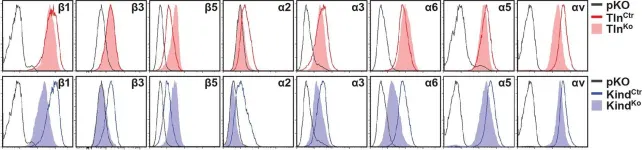
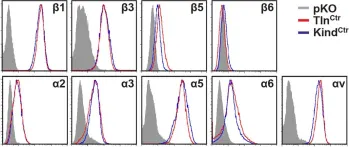
In The Journal of Cell Biology on 6 November 2017 by Böttcher, R. T., Veelders, M., et al.
Cell spreading requires the coupling of actin-driven membrane protrusion and integrin-mediated adhesion to the extracellular matrix. The integrin-activating adaptor protein kindlin-2 plays a central role for cell adhesion and membrane protrusion by directly binding and recruiting paxillin to nascent adhesions. Here, we report that kindlin-2 has a dual role during initial cell spreading: it binds paxillin via the pleckstrin homology and F0 domains to activate Rac1, and it directly associates with the Arp2/3 complex to induce Rac1-mediated membrane protrusions. Consistently, abrogation of kindlin-2 binding to Arp2/3 impairs lamellipodia formation and cell spreading. Our findings identify kindlin-2 as a key protein that couples cell adhesion by activating integrins and the induction of membrane protrusions by activating Rac1 and supplying Rac1 with the Arp2/3 complex.
© 2017 Böttcher et al.
-
FC/FACS
-
Mus musculus (House mouse)
-
Cell Biology
In Elife on 27 January 2016 by Theodosiou, M., Widmaier, M., et al.
Fig.2.A

-
FC/FACS
-
Mus musculus (House mouse)
Collected and cropped from Elife by CiteAb, provided under a CC-BY license
Image 1 of 2
In Elife on 27 January 2016 by Theodosiou, M., Widmaier, M., et al.
Fig.1.A

-
FC/FACS
-
Mus musculus (House mouse)
Collected and cropped from Elife by CiteAb, provided under a CC-BY license
Image 1 of 2