Most exhaustion studies have focused on CD8+ T cells. Here, we demonstrated reciprocal growth inhibition of CD4+ T cells and colorectal cancer cells, which induced the expression of PD-1, PD-L1, and PD-L2 in CD4+ T cells. The accelerated exhaustion of CD4+ T cells was evidenced by the reduced secretion of several cytokines, including IL-2, IFN-γ, or TNFα, and elevated secretion of CXCL family chemokines. Progressive expression of PD-L1, CTLA4, and IDO1 exhaustion markers occurred concomitantly with tumor growth in vivo in a mouse model. The pattern of CD4+ T cell exhaustion was analogous to that observed in CD8+ T cells, although with altered dynamics. The PD-L1-high phenotype can be induced by co-culture with tumor cells and is mediated by secreted factors in addition to cell contact. Our findings revealed that IFN-γ receptor knockout T cells exhibited PD-L1 protein expression when cultured with tumor cells, suggesting that PD-L1 expression is not fully dependent on IFN-γ. The TIL population undergoing exhaustion due to persistent antigen stimulation in the presence of cancer cells gradually acquires an immunosuppressive phenotype. The accumulation of inhibitory signals exerted by both cancer cells and T cells, which had converted to a suppressive phenotype, accelerated T cell exhaustion.
Product Citations: 59
Cancer cells accelerate exhaustion of persistently activated mouse CD4+ T cells.
In Oncoimmunology on 1 December 2025 by Stachowiak, M., Becker, W., et al.
-
Cancer Research
-
Immunology and Microbiology
In Frontiers in Immunology on 17 July 2025 by Guldenpfennig, C., Guan, Y., et al.
Upon antigen recognition, naive CD8 T cells must induce c-JUN N-terminal kinase (JNK), NF-κB, and Akt signaling to drive differentiation and generate a heterogeneous effector response. While the roles of these three pathways individually in mediating essential cellular responses for CD8 T cell differentiation are well established, the mechanisms of signal integration and crosstalk between these pathways to produce a diverse and heterogeneous response to infection remain poorly understood. Here, we establish the critical role of the Plenty of SH3 Domains (POSH) scaffold protein in coordinating signals from all three pathways to support CD8 T cell differentiation and fate.
Using novel conditional T cell POSH knockout reporter mouse models (as POSHfl/fl CD4-Cre eGFP, POSHfl/fl GzmB-Cre eGFP), we determined the phenotype of T cells in the thymus and periphery through flow cytometry. Polyclonal and OT1 TCR transgenic POSH cKO CD8 T cells were stimulated in vitro and analyzed by flow cytometry to assess cell fate. JNK, NF-κB, and Akt pathways were examined via flow cytometry and immunoblotting. Purified OT1 CD8 T cells from these mice were adoptively transferred and subsequently challenged with VSV-OVA infection; their phenotype, effector function, and signaling were then assessed ex vivo by flow cytometry.
We demonstrate that POSH is essential for proper induction of the JNK, NF-κB, and Akt pathways. Furthermore, the absence of these signals due to POSH deficiency results in reduced differentiation into short-lived effector cells (SLECs), delayed proliferation, and decreased survival of memory precursor cells (MPECs) during the contraction phase.
Collectively, these data identify POSH as a key regulator of CD8 T cell fate and enhance our understanding of the complex mechanisms governing signal integration during CD8 T cell responses to infection.
Copyright © 2025 Guldenpfennig, Guan, Cseri, Lopez, Teixeiro and Daniels.
-
Immunology and Microbiology
In Molecular Cancer on 3 July 2025 by Cui, Y., Qiu, T., et al.
-
Cancer Research
-
Immunology and Microbiology
In Journal of Nanobiotechnology on 3 July 2025 by Liang, C., Li, R., et al.
The emergence of SARS-CoV-2 variants has underscored the urgent need for innovative vaccine strategies that provide robust and enduring protection against diverse strains. Our study introduces the FP-HR5 nanoparticle vaccine, designed to target the highly conserved S2 subunit of the spike protein, including the fusion peptide (FP) and heptad repeats (HR1 and HR2), using a 24-mer Helicobacter pylori ferritin platform. Administered intranasally, the FP-HR5-NP vaccine elicits robust systemic and mucosal immune responses in vivo, generating high titers of FP- and HR5-specific IgG antibodies. Notably, intranasal immunization resulted in elevated levels of secretory IgA and IgG in bronchoalveolar lavage fluid (BALF) and stimulated T-cell immune responses, significantly increasing resident memory B cells (BRM) and resident memory T cells (TRM) in the lungs. In hACE2 transgenic mice, three doses of FP-HR5-NP conferred substantial protection against Delta and Omicron variant challenges, with undetectable viral RNA levels in the lungs and no pathological changes observed. Overall, the FP-HR5-NP vaccine triggers comprehensive humoral and cellular immune responses at the mucosa, providing broad defense against SARS-CoV-2 variants and positioning it as a promising candidate for a universal COVID-19 vaccine solution.
© 2025. The Author(s).
-
COVID-19
-
Immunology and Microbiology
In Nature Immunology on 30 June 2025 by Venzin, V., Beccaria, C. G., et al.
Chronic hepatitis B virus (HBV) infection is marked by dysfunctional HBV-specific CD8+ T cells, and restoring their effector activity is a major therapeutic goal. Here, we generated HBV-specific CD4+ T cell receptor transgenic mice to show that CD4+ effector T cells can prevent and reverse the CD8⁺ T cell dysfunction induced by hepatocellular priming. This rescue enhances antiviral CD8+ T cell function and suppresses viral replication. CD4+ T cell help occurs directly within the liver, independent of secondary lymphoid organs, and requires local antigen recognition. Kupffer cells, rather than dendritic cells, are the critical antigen-presenting platform. CD4+ T cells license Kupffer cells via CD40-CD40L interactions, triggering interleukin (IL)-12 and IL-27 production. IL-12 expands the CD4+ T cell pool, while IL-27 is essential for CD8+ T cell rescue. Exogenous IL-27 similarly restores HBV-specific CD8+ T cell function in mice and in T cells isolated from chronically infected patients. These findings identify IL-27 as a tractable immunotherapeutic target in chronic HBV infection.
© 2025. The Author(s).
-
Immunology and Microbiology
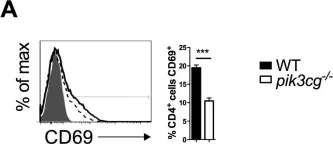
In Elife on 10 September 2018 by Lian, G., Gnanaprakasam, J. R., et al.
Fig.2.A

-
FC/FACS
-
Mus musculus (House mouse)
Collected and cropped from Elife by CiteAb, provided under a CC-BY license
Image 1 of 2
In PLoS One on 3 October 2012 by Comerford, I., Litchfield, W., et al.
Fig.2.A

-
FC/FACS
-
Mus musculus (House mouse)
Collected and cropped from PLoS One by CiteAb, provided under a CC-BY license
Image 1 of 2