Infection with the helminth Schistosoma mansoni can cause exacerbated morbidity and mortality via a pathogenic host CD4 T cell-mediated immune response directed against parasite egg antigens, with T helper (Th) 17 cells playing a major role in the development of severe granulomatous hepatic immunopathology. The role of inflammasomes in intensifying disease has been reported; however, neither the types of caspases and inflammasomes involved, nor their impact on the Th17 response are known. Here we show that enhanced egg-induced IL-1β secretion and pyroptotic cell death required both caspase-1 and caspase-8 as well as NLRP3 and AIM2 inflammasome activation. Schistosome genomic DNA activated AIM2, whereas reactive oxygen species, potassium efflux and cathepsin B, were the major activators of NLRP3. NLRP3 and AIM2 deficiency led to a significant reduction in pathogenic Th17 responses, suggesting their crucial and non-redundant role in promoting inflammation. Additionally, we show that NLRP3- and AIM2-induced IL-1β suppressed IL-4 and protective Type I IFN (IFN-I) production, which further enhanced inflammation. IFN-I signaling also curbed inflammasome- mediated IL-1β production suggesting that these two antagonistic pathways shape the severity of disease. Lastly, Gasdermin D (Gsdmd) deficiency resulted in a marked decrease in egg-induced granulomatous inflammation. Our findings establish NLRP3/AIM2-Gsdmd axis as a central inducer of pathogenic Th17 responses which is counteracted by IFN-I pathway in schistosomiasis.
Copyright: © 2025 Suresh Kumar Meena Kumari et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Product Citations: 158
In PLoS Pathogens on 1 March 2025 by Suresh Kumar Meena Kumari, M., Liu, P., et al.
-
FC/FACS
-
Immunology and Microbiology
In Cell Death and Differentiation on 1 December 2024 by Weiss, F. D., Alvárez, Y., et al.
Immune response genes are highly polymorphic in humans and mice, with heterogeneity amongst loci driving strain-specific host defence responses. The inadvertent retention of polymorphic loci can introduce confounding phenotypes, leading to erroneous conclusions, and impeding scientific advancement. In this study, we employ a combination of RNAseq and variant calling analyses to identify a substantial region of 129S genome, including the highly polymorphic Nlrp1 locus, proximal to Nlrp3, in one of the most commonly used mouse models of NLRP3 deficiency (Nlrp3tm1Flv). We show that the presence of the Nlrp1129S locus leads to an increase in NLRP1B protein expression, and a sensitising of Nlrp3tm1Flv macrophages to NLRP1 inflammasome activation, independent of NLRP3 deficiency. Retention of 129S genome further leads to protein sequence differences and altered gene regulation across multiple cell types, including of the key tissue-resident macrophage marker, TIM4. Using alternative models of NLRP3 deficiency, including a previously undescribed conditional Nlrp3 allele enabling precise temporal and cell-type specific control over Nlrp3 deletion, we further show that NLRP3 contributes to Talabostat-driven IL-1β release. Our study also establishes a generic framework to identify functionally relevant SNPs and assess genomic contamination in transgenic mice using RNAseq data. This allows for unambiguous attribution of phenotypes to the target gene and advances the precision and reliability of research in the field of host defence responses.
© 2024. The Author(s).
-
FC/FACS
-
Mus musculus (House mouse)
-
Biochemistry and Molecular biology
-
Cell Biology
-
Stem Cells and Developmental Biology
Protein arginine methyltransferase 2 controls inflammatory signaling in acute myeloid leukemia.
In Communications Biology on 20 June 2024 by Sauter, C., Morin, T., et al.
Arginine methylation is catalyzed by protein arginine methyltransferases (PRMTs) and is involved in various cellular processes, including cancer development. PRMT2 expression is increased in several cancer types although its role in acute myeloid leukemia (AML) remains unknown. Here, we investigate the role of PRMT2 in a cohort of patients with AML, PRMT2 knockout AML cell lines as well as a Prmt2 knockout mouse model. In patients, low PRMT2 expressors are enriched for inflammatory signatures, including the NF-κB pathway, and show inferior survival. In keeping with a role for PRMT2 in control of inflammatory signaling, bone marrow-derived macrophages from Prmt2 KO mice display increased pro-inflammatory cytokine signaling upon LPS treatment. In PRMT2-depleted AML cell lines, aberrant inflammatory signaling has been linked to overproduction of IL6, resulting from a deregulation of the NF-κB signaling pathway, therefore leading to hyperactivation of STAT3. Together, these findings identify PRMT2 as a key regulator of inflammation in AML.
© 2024. The Author(s).
-
FC/FACS
-
Mus musculus (House mouse)
-
Cancer Research
-
Immunology and Microbiology
In Cell Death Discovery on 19 June 2024 by Krug, A., Mhaidly, R., et al.
Cancer metabolic reprogramming has been recognized as one of the cancer hallmarks that promote cell proliferation, survival, as well as therapeutic resistance. Up-to-date regulation of metabolism in T-cell lymphoma is poorly understood. In particular, for human angioimmunoblastic T-cell lymphoma (AITL) the metabolic profile is not known. Metabolic intervention could help identify new treatment options for this cancer with very poor outcomes and no effective medication. Transcriptomic analysis of AITL tumor cells, identified that these cells use preferentially mitochondrial metabolism. By using our preclinical AITL mouse model, mimicking closely human AITL features, we confirmed that T follicular helper (Tfh) tumor cells exhibit a strong enrichment of mitochondrial metabolic signatures. Consistent with these results, disruption of mitochondrial metabolism using metformin or a mitochondrial complex I inhibitor such as IACS improved the survival of AITL lymphoma-bearing mice. Additionally, we confirmed a selective elimination of the malignant human AITL T cells in patient biopsies upon mitochondrial respiration inhibition. Moreover, we confirmed that diabetic patients suffering from T-cell lymphoma, treated with metformin survived longer as compared to patients receiving alternative treatments. Taking together, our findings suggest that targeting the mitochondrial metabolic pathway could be a clinically efficient approach to inhibit aggressive cancers such as peripheral T-cell lymphoma.
© 2024. The Author(s).
-
Mus musculus (House mouse)
-
Cancer Research
-
Cell Biology
-
Immunology and Microbiology
Preprint on BioRxiv : the Preprint Server for Biology on 13 March 2024 by Suresh Kumar Meena Kumari, M., Liu, P., et al.
Infection with the helminth Schistosoma mansoni can cause exacerbated morbidity and mortality via a pathogenic host CD4 T cell-mediated immune response directed against parasite egg antigens, with T helper (Th) 17 cells playing a major role in the development of severe granulomatous hepatic immunopathology. The role of inflammasomes in intensifying disease has been reported; however, neither the types of caspases and inflammasomes involved, nor their impact on the Th17 response are known. Here we show that enhanced egg-induced IL-1β secretion and pyroptotic cell death required both caspase-1 and caspase-8 as well as NLRP3 and AIM2 inflammasome activation. Schistosome genomic DNA activated AIM2, whereas reactive oxygen species, potassium efflux and cathepsin B, were the major activators of NLRP3. NLRP3 and AIM2 deficiency led to a significant reduction in pathogenic Th17 responses, suggesting their crucial and non-redundant role in promoting inflammation. Additionally, we show that NLRP3- and AIM2-induced IL-1β suppressed IL-4 and protective Type I IFN (IFN-I) production, which further enhanced inflammation. IFN-I signaling also curbed inflammasome-mediated IL-1β production suggesting that these two antagonistic pathways shape the severity of disease. Lastly, Gasdermin D (Gsdmd) deficiency resulted in a marked decrease in egg-induced granulomatous inflammation. Our findings establish NLRP3/AIM2-Gsdmd axis as a central inducer of pathogenic Th17 responses which is counteracted by IFN-I pathway in schistosomiasis. Summary Schistosomiasis is a major tropical parasitic disease caused by trematode worms of the genus Schistosoma. Morbidity and mortality in infection with the species Schistosoma mansoni are due to a pathogenic CD4 T cell-mediated immune response directed against parasite eggs, resulting in granulomatous inflammation. In severe cases of schistosomiasis, there is liver fibrosis, hepatosplenomegaly, portal hypertension, gastro-intestinal hemorrhage and death. Here we describe the role of two proteins, the NLRP3 and AIM2 inflammasomes, in intensifying disease. We found that upstream proteins which activate these inflammasomes are caspase-1 and caspase 8; these in turn lead to the activation of another protein, Gasdermin D (Gsdmd), which facilitates the release of the proinflammatory cytokine IL-1β. Importantly, we observed that mice deficient in Gsdmd exhibit diminished pathology. Finally, we discovered that the protective Type I Interferon (IFN-I) pathway counteracts the caspase/inflammasome/Gsdmd axis thereby controlling egg mediated inflammation. These results give us a deeper understanding of the functional features of the crosstalk between inflammasome and IFN-I pathway, which may lead to the identification of novel targets for therapeutic intervention.
-
Mus musculus (House mouse)
In Elife on 16 January 2019 by Gerber, D., Ghidinelli, M., et al.
Fig.3.C

-
ICC
-
Mus musculus (House mouse)
Collected and cropped from Elife by CiteAb, provided under a CC-BY license
Image 1 of 2
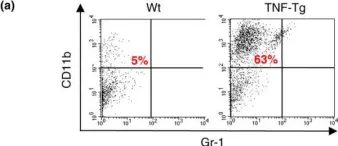
In Arthritis Res Ther on 14 November 2007 by Zhang, Q., Lu, Y., et al.
Fig.4.A

-
FC/FACS
-
Collected and cropped from Arthritis Res Ther by CiteAb, provided under a CC-BY license
Image 1 of 2