Ribonucleases (RNases) are ubiquitous enzymes that process or degrade RNA, essential for cellular functions and immune responses. The EndoU-like superfamily includes endoribonucleases conserved across bacteria, eukaryotes, and certain viruses, with an ancient evolutionary link to the ribonuclease A-like superfamily. Both bacterial EndoU and animal RNase A share a similar fold and function independently of cofactors. In contrast, the eukaryotic EndoU catalytic domain requires divalent metal ions for catalysis, possibly due to an N-terminal extension near the catalytic core. In this study, we use biophysical and computational techniques along with in vitro assays to investigate the calcium-dependent activation of human EndoU. We determine the crystal structure of EndoU bound to calcium and find that calcium binding remote from the catalytic triad triggers water-mediated intramolecular signaling and structural changes, activating the enzyme through allostery. Calcium binding involves residues from both the catalytic core and the N-terminal extension, indicating that the N-terminal extension interacts with the catalytic core to modulate activity in response to calcium. Our findings suggest that similar mechanisms may be present across all eukaryotic EndoUs, highlighting a unique evolutionary adaptation that connects endoribonuclease activity to cellular signaling in eukaryotes.
© 2025. The Author(s).
Product Citations: 436
Molecular basis for the calcium-dependent activation of the ribonuclease EndoU.
In Nature Communications on 1 April 2025 by Malard, F., Dias, K., et al.
-
RT-qPCR
-
Mus musculus (House mouse)
Acidic pH can attenuate immune killing through inactivation of perforin.
In EMBO Reports on 1 February 2025 by Hodel, A. W., Rudd-Schmidt, J. A., et al.
Cytotoxic lymphocytes are crucial to our immune system, primarily eliminating virus-infected or cancerous cells via perforin/granzyme killing. Perforin forms transmembrane pores in the plasma membrane, allowing granzymes to enter the target cell cytosol and trigger apoptosis. The prowess of cytotoxic lymphocytes to efficiently eradicate target cells has been widely harnessed in immunotherapies against haematological cancers. Despite efforts to achieve a similar outcome against solid tumours, the immunosuppressive and acidic tumour microenvironment poses a persistent obstacle. Using different types of effector cells, including therapeutically relevant anti-CD19 CAR T cells, we demonstrate that the acidic pH typically found in solid tumours hinders the efficacy of immune therapies by impeding perforin pore formation within the immunological synapse. A nanometre-scale study of purified recombinant perforin undergoing oligomerization reveals that pore formation is inhibited specifically by preventing the formation of a transmembrane β-barrel. The absence of perforin pore formation directly prevents target cell death. This finding uncovers a novel layer of immune effector inhibition that must be considered in the development of effective immunotherapies for solid tumours.
© 2025. The Author(s).
-
Immunology and Microbiology
Itaconate suppresses house dust mite-induced allergic airways disease and Th2 cell differentiation.
In Mucosal Immunology on 1 December 2024 by Li, Y., Singh, S., et al.
Itaconate was initially identified as an antimicrobial compound produced by myeloid cells. Beyond its antimicrobial role, itaconate may also serve as a crucial metabolic and immune modulator. We therefore examined the roles of aconitate decarboxylase 1 (Acod1) and itaconate in house dust mite (HDM)-sensitized and -challenged mice, a model of T helper 2 (Th2)-driven allergic airways disease. HDM treatment induced lung Acod1 mRNA expression and bronchoalveolar lavage (BAL) itaconate levels in wild-type C57BL/6 mice. Acod1 knockout mice (Acod1-KO) with negligible BAL itaconate showed heightened HDM-induced type 2 cytokine expression, increased serum IgE, and enhanced recruitment of Th2 cells in the lung, indicating a shift towards a more pronounced Th2 immune response. Acod1-KO mice also showed increased eosinophilic airway inflammation and hyperresponsiveness. Experiments in chimeric mice demonstrated that bone marrow from Acod1-KO mice is sufficient to increase type 2 cytokine expression in wild-type mice, and that restitution of bone marrow from wild type mice attenuates mRNA expression of Th2 cytokines in Acod1-KO mice. Specific deletion of Acod1 in lysozyme-secreting macrophages (LysM-cre+Acod1flox/flox) recapitulated the exaggerated phenotype observed in whole-body Acod1-KO mice. Adoptive transfer of Acod1-KO bone marrow-derived macrophages also increased lung mRNA expression of Th2 cytokines. In addition, treatment of Th2-polarized CD4 cells with itaconate impeded Th2 cell differentiation, as shown by reduced expression of Gata3 and decreased release of IL-5 and IL-13. Finally, public datasets of human samples show lower Acod1 expression in subjects with allergic asthma, consistent with a protective role of itaconate in asthma pathogenesis. Together, these data suggest that itaconate plays a protective, immunomodulatory role in limiting airway type 2 inflammation after allergen challenge by attenuating T cell responses.
Copyright © 2024 The Authors. Published by Elsevier Inc. All rights reserved.
-
Immunology and Microbiology
In The Journal of Clinical Investigation on 15 October 2024 by Chowdhury, N. U., Cephus, J. Y., et al.
Female individuals have an increased prevalence of many Th17 cell-mediated diseases, including asthma. Androgen signaling decreases Th17 cell-mediated airway inflammation, and Th17 cells rely on glutaminolysis. However, it remains unclear whether androgen receptor (AR) signaling modifies glutamine metabolism to suppress Th17 cell-mediated airway inflammation. We show that Th17 cells from male humans and mice had decreased glutaminolysis compared with female individuals, and that AR signaling attenuated Th17 cell mitochondrial respiration and glutaminolysis in mice. Using allergen-induced airway inflammation mouse models, we determined that females had a selective reliance upon glutaminolysis for Th17-mediated airway inflammation, and that AR signaling attenuated glutamine uptake in CD4+ T cells by reducing expression of glutamine transporters. In patients with asthma, circulating Th17 cells from men had minimal reliance upon glutamine uptake compared to Th17 cells from women. AR signaling thus attenuates glutaminolysis, demonstrating sex-specific metabolic regulation of Th17 cells with implications for Th17 or glutaminolysis targeted therapeutics.
-
Biochemistry and Molecular biology
-
Cell Biology
-
Endocrinology and Physiology
-
Immunology and Microbiology
In IScience on 16 August 2024 by Wenzek, C., Siemes, D., et al.
The immune system has emerged as an important target of thyroid hormones (THs); however, the role of TH in T cells has so far remained elusive. In this study, we assessed the effect of TH receptor α (TRα) signaling on activation and function of T cells. Our findings show that lack of canonical TRα action not only increased the frequency of regulatory T cells (Treg) but propelled an activated and migratory Treg phenotype and nuclear factor κB (NF-κB) activation in Treg. Conversely, canonical TRα action reduced activation of the NF-κB pathway previously shown to play a pivotal role in Treg differentiation and function. Taken together, our findings demonstrate that TRα impacts T cell differentiation and phenotype. Given the well-known interaction of inflammation, immune responses, and TH axis in e.g., severe illness, altered TH-TRα signaling may have an important role in regulating T cell responses during disease.
© 2024 The Author(s).
-
Mus musculus (House mouse)
-
Endocrinology and Physiology
-
Immunology and Microbiology
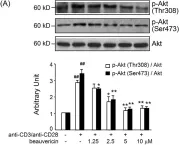
In PLoS One on 18 December 2013 by Wu, X. F., Xu, R., et al.
Fig.7.A

-
WB
-
Collected and cropped from PLoS One by CiteAb, provided under a CC-BY license
Image 1 of 1