Naturally circulating strains of eastern equine encephalitis virus (EEEV) bind heparan sulfate (HS) receptors and this interaction has been linked to neurovirulence. Previous studies associated EEEV-HS interactions with three positively charged amino acid clusters on the E2 glycoprotein. One of these sites has recently been reported to be critical for binding EEEV to the very-low-density lipoprotein receptor (VLDLR), an EEEV receptor protein. The proteins apolipoprotein E receptor 2 (ApoER2) isoforms 1 and 2, and LDLR have also been shown to function as EEEV receptors. Herein, we investigate the individual contribution of each HS interaction site to EEEV HS- and protein receptor-dependent infection in vitro and EEEV replication in animals. We show that each site contributes to both EEEV-HS and EEEV-protein receptor interactions, providing evidence that altering these interactions can affect disease in mice and eliminate mosquito infectivity. Thus, multiple HS-binding sites exist in EEEV E2, and these sites overlap functionally with protein receptor interaction sites, with each type of interaction contributing to tissue infectivity and disease phenotypes.
© 2025. The Author(s).
Product Citations: 13
In Nature Communications on 5 August 2025 by Alcorn, M. D. H., Sun, C., et al.
-
Immunology and Microbiology
-
Veterinary Research
CD30 influences germinal center B-cell dynamics and the expansion of IgG1-switched B cells.
In Cellular Molecular Immunology on 1 December 2024 by Wang, Y., Rambold, U., et al.
Initially, identified as a Hodgkin lymphoma marker, CD30 was subsequently detected on a subset of human B cells within and around germinal centers (GCs). While CD30 expression is typically restricted to a few B cells, expansion of CD30-expressing B cells occurs in certain immune disorders and during viral infections. The role of CD30 in B cells remains largely unclear. To address this gap in knowledge, we established a conditional CD30-knockin mouse strain. In these mice, B-cell-specific CD30 expression led to a normal B-cell phenotype in young mice, but most aged mice exhibited significant expansion of B cells, T cells and myeloid cells and increased percentages of GC B cells and IgG1-switched cells. This may be driven by the expansion of CD4+ senescence-associated T cells and T follicular helper cells, which partially express CD30-L (CD153) and may stimulate CD30-expressing B cells. Inducing CD30 expression in antigen-activated B cells accelerates the GC reaction and augments plasma cell differentiation, possibly through the posttranscriptional upregulation of CXCR4. Furthermore, CD30 expression in GC B cells promoted the expansion of IgG1-switched cells, which displayed either a GC or memory-like B-cell phenotype, with abnormally high IgG1 levels compared with those in controls. These findings shed light on the role of CD30 signaling in GC B cells and suggest that elevated CD30+ B-cell numbers lead to pathological lymphocyte activation and proliferation.
© 2024. The Author(s).
-
Mus musculus (House mouse)
-
Immunology and Microbiology
PRO-2000 exhibits SARS-CoV-2 antiviral activity by interfering with spike-heparin binding.
In Antiviral Research on 1 September 2023 by Vanderlinden, E., Boonen, A., et al.
Here, we report on the anti-SARS-CoV-2 activity of PRO-2000, a sulfonated polyanionic compound. In Vero cells infected with the Wuhan, alpha, beta, delta or omicron variant, PRO-2000 displayed EC50 values of 1.1 μM, 2.4 μM, 1.3 μM, 2.1 μM and 0.11 μM, respectively, and an average selectivity index (i.e. ratio of cytotoxic versus antiviral concentration) of 172. Its anti-SARS-CoV-2 activity was confirmed by virus yield assays in Vero cells, Caco2 cells and A549 cells overexpressing ACE2 and TMPRSS2 (A549-AT). Using pseudoviruses bearing the SARS-CoV-2 spike (S), PRO-2000 was shown to block the S-mediated pseudovirus entry in Vero cells and A549-AT cells, with EC50 values of 0.091 μM and 1.6 μM, respectively. This entry process is initiated by interaction of the S glycoprotein with angiotensin-converting enzyme 2 (ACE2) and heparan sulfate proteoglycans. Surface Plasmon Resonance (SPR) studies showed that PRO-2000 binds to the receptor-binding domain (RBD) of S with a KD of 1.6 nM. Similar KD values (range: 1.2 nM-2.1 nM) were obtained with the RBDs of the alpha, beta, delta and omicron variants. In an SPR neutralization assay, PRO-2000 had no effect on the interaction between the RBD and ACE2. Instead, PRO-2000 was proven to inhibit binding of the RBD to a heparin-coated sensor chip, yielding an IC50 of 1.1 nM. To conclude, PRO-2000 has the potential to inhibit a broad range of SARS-CoV-2 variants by blocking the heparin-binding site on the S protein.
Copyright © 2023 The Authors. Published by Elsevier B.V. All rights reserved.
-
COVID-19
Differentiation of Brain Pericyte-Like Cells from Human Pluripotent Stem Cell-Derived Neural Crest.
In Current Protocols on 1 January 2021 by Gastfriend, B. D., Stebbins, M. J., et al.
Brain pericytes regulate diverse aspects of neurovascular development and function, including blood-brain barrier (BBB) induction and maintenance. Primary brain pericytes have been widely employed in coculture-based in vitro models of the BBB, and a method to generate brain pericytes from human pluripotent stem cells (hPSCs) could provide a renewable, genetically tractable source of cells for BBB modeling and studying pericyte roles in development and disease. Here, we describe a protocol to differentiate hPSCs to NG2+ PDGFRβ+ αSMAlow brain pericyte-like cells in 22-25 days through a p75-NGFR+ HNK-1+ neural crest intermediate, which mimics the developmental origin of forebrain pericytes. The resulting brain pericyte-like cells have molecular and functional attributes of brain pericytes. We also provide protocols for maintenance, cryopreservation, and recovery of the neural crest intermediate, and for molecular and functional characterization of the resulting cells. © 2021 Wiley Periodicals LLC. Basic Protocol 1: Differentiation of hPSCs to neural crest Basic Protocol 2: Differentiation of neural crest to brain pericyte-like cells Support Protocol 1: Flow cytometry analysis of neural crest cells Support Protocol 2: Maintenance, cryopreservation, and recovery of neural crest cells Support Protocol 3: Molecular characterization of brain pericyte-like cells Support Protocol 4: Cord formation assay with endothelial cells and brain pericyte-like cells.
© 2021 Wiley Periodicals LLC.
-
Homo sapiens (Human)
-
Stem Cells and Developmental Biology
Use of Specialized Pro-Resolving Mediators to Alleviate Cold Platelet Storage Lesion.
In Transfusion on 1 June 2020 by Reddoch-Cardenas, K. M., Sharma, U., et al.
Cold-stored platelets are an attractive option for treatment of actively bleeding patients due to a reduced risk of septic complications and preserved hemostatic function compared to conventional room temperature-stored platelets. However, refrigeration causes increased platelet activation and aggregate formation. Specialized pro-resolving mediators (SPMs), cell signaling mediators biosynthesized from essential fatty acids, have been shown to modulate platelet function and activation. In this study, we sought to determine if SPMs could be used to inhibit cold-stored platelet activation.
Platelets were collected from healthy donors (n = 4-7) and treated with SPMs (resolvin E1 [RvE1], maresin 1 [MaR1], and resolvin D2 [RvD2]) or vehicle (VEH; 0.1% EtOH). Platelets were stored without agitation in the cold and assayed on Days 0 and 7 of storage for platelet activation levels using flow cytometry, platelet count, aggregation response using impedance aggregometry, and nucleotide content using mass spectrometry.
Compared to VEH, SPM treatment inhibited GPIb shedding (all compounds), significantly reduced both PS exposure and activation of GPIIb/IIIa receptor (RvD2, MaR1), and preserved aggregation response to TRAP (RvD2, MaR1) after 7 days of storage. Similar to untreated cold-stored platelets, SPM-treated samples did not preserve platelet counts or block the release of P-Selectin. Nucleotide content was unaffected by SPM treatment in cold-stored platelets.
SPM treatment, particularly Mar1 and RvD2, led to reduced platelet activation and preserved platelet function after 7 days of storage in the cold. Future work is warranted to better elucidate the mechanism of action of SPMs on cold platelet function and activation.
Published 2020. This article is a U.S. Government work and is in the public domain in the USA.
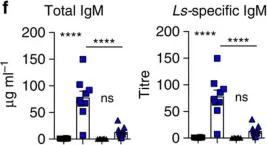
In Nat Commun on 1 September 2016 by Jackson-Jones, L. H., Duncan, S. M., et al.
Fig.5.E

-
ELISA
-
Mus musculus (House mouse)
Collected and cropped from Nat Commun by CiteAb, provided under a CC-BY license
Image 1 of 7
In Nat Commun on 1 September 2016 by Jackson-Jones, L. H., Duncan, S. M., et al.
Fig.5.F

-
ELISA
-
Mus musculus (House mouse)
Collected and cropped from Nat Commun by CiteAb, provided under a CC-BY license
Image 1 of 7
In Nat Commun on 1 September 2016 by Jackson-Jones, L. H., Duncan, S. M., et al.
Fig.1.A,B

-
ELISA
-
Mus musculus (House mouse)
Collected and cropped from Nat Commun by CiteAb, provided under a CC-BY license
Image 1 of 7
In Nat Commun on 1 September 2016 by Jackson-Jones, L. H., Duncan, S. M., et al.
Fig.1.G

-
ELISA
-
Mus musculus (House mouse)
Collected and cropped from Nat Commun by CiteAb, provided under a CC-BY license
Image 1 of 7
In Nat Commun on 1 September 2016 by Jackson-Jones, L. H., Duncan, S. M., et al.
Fig.3.F

-
ELISA
-
Mus musculus (House mouse)
Collected and cropped from Nat Commun by CiteAb, provided under a CC-BY license
Image 1 of 7
In Nat Commun on 1 September 2016 by Jackson-Jones, L. H., Duncan, S. M., et al.
Fig.6.G

-
ELISA
-
Mus musculus (House mouse)
Collected and cropped from Nat Commun by CiteAb, provided under a CC-BY license
Image 1 of 7
In Nat Commun on 1 September 2016 by Jackson-Jones, L. H., Duncan, S. M., et al.
Fig.6.K

-
ELISA
-
Mus musculus (House mouse)
Collected and cropped from Nat Commun by CiteAb, provided under a CC-BY license
Image 1 of 7