Tubular aggregate myopathy (TAM) and Stormorken syndrome (STRMK) are clinically overlapping disorders characterized by childhood-onset muscle weakness and a variable occurrence of multisystemic signs, including short stature, thrombocytopenia, and hyposplenism. TAM/STRMK is caused by gain-of-function mutations in the Ca2+ sensor STIM1 or the Ca2+ channel ORAI1, both of which regulate Ca2+ homeostasis through the ubiquitous store-operated Ca2+ entry (SOCE) mechanism. Functional experiments in cells have demonstrated that the TAM/STRMK mutations induce SOCE overactivation, resulting in excessive influx of extracellular Ca2+. There is currently no treatment for TAM/STRMK, but SOCE is amenable to manipulation. Here, we crossed Stim1R304W/+ mice harboring the most common TAM/STRMK mutation with Orai1R93W/+ mice carrying an ORAI1 mutation partially obstructing Ca2+ influx. Compared with Stim1R304W/+ littermates, Stim1R304W/+Orai1R93W/+ offspring showed a normalization of bone architecture, spleen histology, and muscle morphology; an increase of thrombocytes; and improved muscle contraction and relaxation kinetics. Accordingly, comparative RNA-Seq detected more than 1,200 dysregulated genes in Stim1R304W/+ muscle and revealed a major restoration of gene expression in Stim1R304W/+Orai1R93W/+ mice. Altogether, we provide physiological, morphological, functional, and molecular data highlighting the therapeutic potential of ORAI1 inhibition to rescue the multisystemic TAM/STRMK signs, and we identified myostatin as a promising biomarker for TAM/STRMK in humans and mice.
Product Citations: 276
In JCI Insight on 5 March 2024 by Silva-Rojas, R., Pérez-Guàrdia, L., et al.
-
Mus musculus (House mouse)
In Communications Biology on 5 February 2024 by Petrova, E., López-Gay, J. M., et al.
Netherton syndrome (NS) is a rare skin disease caused by loss-of-function mutations in the serine peptidase inhibitor Kazal type 5 (SPINK5) gene. Disease severity and the lack of efficacious treatments call for a better understanding of NS mechanisms. Here we describe a novel and viable, Spink5 conditional knock-out (cKO) mouse model, allowing to study NS progression. By combining transcriptomics and proteomics, we determine a disease molecular profile common to mouse models and NS patients. Spink5 cKO mice and NS patients share skin barrier and inflammation signatures defined by up-regulation and increased activity of proteases, IL-17, IL-36, and IL-20 family cytokine signaling. Systemic inflammation in Spink5 cKO mice correlates with disease severity and is associated with thymic atrophy and enlargement of lymph nodes and spleen. This systemic inflammation phenotype is marked by neutrophils and IL-17/IL-22 signaling, does not involve primary T cell immunodeficiency and is independent of bacterial infection. By comparing skin transcriptomes and proteomes, we uncover several putative substrates of tissue kallikrein-related proteases (KLKs), demonstrating that KLKs can proteolytically regulate IL-36 pro-inflammatory cytokines. Our study thus provides a conserved molecular framework for NS and reveals a KLK/IL-36 signaling axis, adding new insights into the disease mechanisms and therapeutic targets.
© 2024. The Author(s).
-
Mus musculus (House mouse)
In Cancer Immunology, Immunotherapy : CII on 1 January 2023 by Nakagawa, N., Hashii, Y., et al.
Wilms' tumor 1 (WT1) is a promising tumor-associated antigen for cancer immunotherapy. We developed an oral protein vaccine platform composed of WT1-anchored, genetically engineered Bifidobacterium longum (B. longum) and conducted an in vivo study in mice to examine its anticancer activity. Mice were orally treated with phosphate-buffered saline, wild-type B. longum105-A, B. longum 2012 displaying only galacto-N-biose/lacto-N-biose I-binding protein (GLBP), and WT1 protein- and GLBP-expressing B. longum 420. Tumor size reduced significantly in the B. longum 420 group than in the B. longum 105-A and 2012 groups (P < 0.00 l each), indicating B. longum 420's antitumor activity via WT1-specific immune responses. CD8+ T cells played a major role in the antitumor activity of B. longum 420. The proportion of CD103+CD11b+CD11c+ dendritic cells (DCs) increased in the Peyer's patches (PPs) from mice in the B. longum 420 group, indicating the definite activation of DCs. In the PPs, the number and proportion of CD8+ T cells capable of producing interferon-gamma were significantly greater in the B. longum 420 group than in the B. longum 2012 group (P < 0.05 or < 0.01). The production of WT1-specific IgG antibody was significantly higher in the B. longum 420 group than in the 2012 group (P < 0.05). The B. longum 420 group showed the most intense intratumoral infiltration of CD4+ and CD8+ T cells primed by activated DCs in the PPs of mice in the B. longum 420 group. Our findings provide insights into a novel, intestinal bacterium-based, cancer immunotherapy through intestinal immunity.
© 2022. The Author(s).
-
Cancer Research
-
Immunology and Microbiology
In Aging (Albany NY) on 25 February 2022 by Yin, H., Miao, Z., et al.
Liver metastasis is the major cause of death in colorectal cancer (CRC) patients. Nevertheless, the underlying mechanisms remain unknown. Gut microbiota intricately affect the initiation and progression of CRC by instigating immune response through the secretion of pro-inflammatory cytokines. In this study, we investigated the contribution of Fusobacterium nucleatum (F.nucleatum) to the microbiota-liver axis of CRC in mice, focusing on the correlation between liver immunity and gut microbiota alterations. When F. nucleatum was orally administered to mice, CRC liver metastasis was evidently exaggerated and accompanied by noticeable deleterious effects on body weight, cecum weight, and overall survival time. Further evaluation of the immune response and cytokine profiles revealed a substantial increase in the levels of pro-inflammatory cytokines such as IL6, IL12, IL9, IL17A, CXCL1, MCP-1, TNF-α, and IFN-γ in the plasma of mice treated with F. nucleatum as compared to that in the untreated control mice. Besides, hepatic immune response was also modulated by recruitment of myeloid-derived suppressor cells, reduction in the infiltration of natural killer (NK) and T helper-17 (Th17) cells, as well as increase in regulatory T cell accumulation in the liver. Additionally, sustained F. nucleatum exposure abridged the murine gut microbiota diversity, inducing an imbalanced and restructured intestinal microflora. In particular, the abundance of CRC-promoting bacteria such as Enterococcus and Escherichia/Shigella was evidently elevated post F. nucleatum treatment. Thus, our findings suggest that F. nucleatum might be an important factor involved in promoting CRC liver metastasis by triggering of liver immunity through the regulation of gut microbiota structure and composition.
-
Cancer Research
-
Immunology and Microbiology
Resistance Against Leishmania major Infection Depends on Microbiota-Guided Macrophage Activation.
In Frontiers in Immunology on 9 November 2021 by Lopes, M. E., dos Santos, L. M., et al.
Innate immune cells present a dual role during leishmaniasis: they constitute the first line of host defense but are also the main host cells for the parasite. Response against the infection that results in the control of parasite growth and lesion healing depends on activation of macrophages into a classical activated phenotype. We report an essential role for the microbiota in driving macrophage and monocyte-derived macrophage activation towards a resistance phenotype against Leishmania major infection in mice. Both germ-free and dysbiotic mice showed a higher number of myeloid innate cells in lesions and increased number of infected cells, mainly dermal resident and inflammatory macrophages. Despite developing a Th1 immune response characterized by the same levels of IFN-γ production as the conventional mice, germ-free mice presented reduced numbers of iNOS+ macrophages at the peak of infection. Absence or disturbance of host microbiota impaired the capacity of bone marrow-derived macrophage to be activated for Leishmania killing in vitro, even when stimulated by Th1 cytokines. These cells presented reduced expression of inos mRNA, and diminished production of microbicidal molecules, such as ROS, while presenting a permissive activation status, characterized by increased expression of arginase I and il-10 mRNA and higher arginase activity. Colonization of germ-free mice with complete microbiota from conventional mice rescued their ability to control the infection. This study demonstrates the essential role of host microbiota on innate immune response against L. major infection, driving host macrophages to a resistance phenotype.
Copyright © 2021 Lopes, dos Santos, Sacks, Vieira and Carneiro.
-
Immunology and Microbiology
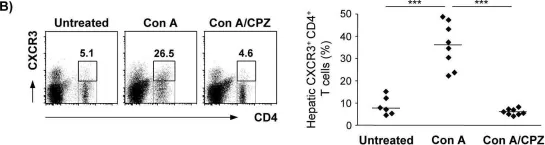
In PLoS One on 9 June 2015 by Neumann, K., Erben, U., et al.
Fig.7.B

-
FC/FACS
-
Collected and cropped from PLoS One by CiteAb, provided under a CC-BY license
Image 1 of 1