Camptothesome is an innovative nanovesicle therapeutic comprising the sphingomyelin-derived camptothecin (CPT) lipid bilayer. In this work, we deciphered that Camptothesome was taken up by colorectal cancer (CRC) cells through primarily the clathrin-mediated endocytotic pathway and displayed the potential of eliciting robust immunogenic cancer cell death (ICD) via upregulating calreticulin, high mobility group box 1 protein (HMGB-1), and adenosine triphosphate (ATP), three hallmarks involved in the induction of ICD. In addition, use of dying MC38 tumor cells treated with Camptothesome as vaccine prevented tumor growth in 60% mice that received subsequent injection of live MC38 cells on the contralateral flank, validating Camptothesome was a legitimate ICD inducer in vivo. Camptothesome markedly reduced the acute bone marrow toxicity and gastrointestinal mucositis associated with free CPT and beat free CPT and Onivyde on anti-CRC efficacy and immune responses in a partially interferon gamma (IFN-γ)-dependent manner. Furthermore, Camptothesome enhanced the efficacy of immune checkpoint inhibitors to shrink late-stage orthotopic MC38 CRC tumors with diminished tumor metastasis and markedly prolonged mice survival.
Copyright © 2022 Elsevier B.V. All rights reserved.
Product Citations: 114
Camptothesome elicits immunogenic cell death to boost colorectal cancer immune checkpoint blockade.
In Journal of Controlled Release : Official Journal of the Controlled Release Society on 1 September 2022 by Wang, Z., Li, W., et al.
-
FC/FACS
-
Mus musculus (House mouse)
-
Cancer Research
-
Immunology and Microbiology
In mBio on 2 February 2021 by Knuschke, T., Kollenda, S., et al.
PD-1-targeted therapies have shown modest antiviral effects in preclinical models of chronic viral infection. Thus, novel therapy protocols are necessary to enhance T cell immunity and viral control to overcome T cell dysfunction and immunosuppression. Here, we demonstrate that nanoparticle-based therapeutic vaccination improved PD-1-targeted therapy during chronic infection with Friend retrovirus (FV). Prevention of inhibitory signals by blocking PD-L1 in combination with therapeutic vaccination with nanoparticles containing the microbial compound CpG and a CD8+ T cell Gag epitope peptide synergistically enhanced functional virus-specific CD8+ T cell responses and improved viral clearance. We characterized the CD8+ T cell populations that were affected by this combination therapy, demonstrating that new effector cells were generated and that exhausted CD8+ T cells were reactivated at the same time. While CD8+ T cells with high PD-1 (PD-1hi) expression turned into a large population of granzyme B-expressing CD8+ T cells after combination therapy, CXCR5-expressing follicular cytotoxic CD8+ T cells also expanded to a high degree. Thus, our study describes a very efficient approach to enhance virus control and may help us to understand the mechanisms of combination immunotherapy reactivating CD8+ T cell immunity. A better understanding of CD8+ T cell immunity during combination therapy will be important for developing efficient checkpoint therapies against chronic viral infections and cancer.IMPORTANCE Despite significant efforts, vaccines are not yet available for every infectious pathogen, and the search for a protective approach to prevent the establishment of chronic infections, i.e., with HIV, continues. Immune checkpoint therapies targeting inhibitory receptors, such as PD-1, have shown impressive results against solid tumors. However, immune checkpoint therapies have not yet been licensed to treat chronic viral infections, since a blockade of inhibitory receptors alone provides only limited benefit, as demonstrated in preclinical models of chronic viral infection. Thus, there is a high interest in the development of potent combination immunotherapies. Here, we tested whether the combination of a PD-L1 blockade and therapeutic vaccination with functionalized nanoparticles is a potent therapy during chronic Friend retrovirus infection. We demonstrate that the combination therapy induced a synergistic reinvigoration of the exhausted virus-specific CD8+ T cell immunity. Taken together, our results provide further information on how to improve PD-1-targeted therapies during chronic viral infection and cancer.
Copyright © 2021 Knuschke et al.
-
FC/FACS
-
Immunology and Microbiology
Hypoxia-inducible factor 1 alpha limits dendritic cell stimulation of CD8 T cell immunity.
In PLoS ONE on 1 January 2021 by Tran, C. W., Gold, M. J., et al.
Dendritic cells are sentinels of the immune system and represent a key cell in the activation of the adaptive immune response. Hypoxia-inducible factor 1 alpha (HIF-1α)-a crucial oxygen sensor stabilized during hypoxic conditions-has been shown to have both activating and inhibitory effects in immune cells in a context- and cell-dependent manner. Previous studies have demonstrated that in some immune cell types, HIF-1α serves a pro-inflammatory role. Genetic deletion of HIF-1α in macrophages has been reported to reduce their pro-inflammatory function. In contrast, loss of HIF-1α enhanced the pro-inflammatory activity of dendritic cells in a bacterial infection model. In this study, we aimed to further clarify the effects of HIF-1α in dendritic cells. Constitutive expression of HIF-1α resulted in diminished immunostimulatory capacity of dendritic cells in vivo, while conditional deletion of HIF-1α in dendritic cells enhanced their ability to induce a cytotoxic T cell response. HIF-1α-expressing dendritic cells demonstrated increased production of inhibitory mediators including IL-10, iNOS and VEGF, which correlated with their reduced capacity to drive effector CD8+ T cell function. Altogether, these data reveal that HIF-1α can promote the anti-inflammatory functions of dendritic cells and provides insight into dysfunctional immune responses in the context of HIF-1α activation.
-
Immunology and Microbiology
In Scientific Reports on 17 August 2020 by Xu, K., Wu, N., et al.
Airway dendritic cells (DCs) are recognized as important factors in the mechanisms of allergic inflammatory diseases. Suppressor of cytokine signaling 3 (SOCS3) is involved in regulating the functions of T cells and macrophages, but the roles of SOCS3-expressing DCs in the pathogeneses of allergic inflammatory diseases are still controversial. We compared the effects of adoptively transferred SOCS3-/- and SOCS3+/+ bone marrow-derived DCs (BMDCs) on airway inflammation in ovalbumin (OVA)-sensitized asthmatic mice. Adoptive transfer of mature DCs (lipopolysaccharide [LPS]-induced DCs, DClps) with or without SOCS3 gene expression significantly ameliorated allergic airway inflammation. SOCS3-/- DCs slightly attenuated BMDC-induced immunogenic tolerance. DClps migrated to OVA-sensitized lungs with higher efficiency than immature DCs (DCim). DClps with or without SOCS3 greatly improved lung pathology scores and alleviated airway inflammatory cell infiltration after adoptive transfer into mice; they also increased interleukin-10 (IL-10) and transforming growth factor-β (TGF-β) production and inhibited signal transducer and activator of transcription (STAT) 4 and STAT6 signaling in the lungs after OVA sensitization. In conclusion, the BMDC adoptive transfer-induced immunogenic tolerance in OVA-sensitized mice might not be due to SOCS3 gene depletion. BMDC adoptive transfer may be developed into a new approach that alleviates asthma by modulating the balance between immune tolerance and inflammation.
-
Immunology and Microbiology
In Cancers on 10 July 2020 by Khou, S., Popa, A., et al.
Cutaneous squamous cell carcinoma (cSCC) development has been linked to immune dysfunctions but the mechanisms are still unclear. Here, we report a progressive infiltration of tumor-associated neutrophils (TANs) in precancerous and established cSCC lesions from chemically induced skin carcinogenesis. Comparative in-depth gene expression analyses identified a predominant protumor gene expression signature of TANs in lesions compared to their respective surrounding skin. In addition, in vivo depletion of neutrophils delayed tumor growth and significantly increased the frequency of proliferating IFN-γ (interferon-γ)-producing CD8+ T cells. Mechanisms that limited antitumor responses involved high arginase activity, production of reactive oxygen species (ROS) and nitrite (NO), and the expression of programmed death-ligand 1 (PD-L1) on TAN, concomitantly with an induction of PD-1 on CD8+ T cells, which correlated with tumor size. Our data highlight the relevance of targeting neutrophils and PD-L1-PD-1 (programmed death-1) interaction in the treatment of cSCC.
-
Cancer Research
-
Immunology and Microbiology
In MBio on 2 February 2021 by Knuschke, T., Kollenda, S., et al.
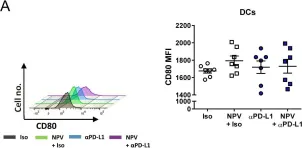
Fig.2.A

-
FC/FACS
-
Collected and cropped from MBio by CiteAb, provided under a CC-BY license
Image 1 of 4
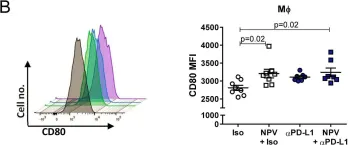
In MBio on 2 February 2021 by Knuschke, T., Kollenda, S., et al.
Fig.2.B

-
FC/FACS
-
Collected and cropped from MBio by CiteAb, provided under a CC-BY license
Image 1 of 4
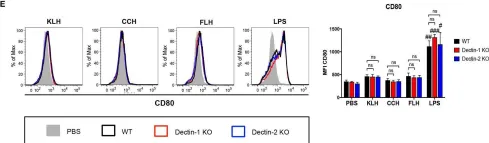
In Front Immunol on 20 June 2019 by Jimenez, J. M., Salazar, M. L., et al.
Fig.3.E

-
FC/FACS
-
Collected and cropped from Front Immunol by CiteAb, provided under a CC-BY license
Image 1 of 4
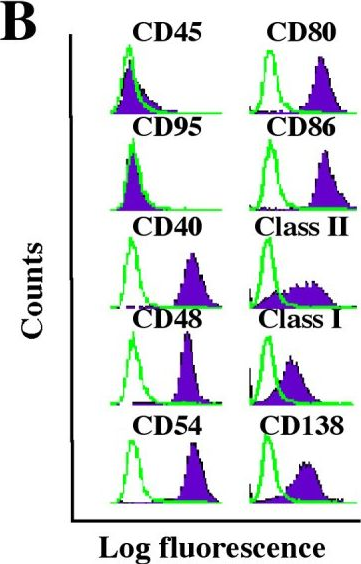
In Mol Cancer on 9 November 2005 by Han, S. S., Shaffer, A. L., et al.
Fig.1.B
-
FC/FACS
-
Mus musculus (House mouse)
Collected and cropped from Mol Cancer by CiteAb, provided under a CC-BY license
Image 1 of 4