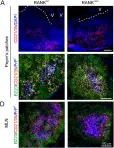
Germinal centers (GCs) are essential for antibody affinity maturation. GC B cells have a unique repertoire of cell surface glycans compared with naive B cells, yet functional roles for changes in glycosylation in the GC have yet to be ascribed. Detection of GCs by the antibody GL7 reflects a downregulation in ligands for CD22, an inhibitory co-receptor of the B cell receptor. To test a functional role for downregulation of CD22 ligands in the GC, we generate a mouse model that maintains CD22 ligands on GC B cells. With this model, we demonstrate that glycan remodeling plays a critical role in the maintenance of B cells in the GC. Sustained expression of CD22 ligands induces higher levels of apoptosis in GC B cells, reduces memory B cell and plasma cell output, and delays affinity maturation of antibodies. These defects are CD22 dependent, demonstrating that downregulation of CD22 ligands on B cells plays a critical function in the GC.
Copyright © 2022 The Author(s). Published by Elsevier Inc. All rights reserved.
Product Citations: 59
Coordinated changes in glycosylation regulate the germinal center through CD22.
In Cell Reports on 15 March 2022 by Enterina, J. R., Sarkar, S., et al.
-
ICC-IF
-
Mus musculus (House mouse)
In Frontiers in Immunology on 27 April 2021 by Pestka, J. J., Akbari, P., et al.
Workplace exposure to respirable crystalline silica dust (cSiO2) has been etiologically linked to the development of lupus and other human autoimmune diseases. Lupus triggering can be recapitulated in female NZBWF1 mice by four weekly intranasal instillations with 1 mg cSiO2. This elicits inflammatory/autoimmune gene expression and ectopic lymphoid structure (ELS) development in the lung within 1 week, ultimately driving early onset of systemic autoimmunity and glomerulonephritis. Intriguingly, dietary supplementation with docosahexaenoic acid (DHA), an ω-3 polyunsaturated fatty acid (PUFA) found in fish oil, beginning 2 week prior to cSiO2 challenge, prevented inflammation and autoimmune flaring in this novel model. However, it is not yet known how ω-3 PUFA intervention influences established autoimmunity in this murine model of toxicant-triggered lupus. Here we tested the hypothesis that DHA intervention after cSiO2-initiated intrapulmonary autoimmunity will suppress lupus progression in the NZBWF1 mouse. Six-week old NZWBF1 female mice were fed purified isocaloric diet for 2 weeks and then intranasally instilled with 1 mg cSiO2 or saline vehicle weekly for 4 consecutive weeks. One week after the final instillation, which marks onset of ELS formation, mice were fed diets supplemented with 0, 4, or 10 g/kg DHA. One cohort of mice (n = 8/group) was terminated 13 weeks after the last cSiO2 instillation and assessed for autoimmune hallmarks. A second cohort of mice (n = 8/group) remained on experimental diets and was monitored for proteinuria and moribund criteria to ascertain progression of glomerulonephritis and survival, respectively. DHA consumption dose-dependently increased ω-3 PUFA content in the plasma, lung, and kidney at the expense of the ω-6 PUFA arachidonic acid. Dietary intervention with high but not low DHA after cSiO2 treatment suppressed or delayed: (i) recruitment of T cells and B cells to the lung, (ii) development of pulmonary ELS, (iii) elevation of a wide spectrum of plasma autoantibodies associated with lupus and other autoimmune diseases, (iv) initiation and progression of glomerulonephritis, and (v) onset of the moribund state. Taken together, these preclinical findings suggest that DHA supplementation at a human caloric equivalent of 5 g/d was an effective therapeutic regimen for slowing progression of established autoimmunity triggered by the environmental toxicant cSiO2.
Copyright © 2021 Pestka, Akbari, Wierenga, Bates, Gilley, Wagner, Lewandowski, Rajasinghe, Chauhan, Lock, Li and Harkema.
-
IHC
-
Mus musculus (House mouse)
-
Immunology and Microbiology
In The Journal of Immunology on 1 January 2021 by Wu, X., Thylur, R. P., et al.
Cytokine responses to malaria play important roles in both protective immunity development and pathogenesis. Although the roles of cytokines such as TNF-α, IL-12, IFN-γ, and IL-10 in immunity and pathogenesis to the blood stage malaria are largely known, the role of IL-4 remains less understood. IL-4 targets many cell types and induces multiple effects, including cell proliferation, gene expression, protection from apoptosis, and immune regulation. Accordingly, IL-4 has been exploited as a therapeutic for several inflammatory diseases. Malaria caused by Plasmodium falciparum manifests in many organ-specific fatal pathologies, including cerebral malaria (CM), driven by a high parasite load, leading to parasite sequestration in organs and consequent excessive inflammatory responses and endothelial damage. We investigated the therapeutic potential of IL-4 against fatal malaria in Plasmodium berghei ANKA-infected C57BL/6J mice, an experimental CM model. IL-4 treatment significantly reduced parasitemia, CM pathology, and mortality. The therapeutic effect of IL-4 is mediated through multiple mechanisms, including enhanced parasite clearance mediated by upregulation of phagocytic receptors and increased IgM production, and decreased brain inflammatory responses, including reduced chemokine (CXCL10) production, reduced chemokine receptor (CXCR3) and adhesion molecule (LFA-1) expression by T cells, and downregulation of cytotoxic T cell lytic potential. IL-4 treatment markedly reduced the infiltration of CD8+ T cells and brain pathology. STAT6, PI3K-Akt-NF-κB, and Src signaling mediated the cellular and molecular events that contributed to the IL-4-dependent decrease in parasitemia. Overall, our results provide mechanistic insights into how IL-4 treatment mitigates experimental CM and have implications in developing treatment strategies for organ-specific fatal malaria.
Copyright © 2020 by The American Association of Immunologists, Inc.
-
Immunology and Microbiology
Targeting Lymph Node Niches Enhances Type 1 Immune Responses to Immunization.
In Cell Reports on 26 May 2020 by Lian, J., Ozga, A. J., et al.
Generating robust CD4+ T-helper cell type 1 (Th1) responses is essential for protective vaccine-induced type 1 immunity. Here, we examine whether immunization formulation associated with enhanced vaccine efficacy promotes antigen targeting and cell recruitment into lymph node (LN) niches associated with optimal type 1 responses. Immunization with antigen and Toll-like receptor agonist emulsified in oil leads to an increased differentiation of IFNγ/TNF-α+ polyfunctional Th1 cells compared to an identical immunization in saline. Oil immunization results in a rapid delivery and persistence of antigen in interfollicular regions (IFRs) of the LN, whereas without oil, antigen is distributed in the medullary region. Following oil immunization, CXCL10-producing inflammatory monocytes accumulate in the IFR, which mobilizes antigen-specific CD4+ T cells into this niche. In this microenvironment, CD4+ T cells are advantageously positioned to encounter arriving IL-12-producing inflammatory dendritic cells (DCs). These data suggest that formulations delivering antigen to the LN IFR create an inflammatory niche that can improve vaccine efficacy.
Copyright © 2020 The Author(s). Published by Elsevier Inc. All rights reserved.
-
Mus musculus (House mouse)
-
Immunology and Microbiology
Widespread organ tolerance to Xist loss and X reactivation except under chronic stress in the gut.
In Proceedings of the National Academy of Sciences of the United States of America on 25 February 2020 by Yang, L., Yildirim, E., et al.
Long thought to be dispensable after establishing X chromosome inactivation (XCI), Xist RNA is now known to also maintain the inactive X (Xi). To what extent somatic X reactivation causes physiological abnormalities is an active area of inquiry. Here, we use multiple mouse models to investigate in vivo consequences. First, when Xist is deleted systemically in post-XCI embryonic cells using the Meox2-Cre driver, female pups exhibit no morbidity or mortality despite partial X reactivation. Second, when Xist is conditionally deleted in epithelial cells using Keratin14-Cre or in B cells using CD19-Cre, female mice have a normal life span without obvious illness. Third, when Xist is deleted in gut using Villin-Cre, female mice remain healthy despite significant X-autosome dosage imbalance. Finally, when the gut is acutely stressed by azoxymethane/dextran sulfate (AOM/DSS) exposure, both Xist-deleted and wild-type mice develop gastrointestinal tumors. Intriguingly, however, under prolonged stress, mutant mice develop larger tumors and have a higher tumor burden. The effect is female specific. Altogether, these observations reveal a surprising systemic tolerance to Xist loss but importantly reveal that Xist and XCI are protective to females during chronic stress.
In PLoS Pathog on 1 December 2016 by Donaldson, D. S., Sehgal, A., et al.
Fig.3.A

-
ICC
-
Mus musculus (House mouse)
Collected and cropped from PLoS Pathog by CiteAb, provided under a CC-BY license
Image 1 of 1