The CD47/SIRPα axis conveys a 'don't eat me' signal, thereby thwarting the phagocytic clearance of tumor cells. Although blocking antibodies targeting CD47 have demonstrated promising anti-tumor effects in preclinical models, clinical trials involving human cancer patients have not yielded ideal results. Exploring the regulatory mechanisms of CD47 is imperative for devising more efficacious combinational therapies. Here, we report that inhibiting USP2 prompts CD47 degradation and reshapes the tumor microenvironment (TME), thereby enhancing anti-PD-1 immunotherapy. Mechanistically, USP2 interacts with CD47, stabilizing it through deubiquitination. USP2 inhibition destabilizes CD47, thereby boosting macrophage phagocytosis. Single-cell RNA sequencing shows USP2 inhibition reprograms TME, evidenced by increasing M1 macrophages and CD8+ T cells while reducing M2 macrophages. Combining ML364 with anti-PD-1 reduces tumor burden in mouse models. Clinically, low USP2 expression predicts a better response to anti-PD-1 treatment. Our findings uncover the regulatory mechanism of CD47 by USP2 and targeting this axis boosts anti-tumor immunity.
© 2025. The Author(s).
Product Citations: 86
USP2 inhibition unleashes CD47-restrained phagocytosis and enhances anti-tumor immunity.
In Nature Communications on 16 May 2025 by Dai, P., Sun, Y., et al.
-
Cancer Research
-
Immunology and Microbiology
In Ultrasonics Sonochemistry on 1 January 2025 by Yang, G., Li, H., et al.
The hypoxic and immunosuppressive tumor microenvironment (TME) remains a major obstacle to impede cancer immunotherapy. Here, we found that sononeoperfusion-a new effect of tumor perfusion enhancement induced by low mechanical index ultrasound stimulated microbubble cavitation (USMC)-ameliorated tumor tissue oxygenation and induced tumor vascular normalization (TVN). This TVN might be associated with the down-regulation of hypoxia-inducible factor 1-alpha (HIF-1α) and vascular endothelial growth factor (VEGF) within tumors. Moreover, the sononeoperfusion effect reduced the accumulation of immunosuppressive cells, such as regulatory T cells (Tregs), myeloid-derived suppressor cells (MDSCs) and M2-like tumor-associated macrophages (M2-TAMs), and decreased the production of immune inhibitory factors like transforming growth factor-β1 (TGF-β1), interleukin 10 (IL-10), chemoattractant chemokines CC-chemokine ligand 22 (CCL22), CCL28, adenosine and lactate within tumors. Notably, flow cytometry analysis revealed that sononeoperfusion not only increased the percentage of tumor infiltrating-CD8+ T cells, but also promoted the generation of interferon-γ (IFN-γ) and tumor necrosis factor-α (TNF-α) by these cells. Furthermore, the improved immune TME by sononeoperfusion effect sensitized anti-PD-L1 treatment both in MC38 colon cancer and Lewis lung carcinoma mice, resulting in tumor regression and prolonged survival. Mechanically, the enhanced efficacy of combination therapy was mainly based on promoting the infiltration and function of CD8+ T cells within tumors. Together, sononeoperfusion could ameliorate hypoxia and immunosuppression in the TME, thereby potentiating anti-PD-L1 therapy for solid tumors. This novel method of USMC generating sononeoperfusion effect may provide a new therapeutic modality for facilitating cancer immunotherapy.
Copyright © 2024 The Author(s). Published by Elsevier B.V. All rights reserved.
-
FC/FACS
-
Cancer Research
In Frontiers in Immunology on 17 September 2024 by Zhou, P., Watt, J., et al.
The outbreak of coronavirus disease 19 (COVID-19) has highlighted the demand for vaccines that are safe and effective in inducing systemic and airway mucosal immunity against the aerosol transmission of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). In this study, we developed a novel helper-dependent adenoviral vector-based COVID-19 mucosal vaccine encoding a full-length SARS-CoV-2 spike protein (HD-Ad-FS). Through intranasal immunization (single-dose and prime-boost regimens), we demonstrated that the HD-Ad-FS was immunogenic and elicited potent systemic and airway mucosal protection in BALB/c mice, transgenic ACE2 (hACE2) mice, and hamsters. We detected high titers of neutralizing antibodies (NAbs) in sera and bronchoalveolar lavages (BALs) in the vaccinated animals. High levels of spike-specific secretory IgA (sIgA) and IgG were induced in the airway of the vaccinated animals. The single-dose HD-Ad-FS elicited a strong immune response and protected animals from SARS-CoV-2 infection. In addition, the prime-boost vaccination induced cross-reactive serum NAbs against variants of concern (VOCs; Beta, Delta, and Omicron). After challenge, VOC infectious viral particles were at undetectable or minimal levels in the lower airway. Our findings highlight the potential of airway delivery of HD-Ad-FS as a safe and effective vaccine platform for generating mucosal protection against SARS-CoV-2 and its VOCs.
Copyright © 2024 Zhou, Watt, Mai, Cao, Li, Chen, Duan, Quan, Gingras, Rini, Hu and Liu.
-
FC/FACS
-
Mus musculus (House mouse)
-
COVID-19
-
Immunology and Microbiology
-
Veterinary Research
In Scientific Reports on 20 August 2024 by Yamamoto, W., Hamada, T., et al.
General anesthesia is thought to suppress the immune system and negatively affect postoperative infection and the long-term prognosis of cancer. However, the mechanism underlying immunosuppression induced by general anesthetics remains unclear. In this study, we focused on propofol, which is widely used for sedation under general anesthesia and intensive care and examined its effects on the T cell function and T cell-dependent immune responses. We found that propofol suppressed T cell glycolytic metabolism, differentiation into effector T cells, and cytokine production by effector T cells. CD8 T cells activated and differentiated into effector cells in the presence of propofol in vitro showed reduced antitumor activity. Furthermore, propofol treatment suppressed the increase in the number of antigen-specific CD8 T cells during Listeria infection. In contrast, the administration of propofol improved inflammatory conditions in mouse models of inflammatory diseases, such as OVA-induced allergic airway inflammation, hapten-induced contact dermatitis, and experimental allergic encephalomyelitis. These results suggest that propofol may reduce tumor and infectious immunity by suppressing the T cell function and T cell-dependent immune responses while improving the pathogenesis and prognosis of chronic inflammatory diseases by suppressing inflammation.
© 2024. The Author(s).
-
Mus musculus (House mouse)
-
Immunology and Microbiology
PTEN acts as a crucial inflammatory checkpoint controlling TLR9/IL-6 axis in B cells.
In IScience on 19 July 2024 by Tsai, P. J., Chen, M. Y., et al.
Phosphatase and tensin homolog (PTEN) is vital for B cell development, acting as a key negative regulator in the PI3K signaling pathway. We used CD23-cre to generate PTEN-conditional knockout mice (CD23-cKO) to examine the impact of PTEN mutation on peripheral B cells. Unlike mb1-cre-mediated PTEN deletion in early B cells, CD23-cKO mutants exhibited systemic inflammation with increased IL-6 production in mature B cells upon CpG stimulation. Inflammatory B cells in CD23-cKO mice showed elevated phosphatidylinositol 3-phosphate [PI(3)P] levels and increased TLR9 endosomal localization. Pharmacological inhibition of PI(3)P synthesis markedly reduced TLR9-mediated IL-6. Single-cell RNA-sequencing (RNA-seq) revealed altered endocytosis, BANK1, and NF-κB1 expression in PTEN-deficient B cells. Ectopic B cell receptor (BCR) expression on non-inflammatory mb1-cKO B cells restored BANK1 and NF-κB1 expression, enhancing TLR9-mediated IL-6 production. Our study highlights PTEN as a crucial inflammatory checkpoint, regulating TLR9/IL-6 axis by fine-tuning PI(3)P homeostasis. Additionally, BCR downregulation prevents the differentiation of inflammatory B cells in PTEN deficiency.
© 2024 The Author(s).
-
Mus musculus (House mouse)
-
Immunology and Microbiology
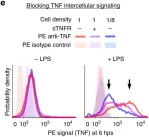
In Nat Commun on 13 February 2020 by Muldoon, J. J., Chuang, Y., et al.
Fig.1.C

-
FC/FACS
-
Collected and cropped from Nat Commun by CiteAb, provided under a CC-BY license
Image 1 of 3
In Nat Commun on 13 February 2020 by Muldoon, J. J., Chuang, Y., et al.
Fig.1.D

-
FC/FACS
-
Collected and cropped from Nat Commun by CiteAb, provided under a CC-BY license
Image 1 of 3
In Nat Commun on 13 February 2020 by Muldoon, J. J., Chuang, Y., et al.
Fig.1.E

-
FC/FACS
-
Collected and cropped from Nat Commun by CiteAb, provided under a CC-BY license
Image 1 of 3