The renin-angiotensin system (RAS) is essential for normal kidney development. Dysregulation of the RAS during embryogenesis can result in kidney abnormalities. To explore how angiotensin type 1 receptor (AT1R) signaling modulates nephron progenitor (NP) fate specification, we used induced pluripotent stem cell (iPSC) derived human kidney organoids treated with angiotensin II (Ang II) or the AT1R blocker losartan during differentiation. Ang II promoted NP proliferation and differentiation preferentially toward a podocyte fate, depleted the podocyte precursor population, and accelerated glomerular maturation. By contrast, losartan expanded the podocyte precursor population, delayed podocyte differentiation, and regressed the transcriptional signature to a more immature fetal state. Overall, using various in silico approaches with validation by RNAscope, we identified a role for AT1R signaling in regulating NP fate during nephrogenesis in kidney organoids. Our work supports the use of RAS modulators to improve organoid maturation and suggests that RAS may be a determinant of nephron endowment in vivo.
© The Author(s) 2025. Published by Oxford University Press.
Product Citations: 73
In Stem Cells on 15 May 2025 by Chung, H., Rahmani, W., et al.
-
Stem Cells and Developmental Biology
Cyclic jetting enables microbubble-mediated drug delivery.
In Nature Physics on 18 April 2025 by Cattaneo, M., Guerriero, G., et al.
The pursuit of targeted therapies capable of overcoming biological barriers, including the blood-brain barrier, has spurred the investigation of stimuli-responsive microagents that can improve therapeutic efficacy and reduce undesirable side effects. Intravenously administered, ultrasound-responsive microbubbles are promising agents with demonstrated potential in clinical trials, but the mechanism underlying drug absorption remains unclear. Here we show that ultrasound-driven single microbubbles puncture the cell membrane and induce drug uptake through stable cyclic microjets. Our theoretical models successfully reproduce the observed bubble and cell dynamic responses. We find that cyclic jets arise from shape instabilities, as opposed to classical inertial jets that are driven by pressure gradients, enabling microjet formation at mild ultrasound pressures below 100 kPa. We also establish a threshold for bubble radial expansion beyond which microjets form and facilitate cellular permeation and show that the stress generated by microjetting outperforms previously suggested mechanisms by at least an order of magnitude. Overall, this work elucidates the physics behind microbubble-mediated targeted drug delivery and provides the criteria for its effective and safe application.
© The Author(s) 2025.
In Frontiers in Pharmacology on 11 April 2025 by Shen, C., Wang, Q., et al.
Celastrol, an active ingredient derived from Tripterygium wilfordii Hook F, has shown therapeutic potential for various kidney renal diseases. The kidney protective activity of celastrol is mainly exerted through anti-inflammatory, and antioxidant effects. However, celastrol causes dose-dependent kidney toxicity, which results in increased risks of mortality among patients. This study aimed to develop a kidney organoid-based prediction system to assess the safety and efficacy of celastrol in reducing cisplatin-induced nephrotoxicity.
We investigated the ability of celastrol to reduce cisplatin-induced nephrotoxicity using kidney organoids. Kidney organoids were cultured and characterized, exhibiting renal tubular and glomerular structures and expressing specific kidney markers such as NPHS1, CD31, LTL, and SLC12A1. Data were obtained from in vitro experiments in which kidney organoids were exposed to therapeutically relevant concentrations or a toxic dosing profile of cisplatin and celastrol, to assess their impact on cell viability using flow cytometry and Acridine Orange/Propidium Iodide (AO/PI) staining. In addition, RNA-seq analyses were performed to determine the mechanisms of celastrol function in the kidney.
Kidney organoids exposed to 50 µM cisplatin showed significantly increased cell death (only 0.37% cells with normal cell structure), whereas celastrol under 5 µM (56% cells with normal cell structure) showed significantly less nephrotoxicity than cisplatin. The protective effects of celastrol against cisplatin-induced nephrotoxicity were further investigated by treating the organoids with both compounds. The results demonstrated that 2 µM celastrol reduced cisplatin-induced nephrotoxicity by downregulating SNORD3A and HIST1H3A gene levels.
This study highlights the potential of celastrol as a protective compound against cisplatin-induced kidney damage and emphasizes the importance of using advanced models, such as iPSC-derived kidney organoids, to predict therapeutic effect and nephrotoxic concentrations of novel drugs.
Copyright © 2025 Shen, Wang, Ye, Zhou, Xing, Pan, Li, Wu and You.
-
IHC-IF
-
Homo sapiens (Human)
-
Pharmacology
In Journal of Extracellular Biology on 1 May 2024 by Arif, S., Richer, M., et al.
Microvesicles (MVs) are a subtype of extracellular vesicles that can transfer biological information from their producer cells to target cells. This communication can in turn affect both normal and pathological processes. Mounting evidence has revealed that dermal wound myofibroblasts (Wmyo) produce MVs, which can transfer biomolecules impacting receptor cells such as human dermal microvascular endothelial cells (HDMECs). While the effects of MVs on HDMECs are generally well described in the literature, little is known about the transport of MVs across the HDMEC barrier, and their potential effect on the barrier integrity remains unknown. Here, we investigated these roles of Wmyo-derived MVs on two sub-populations of HDMECs, blood endothelial cells (BECs) and lymphatic endothelial cells (LECs). Using an in vitro model to mimic the endothelial barrier, we showed that MVs crossed the LEC barrier but not the BEC barrier. In addition, we demonstrated that MVs were able to influence the cell-cell junctions of HDMECs. Specifically, we observed that after internalization via the predominantly caveolin-dependent pathway, MVs induced the opening of junctions in BECs. Conversely, in LECs, MVs mainly use the macropinocytosis pathway and induce closure of these junctions. Moreover, proteins in the MV membrane were responsible for this effect, but not specifically those belonging to the VEGF family. Finally, we found that once the LEC barrier permeability was reduced by MV stimuli, MVs ceased to cross the barrier. Conversely, when the BEC barrier was rendered permeable following stimulation with MVs, they were subsequently able to cross the barrier via the paracellular pathway. Taken together, these results suggest that the study of Wmyo-derived MVs offers valuable insights into their interaction with the HDMEC barrier in the context of wound healing. They highlight the potential significance of these MVs in the overall process.
© 2024 The Authors. Journal of Extracellular Biology published by Wiley Periodicals LLC on behalf of International Society for Extracellular Vesicles.
-
Homo sapiens (Human)
-
Cardiovascular biology
SLC2A9 rs16890979 reduces uric acid absorption by kidney organoids.
In Frontiers in Cell and Developmental Biology on 25 January 2024 by Wu, S., Li, C., et al.
Introduction: The excretion and absorption of uric acid (UA) by the kidneys helps regulate serum UA levels. GLUT9, encoded by SLC2A9, is mainly expressed in the renal tubules responsible for UA absorption. SLC2A9 polymorphisms are associated with different serum UA levels. However, the lack of proper in vitro models has stalled research on the mechanisms of single nucleotide polymorphisms (SNPs) that affect UA metabolism in human urate transporters. Methods: In this study, we constructed a gene-edited human embryonic stem cells-9 (ESC-H9) derived kidney organoid bearing rs16890979, an SLC2A9 missense mutation with undetermined associations with hyperuricemia or hypouricemia. Kidney organoids derived from ESC-H9 with genetical overexpression (OE) and low expression (shRNA) of SLC2A9 to serve as controls to study the function of SLC2A9. The function of rs16890979 on UA metabolism was evaluated after placing the organoids to urate-containing medium and following histopathological analysis. Results: The kidney organoids with heterozygous or homozygous rs16890979 mutations showed normal SLC2A9 expression levels and histological distribution, phenotypically similar to the wild-type controls. However, reduced absorption of UA by the kidney organoids with rs16890979 mutants was observed. This finding together with the observation that UA absorption is increased in organoids with SLC2A9 overexpression and decreased in those with SLC2A9 knockdown, suggest that GLUT9 is responsible for UA absorption, and the rs16890979 SNP may compromise this functionality. Moreover, epithelial-mesenchymal transition (EMT) was detected in organoids after UA treatment, especially in the kidney organoid carrying GLUT9OE, suggesting the cytobiological mechanism explaining the pathological features in hyperuricosuria-related renal injury. Discussion: This study showing the transitional value of kidney organoid modeling the function of SNPs on UA metabolism. With a defined genetic background and a confirmed UA absorption function should be useful for studies on renal histological, cellular, and molecular mechanisms with this organoid model.
Copyright © 2024 Wu, Li, Li, Liu, Rong, Pei, Li, Zeng and Mao.
In Nat Commun on 29 November 2019 by Subramanian, A., Sidhom, E. H., et al.
Fig.4.D

-
IHC
-
Homo sapiens (Human)
Collected and cropped from Nat Commun by CiteAb, provided under a CC-BY license
Image 1 of 2
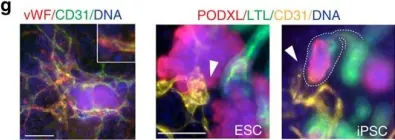
In Nat Commun on 23 October 2015 by Freedman, B. S., Brooks, C. R., et al.
Fig.2.G

-
ICC
-
Homo sapiens (Human)
Collected and cropped from Nat Commun by CiteAb, provided under a CC-BY license
Image 1 of 2