Monomorphic epitheliotropic intestinal T-cell lymphoma (MEITL) is a rare and aggressive primary intestinal lymphoma with a poor prognosis. MEITL can metastasize to the central nervous system, liver and spleen, but gallbladder involvement has not yet been reported. The present study describes the case of a 57-year-old woman who presented with abdominal distention, pain and vomiting. Contrast-enhanced computed tomography revealed thickening and perforation of the small intestinal wall, and a gallbladder mass. Histopathological analysis of the affected small intestine and gallbladder revealed a dense infiltrate of medium-sized monomorphic lymphocytes with a CD3+, CD4-, CD8+ and TIA-1+ phenotype. Based on the absence of celiac disease, aggressive clinical course, and characteristic histopathological and immunophenotypic features, a diagnosis of MEITL with gallbladder involvement was established. The patient underwent small intestinal resection and cholecystectomy, followed by chemotherapy, which was completed without gastrointestinal or gallbladder perforation. Diagnostic resection is currently the best approach for suspected malignant lymphoma of the gallbladder. This rare case of MEITL with gallbladder involvement highlights the importance of considering this diagnosis in similar clinical scenarios and the role of cholecystectomy, which can serve both diagnostic and therapeutic purposes.
Copyright: © 2025 Okuda et al.
Product Citations: 91
Monomorphic epitheliotropic intestinal T-cell lymphoma with gallbladder involvement: A case report.
In Molecular and Clinical Oncology on 1 July 2025 by Okuda, T., Shirase, T., et al.
-
Cancer Research
-
Immunology and Microbiology
KSHV vIL6 Inhibits Functional B Cell Maturation During De Novo Infection.
In Journal of Medical Virology on 1 July 2025 by Zakir, W., Osborn, J. M., et al.
Despite causative links to lymphoproliferative disorders, little is known about early events governing KSHV infection in B lymphocytes. IL-6 signaling plays a critical role in KSHV-mediated disease, with human IL-6 (hIL6) levels correlating with viral load and disease progression. This dynamic is even more complex due to the coexistence of hIL6 and KSHV-encoded viral IL-6 (vIL6) in these diseases. We hypothesize that hIL6 and vIL6 play critical, separable and collective roles in the early stages of KSHV infection in B cells. In this study, we use our ex vivo model of KSHV infection in human tonsil lymphocytes to investigate the relative contributions of vIL6 and hIL6 to early infection events in human B cells. We demonstrate that vIL6 and hIL6 collectively suppress KSHV infection in B cells restricting the distribution of KSHV within B cell subsets. We show that vIL6 manipulates hIL6 expression in a subset-specific manner, and that vIL6 and hIL6 differentially influence the differentiation of germinal center and plasmablasts. Taken together, these results suggest a novel paradigm in which KSHV uses vIL6 to abrogate the GC-mediated maturation pathway for antibody secreting cells that is driven by hIL6 signaling.
© 2025 The Author(s). Journal of Medical Virology published by Wiley Periodicals LLC.
-
Immunology and Microbiology
In Nature Communications on 30 May 2025 by Wu, Z., Gao, S., et al.
Severe immune aplastic anemia is a fatal disease due to the destruction of marrow hematopoietic cells by cytotoxic lymphocytes, serving as a paradigm for marrow failure syndromes and autoimmune diseases. To better understand its pathophysiology, we apply advanced single cell methodologies, including mass cytometry, single-cell RNA, and TCR/BCR sequencing, to patient samples from a clinical trial of immunosuppression and growth factor stimulation. We observe opposing changes in the abundance of myeloid cells and T cells, with T cell clonal expansion dominated by effector memory cells. Therapy reduces and suppresses cytotoxic T cells, but new T cell clones emerge hindering robust hematopoietic recovery. Enhanced cell-cell interactions including between hematopoietic cells and immune cells, in particular evolving IFNG and IFNGR, are noted in patients and are suppressed post-therapy. Hematologic recovery occurs with increases in the progenitor rather than stem cells. Genetic predispositions linked to immune activation genes enhances cytotoxic T cell activity and crosstalk with target cells.
© 2025. This is a U.S. Government work and not under copyright protection in the US; foreign copyright protection may apply.
-
Immunology and Microbiology
In Frontiers in Nutrition on 8 May 2025 by Lian, J., Men, Z., et al.
Folic acid has been associated with fetal development, especially in fetal immunity. Therefore, limited evidence regarding the effects of different folic acid supplementation of hepatitis B surface antigen (HBsAg) positive mothers in innate immunity in offspring. Herein, this study aimed to explore the association between folic acid supplementation and the innate immunity of neonates and the immunological efficacy of hepatitis B vaccine (HepB), which may provide insights that could inform pre-pregnancy health management in HBsAg-positive mothers.
It is an ambispective cohort study with 293 pairs of HBsAg-positive mothers-offspring in Taiyuan, Shanxi Province, China. Mothers were classified into three groups according to the time of starting folic acid supplementation, non-supplementation group, pre-pregnancy group and post-pregnancy supplementation group. Immunological indexes such as immune cells proportion and innate immune mediators in cord blood and anti-HBs in infants were measured. Differences in immunological indexes were analyzed by One-Way ANOVA test. Univariate and multivariate analyses were performed for factors associated with abnormal immunological indexes and potential confounders were adjusted.
The preconception folic acid group showed a significantly higher expression levels of STING (P = 0.005) and pNF-κB (P = 0.010) in cord blood along with higher anti-HBs titres (P = 0.006), when compared to both non-supplementation group and post-pregnancy supplementation group. Higher anti-HBs levels indicate a stronger immune response to HepB and may enhance protection against HBV infection during early life. Infants in the high pNF-κB expression group exhibited a significantly elevated seropositive rate of HepB compared to those in the low pNF-κB expression group (P = 0.037). There were no mediation effects and no moderation effects in this study, potentially due to the direct influence of folic acid supplementation on immune responses or the limited sample size.
In conclusion, our findings demonstrate that preconception folic acid supplementation may enhance HepB vaccine responsiveness in infants of HBsAg-positive mothers. Meanwhile, high pNF-κB expression in cord blood can increase seropositive rates in infants. This discovery has significant public health implications, as it may provide a simple and accessible intervention to improve vaccination outcomes and reduce HBV transmission in endemic regions.
Copyright © 2025 Lian, Men, Xu, Li, Li, Wang, Yao, Li, Qu, Feng and Wang.
-
Immunology and Microbiology
In Nature Communications on 1 April 2025 by Feist, W. N., Luna, S. E., et al.
Allogeneic transplantation of CCR5 null hematopoietic stem and progenitor cells (HSPCs) is the only known cure for HIV-1 infection. However, this treatment is limited because of the rarity of CCR5-null matched donors, the morbidities associated with allogeneic transplantation, and the prevalence of HIV-1 strains resistant to CCR5 knockout (KO) alone. Here, we propose a one-time therapy through autologous transplantation of HSPCs genetically engineered ex vivo to produce both CCR5 KO cells and long-term secretion of potent HIV-1 inhibiting antibodies from B cell progeny. CRISPR-Cas9-engineered HSPCs engraft and reconstitute multiple hematopoietic lineages in vivo and can be engineered to express multiple antibodies simultaneously (in pre-clinical models). Human B cells engineered to express each antibody secrete neutralizing concentrations capable of inhibiting HIV-1 pseudovirus infection in vitro. This work lays the foundation for a potential one-time functional cure for HIV-1 through combining the long-term delivery of therapeutic antibodies against HIV-1 and the known efficacy of CCR5 KO HSPC transplantation.
© 2025. The Author(s).
-
Immunology and Microbiology
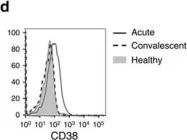
In Nat Commun on 23 June 2016 by van Wilgenburg, B., Scherwitzl, I., et al.
Fig.1.D

-
FC/FACS
-
Collected and cropped from Nat Commun by CiteAb, provided under a CC-BY license
Image 1 of 1