The ovary is the first organ to age in the human body, affecting both fertility and overall health. However, the biological mechanisms underlying human ovarian aging remain poorly understood. Here we present a comprehensive single-nuclei multi-omics atlas of four young (ages 23-29 years) and four reproductively aged (ages 49-54 years) human ovaries. Our analyses reveal coordinated changes in transcriptomes and chromatin accessibilities across cell types in the ovary during aging, notably mTOR signaling being a prominent ovary-specific aging pathway. Cell-type-specific regulatory networks reveal enhanced activity of the transcription factor CEBPD across cell types in the aged ovary. Integration of our multi-omics data with genetic variants associated with age at natural menopause demonstrates a global impact of functional variants on gene regulatory networks across ovarian cell types. We nominate functional non-coding regulatory variants, their target genes and ovarian cell types and regulatory mechanisms. This atlas provides a valuable resource for understanding the cellular, molecular and genetic basis of human ovarian aging.
© 2024. The Author(s).
Product Citations: 227
Molecular and genetic insights into human ovarian aging from single-nuclei multi-omics analyses.
In Nature Aging on 1 February 2025 by Jin, C., Wang, X., et al.
-
Genetics
Noggin Combined With Human Dental Pulp Stem Cells to Promote Skeletal Muscle Regeneration.
In Stem Cells International on 6 January 2025 by Zhang, M. H., Yu, L., et al.
A proper source of stem cells is key to muscle injury repair. Dental pulp stem cells (DPSCs) are an ideal source for the treatment of muscle injuries due to their high proliferative and differentiation capacities. However, the current myogenic induction efficiency of human DPSCs hinders their use in muscle regeneration due to the unknown induction mechanism. In this study, we treated human DPSCs with Noggin, a secreted antagonist of bone morphogenetic protein (BMP), and discovered that Noggin can effectively promote myotube formation. We also found that Noggin can accelerate the skeletal myogenic differentiation (MyoD) of DPSCs and promote the generation of Pax7+ satellite-like cells. Noggin increased the expression of myogenic markers and the transcriptional and translational abundance of satellite cell (SC) markers in DPSCs. Moreover, BMP4 inhibited Pax7 expression and activated p-Smad1/5/9, while Noggin eliminated BMP4-induced p-Smad1/5/9 in DPSCs. This finding suggests that Noggin antagonizes BMP by downregulating p-Smad and facilitates the MyoD of DPSCs. Then, we implanted Noggin-pretreated DPSCs combined with Matrigel into the mouse tibialis anterior muscle with volumetric muscle loss (VML) and observed a 73% reduction in the size of the defect and a 69% decrease in scar tissue. Noggin-treated DPSCs can benefit the Pax7+ SC pool and promote muscle regeneration. This work reveals that Noggin can enhance the production of satellite-like cells from the MyoD of DPSCs by regulating BMP/Smad signaling, and these satellite-like cell bioconstructs might possess a relatively fast capacity for muscle regeneration.
Copyright © 2024 Meng-Han Zhang et al.
-
FC/FACS
-
Homo sapiens (Human)
-
Stem Cells and Developmental Biology
Identification of human cranio-maxillofacial skeletal stem cells for mandibular development.
In Science Advances on 3 January 2025 by Wang, Z., Wang, K., et al.
Compared with long bone that arises from the mesoderm, the major portion of the maxillofacial bones and the front bone of the skull are derived from cranial neural crest cells and undergo intramembranous ossification. Human skeletal stem cells have been identified in embryonic and fetal long bones. Here, we describe a single-cell atlas of the human embryonic mandible and identify a population of cranio-maxillofacial skeletal stem cells (CMSSCs). These CMSSCs are marked by interferon-induced transmembrane protein 5 (IFITM5) and are specifically located around the periosteum of the jawbone and frontal bone. Additionally, these CMSSCs exhibit strong self-renewal and osteogenic differentiation capacities but lower chondrogenic differentiation potency, mediating intramembranous bone formation without cartilage formation. IFITM5+ cells are also observed in the adult jawbone and exhibit functions similar to those of embryonic CMSSCs. Thus, this study identifies CMSSCs that orchestrate the intramembranous ossification of cranio-maxillofacial bones, providing a deeper understanding of cranio-maxillofacial skeletal development and promising seed cells for bone repair.
-
Stem Cells and Developmental Biology
In Materials Today. Bio on 1 December 2024 by Goluba, K., Parfejevs, V., et al.
Pancreatic ductal adenocarcinoma (PDAC) is a highly aggressive cancer characterised by poor survival rates and an increasing global incidence. Advances in the staging and categorization of pancreatic tumours, along with the discovery of functional mutations, have made precision treatments possible, which may lead to better clinical results. To further improve customized treatment approaches, in vitro models that can be used for functional drug sensitivity testing and precisely mimic the disease at the organ level are required. In this study, we present a workflow for creating a personalized PDAC chip utilising primary tumour-derived human pancreatic organoids (hPOs) and Human Umbilical Vein Endothelial Cells (HUVECs) to simulate the vascular barrier and tumour interactions within a PDMS-free organ-on-a-chip system. The patient PDAC tissue, expanded as tumour hPOs, could be cultured as adherent cells on the chip for more than 50 days, allowing continuous monitoring of cell viability through outflows from tumour and endothelial channels. Our findings demonstrate a gradual increase in cell density and cell turnover in the pancreatic tumor channel. Tumour-specific biomarkers, including CA-19.9, TIMP-1, Osteopontin, MIC-1, ICAM-1 and sAXL were consistently detected in the PDAC chip outflows. Comparative analyses between tissue culture plates and microfluidic conditions revealed significant differences in biomarker secretion patterns, highlighting the advantages of the microfluidics approach. This PDAC chip provides a stable, reproducible tumour model system with a functional endothelial cell barrier, suitable for drug sensitivity and secretory biomarker studies, thus serving as a platform for functional precision medicine application and multi-organ chip development.
© 2024 The Authors.
-
FC/FACS
-
Homo sapiens (Human)
-
Cancer Research
CRISPR screening uncovers nucleolar RPL22 as a heterochromatin destabilizer and senescence driver.
In Nucleic Acids Research on 28 October 2024 by Li, H. Y., Wang, M., et al.
Dysfunction of the ribosome manifests during cellular senescence and contributes to tissue aging, functional decline, and development of aging-related disorders in ways that have remained enigmatic. Here, we conducted a comprehensive CRISPR-based loss-of-function (LOF) screen of ribosome-associated genes (RAGs) in human mesenchymal progenitor cells (hMPCs). Through this approach, we identified ribosomal protein L22 (RPL22) as the foremost RAG whose deficiency mitigates the effects of cellular senescence. Consequently, absence of RPL22 delays hMPCs from becoming senescent, while an excess of RPL22 accelerates the senescence process. Mechanistically, we found in senescent hMPCs, RPL22 accumulates within the nucleolus. This accumulation triggers a cascade of events, including heterochromatin decompaction with concomitant degradation of key heterochromatin proteins, specifically heterochromatin protein 1γ (HP1γ) and heterochromatin protein KRAB-associated protein 1 (KAP1). Subsequently, RPL22-dependent breakdown of heterochromatin stimulates the transcription of ribosomal RNAs (rRNAs), triggering cellular senescence. In summary, our findings unveil a novel role for nucleolar RPL22 as a destabilizer of heterochromatin and a driver of cellular senescence, shedding new light on the intricate mechanisms underlying the aging process.
© The Author(s) 2024. Published by Oxford University Press on behalf of Nucleic Acids Research.
-
Biochemistry and Molecular biology
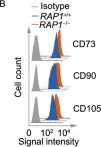
In Protein Cell on 1 September 2019 by Zhang, X., Liu, Z., et al.
Fig.2.B

-
FC/FACS
-
Homo sapiens (Human)
Collected and cropped from Protein Cell by CiteAb, provided under a CC-BY license
Image 1 of 2
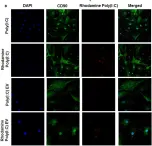
In Front Immunol on 13 February 2018 by Frank-Bertoncelj, M., Pisetsky, D. S., et al.
Fig.4.B

-
ICC-IF
-
Collected and cropped from Front Immunol by CiteAb, provided under a CC-BY license
Image 1 of 2