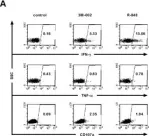
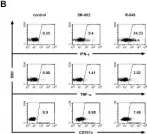
Chimeric antigen receptor (CAR) T-cells targeting CD19 have shown impressive outcomes in refractory/relapsed B-cell acute lymphoblastic leukaemia (r/r B-ALL); however, frequent relapse demands multi-targeted approaches.
We report Spanish clinical data on the safety and efficacy of tandem anti-CD19/CD22 CAR T-cells administered on a compassionate use basis in a cohort of 10 heavily pretreated paediatric, adolescent, and young adult (AYA) patients with r/r B-ALL.
Most (9/10) of the patients had relapsed B-ALL, 7 having received previous anti-CD19 CAR T-cell therapy and 6 haematopoietic stem cell transplantation (HSCT). Two patients had Down syndrome. Increased high-grade CRS/ICANS and proinflammatory markers (IL-6, LDH and ferritin) correlated with patients with a high tumour burden (TB) before lymphodepletion. Complete remission on day +28 post-infusion was achieved in 8/10 patients (7 with MRD-), and 5/7 patients received HSCT as consolidative therapy within three months post-infusion. Two patients with early relapse after tandem anti-CD19/CD22 CAR received rescue therapy and HSCT. At the 18-month follow up, overall survival (OS) was 70% (95% CI, 47%-100%).
Tandem anti-CD19/CD22 CAR T-cell administration combined with consolidative HSCT is a promising therapeutic approach, though managing bridging therapy and reducing the TB prior to infusion remain key challenges (REALL_CART trial, NCT06709469, EudraCT 2023-509723-41-01).
This work was supported by a grant from the Instituto de Salud Carlos III to APM PI22/01226, two grants from CRIS Cancer Foundation to Beat Cancer as part of the projects "Advanced Cell Therapy Unit Hospital Universitario La Paz" and JM "Proyecto Mateo: CAR T-cell therapy for juvenile myelomonocytic leukaemia" and "Terapia avanzada CAR-T CD19/CD22", Ayuda Nominativa de la Consejería de Investigación, Comunidad de Madrid, Spain. Work in MI lab was funded by a grant from the Spanish Ministry of Science and Innovation (PID2020-114148RB-I00). VGG was granted with Río Hortega (AES 2022 exp. Nº. CM22/00078) and Juan Rodés (AES 2024 exp. Nº. JR24/00003) contracts from the Carlos III Health Institute (ISCIII) through the European Funds of the Recovery, Transformation and Resilience Plan and financed by the European Union NextGenerationEU.
Copyright © 2025 The Author(s). Published by Elsevier B.V. All rights reserved.
Product Citations: 151
In EBioMedicine on 1 August 2025 by González-Martínez, B., Galán-Gómez, V., et al.
-
Immunology and Microbiology
In Cell Discovery on 10 June 2025 by Wang, Y., Ji, P., et al.
The discovery of broadly neutralizing antibodies (bNAbs) that target conserved epitopes on the HIV-1 envelope glycoprotein (Env) has garnered significant attention for its potential in the development of effective therapeutic and vaccine strategies. In this study, we isolated and characterized a CD4 binding site (CD4bs) antibody, FD22, from an elite neutralizer in China who had been infected with a clade B virus through contaminated blood plasma for 23 years. The heavy chain of FD22 was derived from a rarely reported IGHV3-30 germline gene and exhibited an exceptionally high degree of somatic hypermutation (SHM) (37%), along with a long and unique CDRH3 loop of 20-amino acids. FD22 exhibited potent and broad neutralizing activity, comparable to that of the well-known bNAb VRC01. It effectively neutralized 82% of a panel of 145 diverse HIV-1 pseudoviruses, including the two major circulating strains in China, CRF01_AE and CRF07_BC. FD22 bound strongly to HIV-1-infected cell lines, efficiently engaged FcγRIIIa receptors, triggered NK cell degranulation and the release of key cytokines such as IFN-γ and β-chemokines, and robustly induced antibody-dependent cellular cytotoxicity (ADCC) against HIV-1-infected target cells. Structural prediction for FD22 and the HIV Env SOSIP trimer performed by AlphaFold3, site-mutagenesis, and autologous virus reverse mutation assays revealed that the epitope of FD22 spans key CD4 binding site, including Loop D, the CD4 binding loop (CD4 BLP), and the V5 Loop. The unique long CDRH3 loop of FD22 interacts with the CD4 binding site through its negatively charged residue R102, distinguishing it from other CD4bs antibodies. Our findings provide valuable insights into the mechanisms of FD22 in viral neutralization and ADCC. The dual functionality of FD22 enhances its potential as a promising therapeutic antibody and offers new avenues for designing CD4bs-targeting vaccines with enhanced ADCC capabilities.
© 2025. The Author(s).
-
Immunology and Microbiology
In Nature Communications on 10 May 2025 by Chen, J. L., Wang, B., et al.
In 2022, a global mpox outbreak occurred, and remains a concern today. The T cell memory response to MPXV (monkeypox virus) infection has not been fully investigated. In this study, we evaluate this response in convalescent and MVA-BN (Modified Vaccinia Ankara - Bavarian Nordic) vaccinated individuals using VACV-infected cells. Strong CD8+ and CD4+ T cell responses are observed, and T cell responses are biased towards viral early expressed proteins. We identify seven immunodominant HLA-A*02:01 restricted MPXV-specific epitopes and focus our detailed phenotypic and scRNAseq analysis on the immunodominant HLA-A*02:01-G5R18-26-specific CD8+ T cell response. While tetramer+CD8+ T cells share similar differentiation and activation phenotypes, T cells from convalescent individuals show greater cytotoxicity, migratory potential to site of infection and TCR clonal expansion. Our data suggest that effective functional profiles of MPXV-specific memory T cells induced by Mpox infection may have an implication on the long-term protective responses to future infection.
© 2025. The Author(s).
-
ICC
-
Homo sapiens (Human)
-
Immunology and Microbiology
Preprint on BioRxiv : the Preprint Server for Biology on 20 April 2025 by Ormhøj, M., Munk, K. K., et al.
Adoptive cell therapy (ACT) has shown promising results in cancer treatment, however, achieving effective ex vivo expansion of potent, functionally active, and cytotoxic T cells remains challenging. To overcome this, we loaded the engineered cytokine Neoleukin-2/15 (Neo2/15) on our recently established artificial antigen-presenting scaffolds (Ag-scaffolds) to expand antigen-specific T cells. Neo2/15 selectively binds to IL-2Rβ/γ receptors, enhancing CD8 + T cell proliferation while limiting regulatory T cell expansion. Our study assessed the efficacy of Neo2/15-loaded Ag-scaffolds (Ag-Neo2/15 scaffolds) in expanding antigen-specific T cells from peripheral blood mononuclear cells (PBMCs) of healthy donors. We optimized Ag-scaffold configurations by varying the number of Neo2/15 molecules loaded on Ag-scaffolds and evaluated their impact on T-cell expansion and functionality. We showed that Ag-Neo2/15 scaffolds promoted significant T-cell expansion, with a comparable frequency of antigen-specific CD8 + T cells compared to IL-2/IL-21-loaded Ag-scaffolds (Ag-IL2/21 scaffolds). The CD8 + T cells expanded with Ag-Neo2/15 scaffolds exhibited potent TNFα and IFNγ production and expressed high levels of α4β7 integrin, a homing molecule which is important for directing T cells to specific tissues, potentially enhancing their therapeutic potential. T cells expanded with Ag-Neo2/15 scaffolds had superior and durable cytotoxicity against tumor target cells compared to T cells expanded with Ag-IL2/21 scaffolds. These findings were further supported by our single-cell analysis revealing that T cells expanded with Ag-Neo2/15 scaffolds had higher cytotoxic scores and lower dysfunctionality scores compared to T cells expanded with Ag-IL2/21 scaffolds. The single-cell analysis also indicated increased expression of genes linked to cell division and enhanced proliferative capacity in Ag-Neo2/15 expanded T cells. Furthermore, TCR clonality analysis demonstrated that Ag-Neo2/15 scaffolds promoted the expansion of functionally superior T-cell clones. The top clones of CD8 + T cells expanded with Ag-Neo2/15 scaffolds exhibited a favorable phenotype, essential for effective antigen recognition and sustained T-cell mediated cytotoxicity. Our findings suggest that Ag-Neo2/15 scaffolds represent an advancement in ACT by producing high-quality, functional antigen-specific T cells. This method has the potential to improve clinical outcomes in cancer therapy by generating large numbers of highly functional T cells, thereby optimizing the balance between cytotoxicity and proliferation capacity with less exhausted T-cells in expansion protocols.
-
Immunology and Microbiology
Developing CAR-T/NK cells that target EphA2 for non-small cell lung cancer treatment.
In Frontiers in Immunology on 4 April 2025 by Kim, S. M., Lee, S. Y., et al.
Chimeric antigen receptor (CAR) immunotherapy has revolutionized anticancer therapy, as it accurately targets cancer cells by recognizing specific antigens expressed in cancer cells. This innovative therapeutic strategy has attracted considerable attention. However, few therapeutics are available for treating non-small cell lung cancer (NSCLC), which accounts for most lung cancer cases and is one of the deadliest cancers with low survival rates.
In this study, we developed a new antibody targeting erythropoietin-producing hepatocellular carcinoma A2 (EphA2), which is highly expressed in NSCLC, and established CAR-T/ natural killer (NK) immune cells to verify its potential for immune cell therapy. The killing capacity, cytokine secretion and solid tumor growth inhibition of EphA2 CAR-T/NK cells were compared to normal T/NK cells.
EphA2 CAR-T cells demonstrated superior killing capacity, enhanced cytokine secretion, and significant solid tumor growth inhibition. Additionally, they exhibited improved tumor infiltration in lung cancer models compared to normal T cells. The anticancer efficacy of the developed EphA2 CAR-NK cells was also confirmed, showcasing their potential as robust candidates for immune cell therapy.
The findings of this study highlight the potential of CAR-T/NK cell therapy targeting EphA2 as an effective treatment for lung cancer, particularly NSCLC with high EphA2 expression. By leveraging the specific targeting capabilities of CAR-T cells and the unique properties of CAR-NK cells, this approach provides a promising therapeutic strategy to address the unmet needs in NSCLC treatment.
Copyright © 2025 Kim, Lee, Kim, Bae, Hong, Jo, Kim, Chung and Kim.
-
FC/FACS
-
Homo sapiens (Human)
-
Cancer Research
-
Immunology and Microbiology
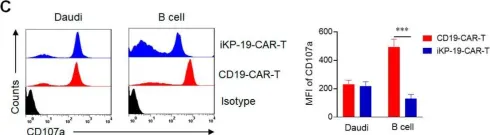
In Cancers (Basel) on 13 September 2020 by Tao, L., Farooq, M. A., et al.
Fig.4.C

-
FC/FACS
-
Collected and cropped from Cancers (Basel) by CiteAb, provided under a CC-BY license
Image 1 of 10
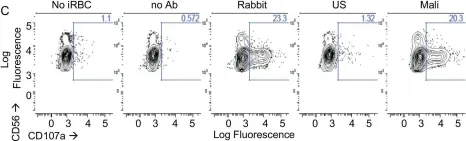
In Front Immunol on 12 November 2019 by Satta, A., Grazia, G., et al.
Fig.6.B

-
FC/FACS
-
Homo sapiens (Human)
Collected and cropped from Front Immunol by CiteAb, provided under a CC-BY license
Image 1 of 10
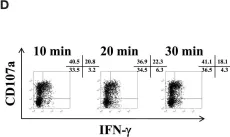
In Elife on 26 June 2018 by Arora, G., Hart, G. T., et al.
Fig.1.C

-
FC/FACS
-
Collected and cropped from Elife by CiteAb, provided under a CC-BY license
Image 1 of 10
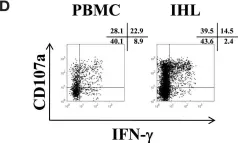
In Front Immunol on 14 March 2013 by Li, N., Puga Yung, G. L., et al.
Fig.3.D

-
FC/FACS
-
Collected and cropped from Front Immunol by CiteAb, provided under a CC-BY license
Image 1 of 10
In Front Immunol on 14 March 2013 by Li, N., Puga Yung, G. L., et al.
Fig.3.B

-
FC/FACS
-
Collected and cropped from Front Immunol by CiteAb, provided under a CC-BY license
Image 1 of 10
In Front Immunol on 14 March 2013 by Li, N., Puga Yung, G. L., et al.
Fig.6.C

-
FC/FACS
-
Collected and cropped from Front Immunol by CiteAb, provided under a CC-BY license
Image 1 of 10
In Front Immunol on 14 March 2013 by Li, N., Puga Yung, G. L., et al.
Fig.7.D

-
FC/FACS
-
Collected and cropped from Front Immunol by CiteAb, provided under a CC-BY license
Image 1 of 10
In Front Immunol on 14 March 2013 by Li, N., Puga Yung, G. L., et al.
Fig.7.C

-
FC/FACS
-
Collected and cropped from Front Immunol by CiteAb, provided under a CC-BY license
Image 1 of 10
In PLoS One on 24 April 2008 by Schlaepfer, E. & Speck, R. F.
Fig.4.A

-
FC/FACS
-
Homo sapiens (Human)
Collected and cropped from PLoS One by CiteAb, provided under a CC-BY license
Image 1 of 10
In PLoS One on 24 April 2008 by Schlaepfer, E. & Speck, R. F.
Fig.4.B

-
FC/FACS
-
Homo sapiens (Human)
Collected and cropped from PLoS One by CiteAb, provided under a CC-BY license
Image 1 of 10