Multiple sclerosis (MS) is a chronic autoimmune disease of the central nervous system (CNS). CD4+ CD25+hi Tregs, which normally suppress immune responses, exhibit impaired function in MS. Treg-derived extracellular vesicles (EVs) carry immunoregulatory proteins and miRNAs that modulate T-cell activity. However, EVs from MS patients show reduced suppressive capacity, suggesting their dysfunction contributes to MS pathogenesis. This highlights EVs' potential role in MS development and therapy.
Tregs were differentiated from naïve T cells isolated from peripheral blood mononuclear cells (PBMCs) of healthy donor, then transduced with B2M-shRNA lentivirus to generate HLA class I-knockdown Tregs. Extracellular vesicles-including natural vesicles, cytochalasin B-induced artificial vesicles, and ultrasound-induced artificial vesicles-were isolated from Tregs and characterized by scanning electron microscopy (SEM), nanoparticle tracking analysis (NTA), flow cytometry, and Western blot. Their effects on healthy donor and MS patient PBMCs were evaluated via flow cytometry and ELISA (IL-6, IL-10, IFN-γ).
Ultrasonication yielded a higher number оf vesicles enriched with key immunosuppressive proteins, including PD-1 and Tim-3, compared to cytochalasin B. Functional assays demonstrated the ability оf ultrasonication-induced AVs to suppress inflammatory markers, such as IFNγ, and modulate the cytokine profile in both healthy and MS-derived PBMCs.
Developing effective MS therapies remains challenging. While cellular therapies face limitations like Treg dysfunction and CNS delivery issues, allogeneic EVs offer a promising alternative due to their scalability, low immunogenicity, and blood-brain barrier penetration. We developed Treg-derived artificial vesicles (TrAVs) that maintain immunosuppressive properties and modulate PBMC responses, suggesting therapeutic potential for MS. Further research is needed to optimize production and validate efficacy in disease models.
© 2025 The Author(s).
Product Citations: 62
In BioImpacts : BI on 9 September 2025 by Ganeeva, I. A., Gilyazova, E. M., et al.
-
Homo sapiens (Human)
-
Immunology and Microbiology
In ACS Biomaterials Science Engineering on 11 August 2025 by Schluck, M., Weiden, J., et al.
Recently, biomaterials have emerged as tools to activate and expand T cells in the context of cancer immunotherapy. Most designs accommodate T cells with a stimulatory two-dimensional (2D) environment. In contrast, three-dimensional (3D) scaffolds, mimicking the complex architecture of the lymph node, have been shown to outperform 2D synthetic constructs, resulting in a more optimal T-cell expansion and phenotype. Here, we used injectable glycidyl methacrylated hyaluronic acid (HAGM)-based cryogel scaffolds to create a modular biodegradable 3D stimulatory immune niche. We developed a strategy to achieve highly specific and efficient covalent linking of immune-activating biomolecules, such as T-cell-activating peptide MHC complexes and antibodies, to HAGM scaffolds without compromising the injectable properties of the cryogels. Importantly, because our conjugation strategy is carried out postcryogelation, biomolecules are not exposed to free radicals and freeze-thawing cycles, facilitating highly reproducible covalent attachment. Our scaffold potently activates human- and murine-T cells, inducing higher levels of multifunctional T cells with a less exhausted phenotype compared to 2D cultures. Following injections, HAGM scaffolds retain up to 60% of highly proliferative T cells. In conclusion, our HAGM scaffolds are an easily adaptable tool for robust T-cell activation, thus further expanding the biomaterial-based immunotherapy toolbox.
-
Immunology and Microbiology
In Cell Reports Medicine on 20 May 2025 by Liu, X. F., Song, B., et al.
Adoptive cell therapy (ACT) using tumor-infiltrating lymphocytes (TILs) has demonstrated clinical success in solid tumors. We analyze 47 TIL infusion products and 62 pretreatment tumor microenvironments (TMEs) from a randomized phase 2 clinical study of concurrent chemoradiotherapy plus TIL-ACT (NCT02421640). Using single-cell and bulk RNA sequencing along with flow cytometry, we identify 14 CD3+ T cell clusters within 26 TIL infusion products: 11 CD3+CD8+ TILs, 2 CD3+CD4+ TILs, and 1 CD3+CD8-CD4- double-negative (DN) TIL. (DN) TILs, significantly associated with poor TIL-ACT outcomes, exhibit an activated regulatory T cell-like phenotype and include two CD56+ and four CD56- subsets. Among them, CD56-KZF2+ (DN) TILs are predominantly suppressive. (DN) TILs inhibit CD8+ TIL expansion via Fas-FasL, transforming growth factor β (TGF-β), and interleukin (IL)-10 signaling. Distinct CD8+ T subsets differentially impact on TIL-ACT outcomes, while 9 baseline TME gene signatures and 14 intracellular T cell genes hold prognostic value. Our findings identify predictive TIL subsets and biomarkers for TIL-ACT outcomes.
Copyright © 2025 The Author(s). Published by Elsevier Inc. All rights reserved.
-
FC/FACS
-
Homo sapiens (Human)
-
Cancer Research
-
Immunology and Microbiology
In Cancer Immunology Research on 4 March 2025 by Majocchi, S., Lloveras, P., et al.
Despite advances in cancer immunotherapy, such as targeting the PD-1/PD-L1 axis, a substantial number of patients harbor tumors that are resistant or relapse. Selective engagement of T-cell co-stimulatory molecules with bispecific antibodies may offer novel therapeutic options by enhancing signal 1-driven activation occurring via T-cell receptor engagement. In this study, we report the development and preclinical characterization of NI-3201, a PD-L1×CD28 bispecific antibody generated on the κλ-body platform that was designed to promote T-cell activity and antitumor function through a dual mechanism of action. We confirmed that NI-3201 blocks the PD-L1/PD-1 immune checkpoint pathway and conditionally provides T-cell co-stimulation via CD28 (signal 2) when engaging PD-L1+ tumors or immune cells. In systems with signal 1-primed T cells, NI-3201 enhanced potent effector functionality: in vitro through antigen-specific recall assays with cytomegalovirus-specific T cells and in vivo by inducing tumor regression and immunologic memory in tumor-associated antigen-expressing MC38 syngeneic mouse models. When T-cell engagers were used to provide synthetic signal 1, the combination with NI-3201 resulted in synergistic T cell-dependent cytotoxicity and potent antitumor activity in two humanized mouse tumor models. Nonhuman primate safety assessments showed favorable tolerability and pharmacokinetics at pharmacologically active doses. Quantitative systems pharmacology modeling predicted that NI-3201 exposure results in antitumor activity in patients, but this remains to be investigated. Overall, this study suggests that by combining PD-L1 blockade with safe and effective CD28 co-stimulation, NI-3201 has the potential to improve cancer immunotherapy outcomes, and the clinical development of NI-3201 for PD-L1+ solid tumors is planned.
©2025 The Authors; Published by the American Association for Cancer Research.
-
FC/FACS
-
Cancer Research
-
Immunology and Microbiology
Transferrin Disassociates TCR from CD3 Signaling Apparatus to Promote Metastasis.
In Research (Washington, D.C.) on 15 January 2025 by Cheng, R., Tang, X., et al.
Immune recognition and activation by the peptide-laden major histocompatibility complex-T cell receptor (TCR)-CD3 complex is essential for anti-tumor immunity. Tumors may escape immune surveillance by dissembling the complex. Here, we report that transferrin, which is overexpressed in patients with liver metastasis, disassociates TCR from the CD3 signaling apparatus by targeting the constant domain (CD) of T cell receptor α (TCRα), consequently suppresses T cell activation, and inhibits anti-metastatic and anti-tumor immunity. In mouse models of melanoma and lymphoma, transferrin overexpression exacerbates liver metastasis, while its knockdown, antibody, designed peptides, and CD mutation interfering with transferrin-TCRα interaction inhibit metastasis. This work reveals a novel strategy of tumor evasion of immune surveillance by blocking the coupling between TCRs and the CD3 signaling apparatus to suppress TCR activation. Given the conservation of CD and transferrin up-regulation in metastatic tumors, the strategy might be a common metastatic mechanism. Targeting transferrin-TCRα holds promise for anti-metastatic treatment.
Copyright © 2025 Ruomei Cheng et al.
-
FC/FACS
-
Homo sapiens (Human)
-
Cancer Research
In Cell Mol Immunol on 1 July 2023 by Hao, Y., Miraghazadeh, B., et al.
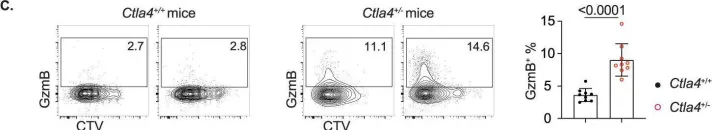
Fig.6.C
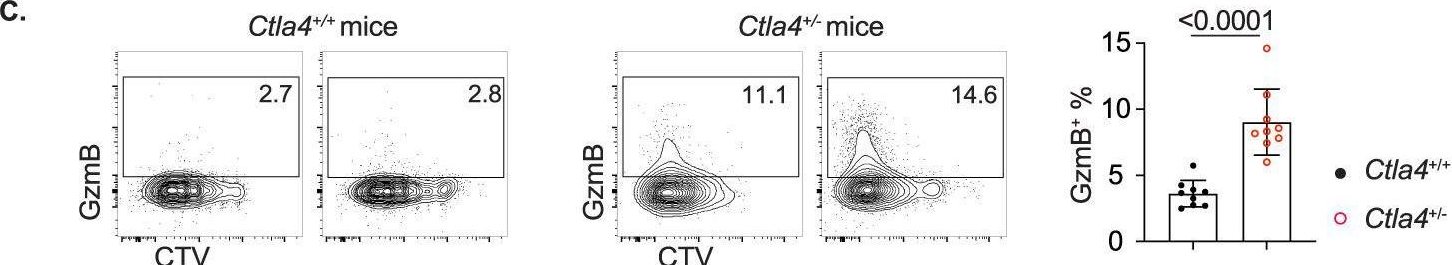
-
FC/FACS
-
Homo sapiens (Human)
Collected and cropped from Cellular Molecular Immunology by CiteAb, provided under a CC-BY license
Image 1 of 4
In Cell Mol Immunol on 1 July 2023 by Hao, Y., Miraghazadeh, B., et al.
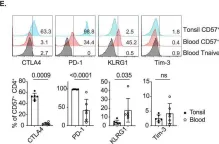
Fig.4.E
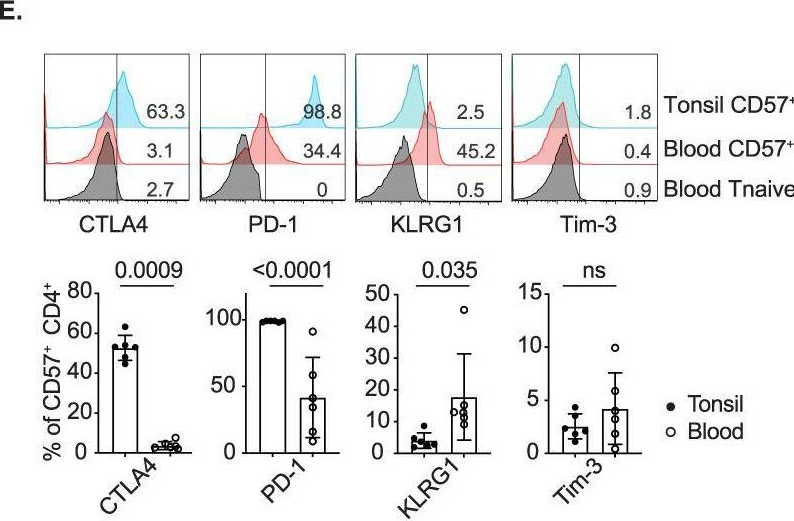
-
FC/FACS
-
Collected and cropped from Cellular Molecular Immunology by CiteAb, provided under a CC-BY license
Image 1 of 4
In Int J Mol Sci on 13 May 2022 by de Campos, N. S. P., Beserra, A. O., et al.
Fig.2.D
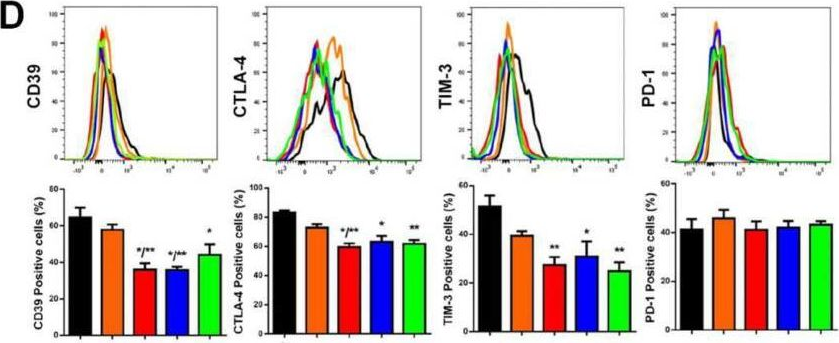
-
FC/FACS
-
Collected and cropped from International Journal of Molecular Sciences by CiteAb, provided under a CC-BY license
Image 1 of 4
In PLoS Biol on 1 April 2014 by Sarrabayrouse, G., Bossard, C., et al.
Fig.3.A
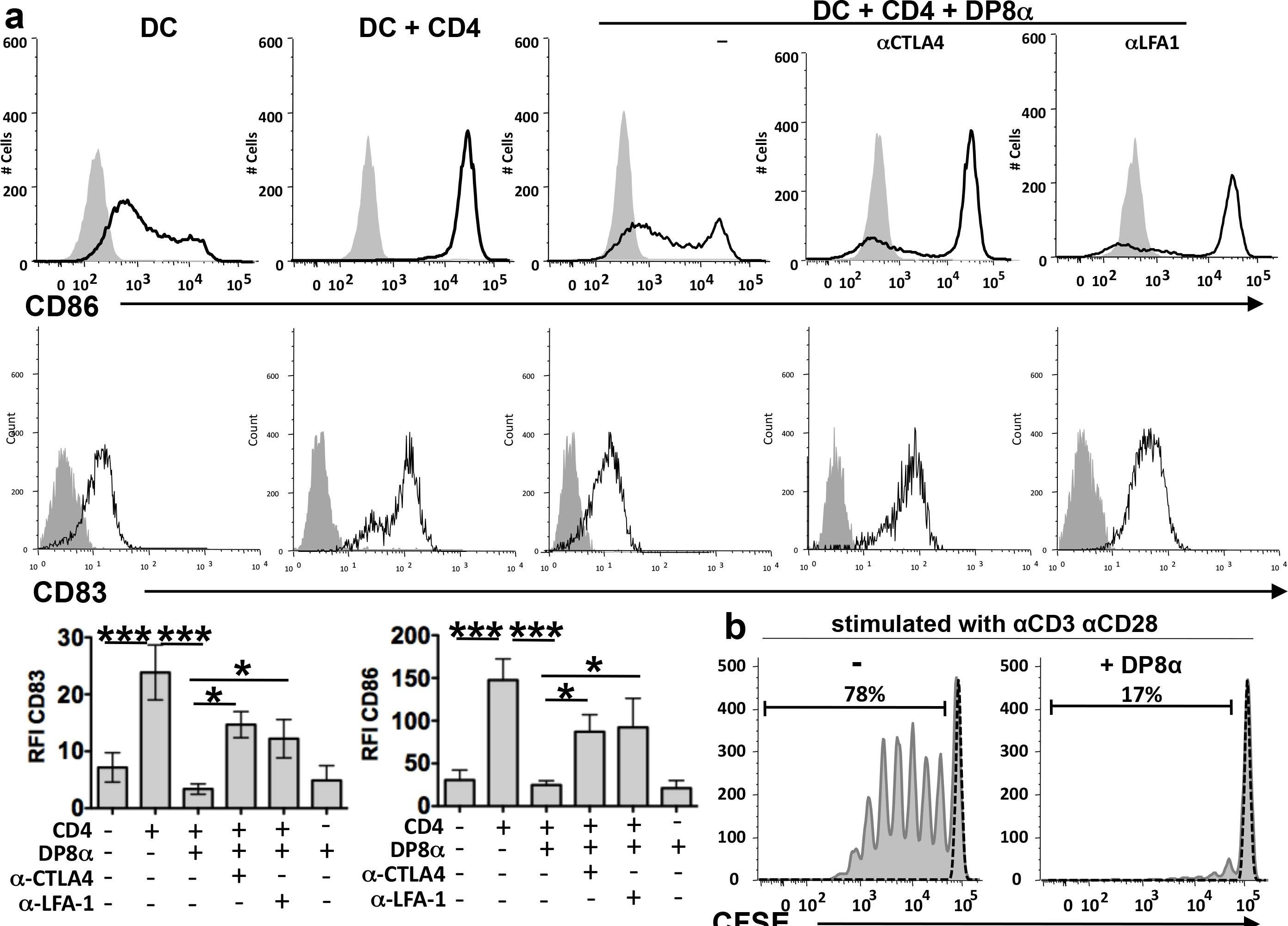
-
FC/FACS
-
Homo sapiens (Human)
Collected and cropped from PLoS Biology by CiteAb, provided under a CC-BY license
Image 1 of 4