Dnah5 is associated with primary ciliary dyskinesia in humans. Dnah5-knockout (Dnah5-/- mice develop acute hydrocephalus shortly after birth owing to impaired ciliary motility and cerebrospinal fluid (CSF) stagnation. In contrast to chronic adult-onset hydrocephalus observed in other models, this rapid ventricular enlargement indicates additional factors beyond CSF stagnation. Herein, we investigated the contributors to rapid ventricular enlargement in congenital hydrocephalus. Dnah5-/- mice were generated using CRISPR/Cas9. The expression of dynein, N-cadherin, and nestin in the cerebral cortex was assessed using microarrays and immunostaining. Real-time PCR and Western blotting were performed for gene and protein quantification, respectively. All Dnah5-/- mice developed hydrocephalus, confirmed by electron microscopy, indicating the absence of axonemal outer dynein arms. Ventricular enlargement occurred rapidly, with a 25% reduction in the number of mature neurons in the motor cortex. Dync1h1 expression was decreased, while cytoplasmic dynein levels were 56.3% lower. Levels of nestin and N-cadherin in the lateral ventricular walls decreased by 31.7% and 33.3%, respectively. Reduced cytoplasmic dynein disrupts neurogenesis and axonal growth and reduces neuron cortical density. Hydrocephalus in Dnah5-/- mice may result from cortical maldevelopment due to cytoplasmic dynein deficiency, further exacerbating ventricular enlargement due to CSF stagnation caused by impaired motile ciliary function.
Product Citations: 166
In Cells on 14 November 2024 by Sakamoto, K., Miyajima, M., et al.
-
Cell Biology
Noggin-Loaded PLA/PCL Patch Inhibits BMP-Initiated Reactive Astrogliosis.
In International Journal of Molecular Sciences on 29 October 2024 by Hawes, J., González-Manteiga, A., et al.
Myelomeningocele (MMC) is a congenital birth defect of the spine and spinal cord, commonly treated clinically through prenatal or postnatal surgery by repairing the unclosed spinal canal. Having previously developed a PLA/PCL polymer smart patch for this condition, we aim to further expand the potential therapeutic options by providing additional cellular and biochemical support in addition to its mechanical properties. Bone morphogenetic proteins (BMPs) are a large class of secreted factors that serve as modulators of development in multiple organ systems, including the CNS. We hypothesize that our smart patch mitigates the astrogenesis induced, at least partly, by increased BMP activity during MMC. To test this hypothesis, neural stem or precursor cells were isolated from rat fetuses and cultured in the presence of Noggin, an endogenous antagonist of BMP action, with recombinant BMPs. We found that the developed PLA/PCL patch not only serves as a biocompatible material for developing neural stem cells but was also able to act as a carrier for BMP-Notch pathway inhibitor Noggin, effectively minimizing the effect of BMP2 or BMP4 on NPCs cultured with the Noggin-loaded patch.
-
Rattus norvegicus (Rat)
In Science Advances on 9 August 2024 by Norton, E. S., Whaley, L. A., et al.
Glioblastoma (GBM) is the most prevalent and aggressive malignant primary brain tumor. GBM proximal to the lateral ventricles (LVs) is more aggressive, potentially because of subventricular zone contact. Despite this, cross-talk between GBM and neural stem/progenitor cells (NSC/NPCs) is not well understood. Using cell-specific proteomics, we show that LV-proximal GBM prevents neuronal maturation of NSCs through induction of senescence. In addition, GBM brain tumor-initiating cells (BTICs) increase expression of cathepsin B (CTSB) upon interaction with NPCs. Lentiviral knockdown and recombinant protein experiments reveal that both cell-intrinsic and soluble CTSB promote malignancy-associated phenotypes in BTICs. Soluble CTSB stalls neuronal maturation in NPCs while promoting senescence, providing a link between LV-tumor proximity and neurogenesis disruption. Last, we show LV-proximal CTSB up-regulation in patients, showing the relevance of this cross-talk in human GBM biology. These results demonstrate the value of proteomic analysis in tumor microenvironment research and provide direction for new therapeutic strategies in GBM.
Restoration of neuronal progenitors by partial reprogramming in the aged neurogenic niche.
In Nature Aging on 1 April 2024 by Xu, L., Ramirez-Matias, J., et al.
Partial reprogramming (pulsed expression of reprogramming transcription factors) improves the function of several tissues in old mice. However, it remains largely unknown how partial reprogramming impacts the old brain. Here we use single-cell transcriptomics to systematically examine how partial reprogramming influences the subventricular zone neurogenic niche in aged mouse brains. Whole-body partial reprogramming mainly improves neuroblasts (cells committed to give rise to new neurons) in the old neurogenic niche, restoring neuroblast proportion to more youthful levels. Interestingly, targeting partial reprogramming specifically to the neurogenic niche also boosts the proportion of neuroblasts and their precursors (neural stem cells) in old mice and improves several molecular signatures of aging, suggesting that the beneficial effects of reprogramming are niche intrinsic. In old neural stem cell cultures, partial reprogramming cell autonomously restores the proportion of neuroblasts during differentiation and blunts some age-related transcriptomic changes. Importantly, partial reprogramming improves the production of new neurons in vitro and in old brains. Our work suggests that partial reprogramming could be used to rejuvenate the neurogenic niche and counter brain decline in old individuals.
© 2024. The Author(s), under exclusive licence to Springer Nature America, Inc.
Autophagy supports PDGFRA-dependent brain tumor development by enhancing oncogenic signaling.
In Developmental Cell on 22 January 2024 by Simpson, J. E., Muir, M. T., et al.
Autophagy is a conserved cellular degradation process. While autophagy-related proteins were shown to influence the signaling and trafficking of some receptor tyrosine kinases, the relevance of this during cancer development is unclear. Here, we identify a role for autophagy in regulating platelet-derived growth factor receptor alpha (PDGFRA) signaling and levels. We find that PDGFRA can be targeted for autophagic degradation through the activity of the autophagy cargo receptor p62. As a result, short-term autophagy inhibition leads to elevated levels of PDGFRA but an unexpected defect in PDGFA-mediated signaling due to perturbed receptor trafficking. Defective PDGFRA signaling led to its reduced levels during prolonged autophagy inhibition, suggesting a mechanism of adaptation. Importantly, PDGFA-driven gliomagenesis in mice was disrupted when autophagy was inhibited in a manner dependent on Pten status, thus highlighting a genotype-specific role for autophagy during tumorigenesis. In summary, our data provide a mechanism by which cells require autophagy to drive tumor formation.
Copyright © 2023 The Author(s). Published by Elsevier Inc. All rights reserved.
-
Cancer Research
-
Cell Biology
-
Stem Cells and Developmental Biology
In Front Cell Neurosci on 11 June 2022 by Zhang, Y. F., Li, X. X., et al.
Fig.2.D

-
WB
-
Collected and cropped from Front Cell Neurosci by CiteAb, provided under a CC-BY license
Image 1 of 6
In Front Cell Neurosci on 11 June 2022 by Zhang, Y. F., Li, X. X., et al.
Fig.5.E

-
WB
-
Collected and cropped from Front Cell Neurosci by CiteAb, provided under a CC-BY license
Image 1 of 6
In Front Cell Neurosci on 11 June 2022 by Zhang, Y. F., Li, X. X., et al.
Fig.6.E

-
WB
-
Collected and cropped from Front Cell Neurosci by CiteAb, provided under a CC-BY license
Image 1 of 6
In Cells on 9 April 2022 by Huang, X., Guo, H., et al.
Fig.1.J

-
WB
-
Collected and cropped from Cells by CiteAb, provided under a CC-BY license
Image 1 of 6
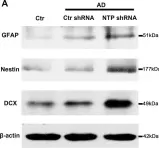
In Front Aging Neurosci on 9 February 2021 by Li, P., Quan, W., et al.
Fig.4.A

-
WB
-
Rattus norvegicus (Rat)
Collected and cropped from Front Aging Neurosci by CiteAb, provided under a CC-BY license
Image 1 of 6
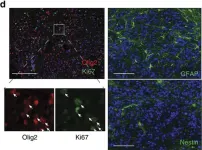
In Nat Commun on 11 July 2017 by Cook, P. J., Thomas, R., et al.
Fig.2.D

-
ICC-IF
-
Mus musculus (House mouse)
Collected and cropped from Nat Commun by CiteAb, provided under a CC-BY license
Image 1 of 6