Peptidylarginine deiminase 4 (PAD4) is an enzyme that modifies proteins by converting positively charged arginine residues to neutral citrulline residues. This process, termed citrullination, has been known to trigger NETosis, a neutrophil cell death pathway involving the release of neutrophil extracellular traps (NETs). Abnormal PAD4 activity and protein citrullination have been linked to various diseases, including those affecting the central nervous system. Herein we investigated the profile of PAD4 expression in an animal model of stroke induced by middle cerebral artery occlusion (MCAO). PAD4 levels were significantly elevated in the ischemic core and penumbra of the affected hemisphere at 3-6 and 6-48 h post-MCAO, respectively. Notably, NETosis induction, indicated by the upregulation of CitH3 (citrullinated histone H3, a NETosis marker), was observed between 48 and 96 h post-MCAO, peaking at 96 h. While PAD4 was present in most brain cell types of sham controls, strong PAD4 induction was primarily observed in neurons during the peak PAD4 induction period (12-24 h post-MCAO). Importantly, intranasal administration of the PAD4 inhibitor BB-Cl-amidine (BBCA) significantly reduced infarct volume and improved neurological and functional outcomes at 24 h post-MCAO, demonstrating a strong protective effect of PAD4 inhibition in ischemic stroke. Staining with an antibody that recognizing citrullinated proteins (F95) revealed an accumulation of these proteins, especially degenerating neurons, however, BBCA treatment significantly suppressed this accumulation in dying neurons. These findings indicate that PAD4-mediated protein citrullination in neurons plays a critical role in promoting ischemic brain damage. Furthermore, delayed administration of BBCA (at 48/72 h post-MCAO) suppresses the NETosis induction observed at 96 h post-MCAO, potentially ameliorating repair processes such as blood vessel regeneration. Collectively, these findings suggest a complex role of PAD4 in cerebral ischemia, with neuroprotective effects (NETosis-independent function) during the acute to subacute period and NETosis-suppressive effects at later time points.
© 2025. The Author(s).
Product Citations: 39
In Acta Neuropathologica Communications on 18 February 2025 by Seol, S. I., Oh, S. A., et al.
-
IHC-IF
In International Journal of Molecular Sciences on 26 January 2024 by Aguado-Garrido, M., García-Rama, C., et al.
The treatment of spinal cord injury (SCI) with uncultivated human bone marrow-derived stromal cells (bmSCs) prepared by negative selection has been proposed to be therapeutically superior to treatment with stem cells that were expanded in vitro. To explore their use in clinical trials, we studied the functional effects of delayed application at 7 days after SCI by testing different doses of bmSCs. Spinal cord contusion injury was induced in adult male Wistar rats at the thoracic level T9. Human bmSCs were prepared by negative selection without expansion in vitro (NeuroCellsTM). Treatment consisted of one 150 µL injection into the cisterna magna containing 0.5 or 2.5 million fresh bmSCs or 2.5 million bmSCs. The recovery of motor functions was evaluated during a surveillance period of six weeks (6 W), during which spinal cords were assessed histologically. Treatment resulted in a significant, dose-dependent therapeutic effect on the recovery of motor performance. The histological analysis revealed a lower degree of axonal degeneration and better survival of neurons and oligodendrocytes in bmSCs treated rats. Our results support delayed intrathecal application of bmSCs prepared by negative selection without expansion in vitro as a treatment of SCI.
-
Neuroscience
In British Journal of Pharmacology on 1 August 2023 by Manni, L., Leotta, E., et al.
Traumatic brain injury (TBI) comprises a primary injury directly induced by impact, which progresses into a secondary injury leading to neuroinflammation, reactive astrogliosis, and cognitive and motor damage. To date, treatment of TBI consists solely of palliative therapies that do not prevent and/or limit the outcomes of secondary damage and only stabilize the deficits. The neurotrophin, nerve growth factor (NGF), delivered to the brain parenchyma following intranasal application, could be a useful means of limiting or improving the outcomes of the secondary injury, as suggested by pre-clinical and clinical data.
We evaluated the effect of acute intranasal treatment of young (20-postnatal day) rats, with NGF in a TBI model (weight drop/close head), aggravated by hypoxic complications. Immediately after the trauma, rats were intranasally treated with human recombinant NGF (50 μg·kg-1 ), and motor behavioural test, morphometric and biochemical assays were carried out 24 h later.
Acute intranasal NGF prevented the onset of TBI-induced motor disabilities, and decreased reactive astrogliosis, microglial activation and IL-1β content, which after TBI develops to the same extent in the impact zone and the hypothalamus.
Intranasal application of NGF was effective in decreasing the motor dysfunction and neuroinflammation in the brain of young rats in our model of TBI. This work forms an initial pre-clinical evaluation of the potential of early intranasal NGF treatment in preventing and limiting the disabling outcomes of TBI, a clinical condition that remains one of the unsolved problems of paediatric neurology.
© 2023 The Authors. British Journal of Pharmacology published by John Wiley & Sons Ltd on behalf of British Pharmacological Society.
-
Neuroscience
-
Pharmacology
In International Journal of Molecular Sciences on 5 July 2023 by Alves-Sampaio, A., Del-Cerro, P., et al.
Biomaterials may enhance neural repair after spinal cord injury (SCI) and testing their functionality in large animals is essential to achieve successful clinical translation. This work developed a porcine contusion/compression SCI model to investigate the consequences of myelotomy and implantation of fibrin gel containing biofunctionalized carbon microfibers (MFs). Fourteen pigs were distributed in SCI, SCI/myelotomy, and SCI/myelotomy/implant groups. An automated device was used for SCI. A dorsal myelotomy was performed on the lesion site at 1 day post-injury for removing cloths and devitalized tissue. Bundles of MFs coated with a conducting polymer and cell adhesion molecules were embedded in fibrin gel and used to bridge the spinal cord cavity. Reproducible lesions of about 1 cm in length were obtained. Myelotomy and lesion debridement caused no further neural damage compared to SCI alone but had little positive effect on neural regrowth. The MFs/fibrin gel implant facilitated axonal sprouting, elongation, and alignment within the lesion. However, the implant also increased lesion volume and was ineffective in preventing fibrosis, thus precluding functional neural regeneration. Our results indicate that myelotomy and lesion debridement can be advantageously used for implanting MF-based scaffolds. However, the implants need refinement and pharmaceuticals will be necessary to limit scarring.
-
IHC
-
Biochemistry and Molecular biology
-
Neuroscience
-
Veterinary Research
Autophagy-Mediated Inflammatory Cytokine Secretion in Sporadic ALS Patient iPSC-Derived Astrocytes.
In Oxidative Medicine and Cellular Longevity on 2 September 2022 by BaofengFeng, Amponsah, A. E., et al.
Astrocytes can be involved in motor neuron toxicity in amyotrophic lateral sclerosis (ALS) induced by noncell autonomous effects, and inflammatory cytokines may play the main role in mediating this process. However, the etiology of aberrant cytokine secretion is unclear. The present study assessed possible involvement of the mTOR-autophagy pathway in aberrant cytokine secretion by ALS patient iPSC-derived astrocytes. Method and Results. PBMCs from sporadic ALS patients and control subjects were reprogrammed into iPSCs, which were then differentiated into astrocytes and/or motor neurons. Comparison with control astrocytes indicated that conditioned medium of ALS astrocytes reduced the viability of the control motor neurons (p < 0.05) assessed using the MTT assay. The results of ELISA showed that the concentrations of TNFα, IL1β, and IL6 in cell culture medium of ALS astrocytes were increased (p < 0.05). ALS astrocytes had higher p62 and mTOR levels and lower LC3BII/LC3BI ratio and ULK1 and p-Beclin-1 (Ser15) levels (p < 0.05), indicating defective autophagy. Exogenous inhibition of the mTOR-autophagy pathway, but not the activation of the pathway in control subject astrocytes, increased the levels of p62 and mTOR and concentration of IL-1β, TNF-α, and IL-6 in cell culture medium and decreased the LC3BII/LC3BI ratio and levels of ULK1 and p-Beclin-1 (Ser15), and these changes were comparable to those in ALS astrocytes. After 48 h of rapamycin (autophagy activator) and 3-methyladenine (autophagy inhibitor) treatments, the exogenous activation of the mTOR-autophagy pathway, but not inhibition of the pathway, in ALS astrocytes significantly reduced the concentrations of TNFα, IL1β, and IL6 in cell culture medium and reduced the levels of p62, while increasing the levels of LC3B-II/LC3B-I, ULK1, and p-Beclin-1 (Ser15), and these changes were comparable to those in control subject astrocytes.
Alteration in the mTOR/ULK1/Beclin-1 pathway regulated cytokine secretion in ALS astrocytes, which was able to lead to noncell autonomous toxicity. Autophagy activation mitigated cytokine secretion by ALS astrocytes.
Copyright © 2022 BaofengFeng et al.
-
Cell Biology
-
Immunology and Microbiology
-
Neuroscience
-
Stem Cells and Developmental Biology
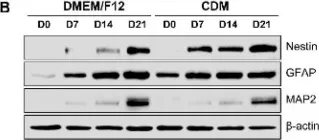
In Int J Mol Sci on 24 June 2022 by Park, S. Y., Kim, D. S., et al.
Fig.2.B

-
WB
-
Collected and cropped from Int J Mol Sci by CiteAb, provided under a CC-BY license
Image 1 of 1