Cutaneous squamous cell carcinoma (cSCC) is a highly prevalent skin cancer. While surgery remains the gold standard treatment, non-invasive methods like photodynamic therapy (PDT) stand out for their high efficacy and minimal cosmetic impact. However, resistance to PDT is still a challenge. Numerous cellular processes involved in cancer biology and therapy resistance are regulated by the TGFβ1/SMAD pathway. Using in vitro bidimensional and tridimensional cultures of cSCC cell lines, we studied the development of resistance to PDT in response to TGFβ1 secreted by cancer associated fibroblasts. Our results highlight the TGFβ1 co-receptor endoglin as a key molecular player in the process. Importantly, targeting endoglin expression with N-acetylcysteine (NAC) or raloxifene significantly reduced TGFβ1 levels and effectively prevented resistance. In addition, the combination of PDT with NAC resulted in an improved therapeutic outcome in vivo in SKH-1 mice with cSCC photogenerated by chronic exposition to ultraviolet light. In conclusion, the combination of PDT with NAC or raloxifene enhances PDT efficacy by mitigating resistance mechanisms, which can open new avenues for the treatment of cSCC.
© The author(s).
Product Citations: 197
In International Journal of Biological Sciences on 19 May 2025 by Gallego-Rentero, M., Botella, L. M., et al.
-
Cancer Research
In Frontiers in Pharmacology on 3 April 2025 by Nizami, Z. N., Al Azzani, M., et al.
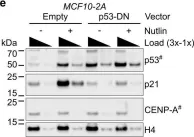
Colorectal cancer is a leading cause of cancer related-death worldwide, and resistance to 5-fluorouracil (5FU, a key component of chemotherapy regimens, is a major clinical concern. We have previously elucidated the effects of Rhus coriaria ethanolic extract (RCE) in triple-negative breast cancer, CRC, and pancreatic cancer cells. Here, we explored the anticancer effects of RCE in parental (HCT-116-WT) and 5FU-resistant HCT-116 (HCT-116-5FU-R) CRC cells.
MTT assay was used to assess cell viability. Muse analyzer was used to assess cell viability, cell cycle distribution, and apoptosis. Additionally, colony formation and growth assays and western blots were performed. In vivo effects of RCE were assessed by an in ovo chick embryo tumor growth assay.
We found that RCE inhibited the viability and colony formation and growth capacities of HCT-116-WT and HCT-116-5FU-R cells. The antiproliferative effects were attributed to DNA damage-mediated impairment of cell cycle at S phase, and induction of Beclin-1-independent autophagy in both cell lines. Mechanistically, inhibition of the mTOR, STAT3 and p38 MAPK pathways was implicated in the latter. Additionally, RCE induced caspase-7-independent apoptosis in HCT-116-WT cells. However, HCT-116-5FU-R cells were resistant to apoptosis through upregulation of survivin, and downregulation of Bax. Using autophagy and proteasome inhibitors, we clarified that autophagy and the proteasome pathway contributed to RCE-mediated cell death in HCT-116-WT and HCT-116-5FU-R cells. Lastly, we confirmed RCE inhibited the growth of both HCT-116-WT and HCT-116-5FU-R xenografts in a chick embryo model.
Collectively, our findings highlight that RCE is a source of phytochemicals that can be used as anticancer agents for 5FU-resistant CRC.
Copyright © 2025 Nizami, Al Azzani, Khaldi, Wali, Magramane, Samad, Eid, Arafat, Al Dhaheri, Attoub and Iratni.
-
Cancer Research
-
Pharmacology
In ASN NEURO on 18 July 2024 by Guardiola-Diaz, H. M., DiBenedictis, B. T., et al.
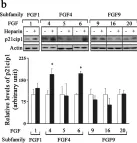
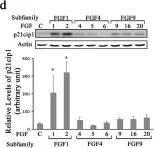
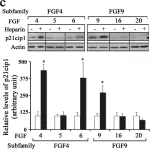
The fifteen canonical paracrine fibroblast growth factors (FGFs) are organized in five subfamilies that interact with four FGF-receptors (FGFRs) and heparan sulfate proteoglycan (HSPG) co-receptors. Many of these FGFs are expressed in CNS regions where oligodendrocyte (OL) progenitors originate, migrate or differentiate. FGF2 (basic FGF) is considered a prototype FGF and the information about the effects of FGF signaling on OL-lineage cells has evolved largely from the study of FGF2. However, other FGFs from four subfamilies ((FGF1 (FGF1,-2), FGF4 (FGF4,-5,-6), FGF8 (FGF8,-17,-18) and FGF9 (FGF9,-16,-20)) that can interact with the isoforms of FGFRs expressed in OL-lineage cells may also play important roles. We previously reported OL-responses to FGF8 family members. Here, we investigate the effects of members of the FGF1,-4, and -9 subfamilies on proliferation and differentiation of OL progenitors (OPCs), and on cell cycle re-entry and down-regulation of myelin proteins by mature OLs. We found that while FGF2 induced all these responses strongly, FGF4,-6,-9 could do so only transiently and in the presence of exogenous HSPGs, and that FGF5,-16,-20 could not do so even in the presence of heparin or at higher concentrations. Furthermore, we noted that structurally similar FGFs within subfamilies did not always show similarities in their biological effects on OL-lineage cells. Taken together, these studies reveal that FGFs differ in the way they regulate the OL-lineage cells, emphasizes the selectivity and importance of HSPGs as FGF co-receptors in OL-lineage cells and suggests that structural similarity among FGF-subfamily members may not always predict their overlapping biological functions.
-
WB
-
Neuroscience
PRMT5-mediated arginine methylation of FXR1 is essential for RNA binding in cancer cells.
In Nucleic Acids Research on 8 July 2024 by Vijayakumar, A., Majumder, M., et al.
Emerging evidence indicates that arginine methylation promotes the stability of arginine-glycine-rich (RGG) motif-containing RNA-binding proteins (RBPs) and regulates gene expression. Here, we report that post-translational modification of FXR1 enhances the binding with mRNAs and is involved in cancer cell growth and proliferation. Independent point mutations in arginine residues of FXR1's nuclear export signal (R386 and R388) and RGG (R453, R455 and R459) domains prevent it from binding to RNAs that form G-quadruplex (G4) RNA structures. Disruption of G4-RNA structures by lithium chloride failed to bind with FXR1, indicating its preference for G4-RNA structure containing mRNAs. Furthermore, loss-of-function of PRMT5 inhibited FXR1 methylation both in vivo and in vitro, affecting FXR1 protein stability, inhibiting RNA-binding activity and cancer cell growth and proliferation. Finally, the enhanced crosslinking and immunoprecipitation (eCLIP) analyses reveal that FXR1 binds with the G4-enriched mRNA targets such as AHNAK, MAP1B, AHNAK2, HUWE1, DYNC1H1 and UBR4 and controls its mRNA expression in cancer cells. Our findings suggest that PRMT5-mediated FXR1 methylation is required for RNA/G4-RNA binding, which promotes gene expression in cancer cells. Thus, FXR1's structural characteristics and affinity for RNAs preferentially G4 regions provide new insights into the molecular mechanism of FXR1 in oral cancer cells.
© The Author(s) 2024. Published by Oxford University Press on behalf of Nucleic Acids Research.
-
Biochemistry and Molecular biology
-
Cancer Research
-
Genetics
Epistatic interactions between NMD and TRP53 control progenitor cell maintenance and brain size.
In Neuron on 3 July 2024 by Lin, L., Zhao, J., et al.
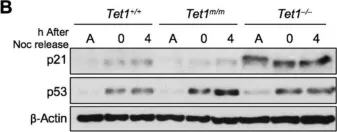
Mutations in human nonsense-mediated mRNA decay (NMD) factors are enriched in neurodevelopmental disorders. We show that deletion of key NMD factor Upf2 in mouse embryonic neural progenitor cells causes perinatal microcephaly but deletion in immature neurons does not, indicating NMD's critical roles in progenitors. Upf2 knockout (KO) prolongs the cell cycle of radial glia progenitor cells, promotes their transition into intermediate progenitors, and leads to reduced upper-layer neurons. CRISPRi screening identified Trp53 knockdown rescuing Upf2KO progenitors without globally reversing NMD inhibition, implying marginal contributions of most NMD targets to the cell cycle defect. Integrated functional genomics shows that NMD degrades selective TRP53 downstream targets, including Cdkn1a, which, without NMD suppression, slow the cell cycle. Trp53KO restores the progenitor cell pool and rescues the microcephaly of Upf2KO mice. Therefore, one physiological role of NMD in the developing brain is to degrade selective TRP53 targets to control progenitor cell cycle and brain size.
Copyright © 2024 Elsevier Inc. All rights reserved.
-
Neuroscience
In ASN Neuro on 18 July 2024 by Guardiola-Diaz, H. M., DiBenedictis, B. T., et al.
Fig.3.B

-
WB
-
Collected and cropped from ASN Neuro by CiteAb, provided under a CC-BY license
Image 1 of 88
In ASN Neuro on 18 July 2024 by Guardiola-Diaz, H. M., DiBenedictis, B. T., et al.
Fig.5.D

-
WB
-
Collected and cropped from ASN Neuro by CiteAb, provided under a CC-BY license
Image 1 of 88
In ASN Neuro on 18 July 2024 by Guardiola-Diaz, H. M., DiBenedictis, B. T., et al.
Fig.6.C

-
WB
-
Collected and cropped from ASN Neuro by CiteAb, provided under a CC-BY license
Image 1 of 88
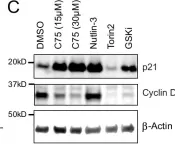
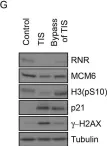
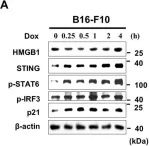
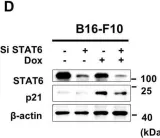
In Nat Commun on 18 March 2024 by Köberlin, M. S., Fan, Y., et al.
Fig.5.C

-
WB
-
Collected and cropped from Nat Commun by CiteAb, provided under a CC-BY license
Image 1 of 88
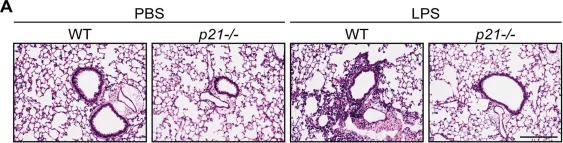
In Aging (Albany NY) on 30 March 2023 by Levi, N., Papismadov, N., et al.
Fig.2.A

-
IHC
-
Mus musculus (House mouse)
Collected and cropped from Aging (Albany NY) by CiteAb, provided under a CC-BY license
Image 1 of 88
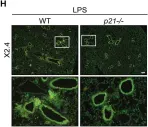
In Aging (Albany NY) on 30 March 2023 by Levi, N., Papismadov, N., et al.
Fig.3.H

-
IHC-IF
-
Mus musculus (House mouse)
Collected and cropped from Aging (Albany NY) by CiteAb, provided under a CC-BY license
Image 1 of 88
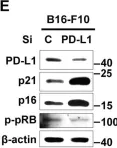
In Cell Death Dis on 15 September 2022 by Lee, J. J., Kim, S. Y., et al.
Fig.1.E

-
WB
-
Collected and cropped from Cell Death Dis by CiteAb, provided under a CC-BY license
Image 1 of 88
In Cell Death Dis on 15 September 2022 by Lee, J. J., Kim, S. Y., et al.
Fig.2.K

-
WB
-
Collected and cropped from Cell Death Dis by CiteAb, provided under a CC-BY license
Image 1 of 88
In Cell Death Dis on 15 September 2022 by Lee, J. J., Kim, S. Y., et al.
Fig.2.F

-
WB
-
Collected and cropped from Cell Death Dis by CiteAb, provided under a CC-BY license
Image 1 of 88
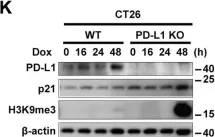
In Cell Death Dis on 15 September 2022 by Lee, J. J., Kim, S. Y., et al.
Fig.3.A

-
WB
-
Collected and cropped from Cell Death Dis by CiteAb, provided under a CC-BY license
Image 1 of 88
In EMBO J on 18 July 2022 by Tátrai, P. & Gergely, F.
Fig.7.A

-
WB
-
Collected and cropped from EMBO J by CiteAb, provided under a CC-BY license
Image 1 of 88
In EMBO J on 18 July 2022 by Tátrai, P. & Gergely, F.
Fig.6.A

-
WB
-
Collected and cropped from EMBO J by CiteAb, provided under a CC-BY license
Image 1 of 88
In Cells on 17 April 2022 by Chrysanthou, S., Flores, J. C., et al.
Fig.2.B

-
WB
-
Collected and cropped from Cells by CiteAb, provided under a CC-BY license
Image 1 of 88
In Biol Open on 1 December 2021 by Del Toro, N., Lessard, F., et al.
Fig.7.G

-
WB
-
Collected and cropped from Biol Open by CiteAb, provided under a CC-BY license
Image 1 of 88
In Commun Biol on 26 March 2021 by Jeffery, D., Gatto, A., et al.
Fig.3.E

-
WB
-
Collected and cropped from Commun Biol by CiteAb, provided under a CC-BY license
Image 1 of 88
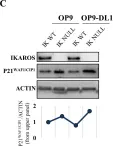
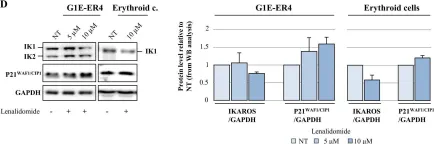
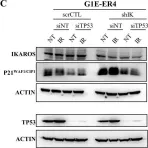
In PLoS Genet on 1 March 2021 by Lemarié, M., Bottardi, S., et al.
Fig.4.C

-
WB
-
Mus musculus (House mouse)
Collected and cropped from PLoS Genet by CiteAb, provided under a CC-BY license
Image 1 of 88
In PLoS Genet on 1 March 2021 by Lemarié, M., Bottardi, S., et al.
Fig.8.D

-
WB
-
Mus musculus (House mouse)
Collected and cropped from PLoS Genet by CiteAb, provided under a CC-BY license
Image 1 of 88
In PLoS Genet on 1 March 2021 by Lemarié, M., Bottardi, S., et al.
Fig.8.C

-
WB
-
Mus musculus (House mouse)
Collected and cropped from PLoS Genet by CiteAb, provided under a CC-BY license
Image 1 of 88
In Cell Death Discov on 8 February 2021 by Lee, J. J., Park, I. H., et al.
Fig.5.A

-
WB
-
Collected and cropped from Cell Death Discov by CiteAb, provided under a CC-BY license
Image 1 of 88
In Cell Death Discov on 8 February 2021 by Lee, J. J., Park, I. H., et al.
Fig.5.D

-
WB
-
Collected and cropped from Cell Death Discov by CiteAb, provided under a CC-BY license
Image 1 of 88
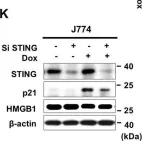
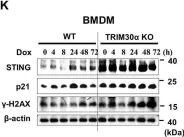
In Cell Death Discov on 8 February 2021 by Lee, J. J., Park, I. H., et al.
Fig.1.K

-
WB
-
Collected and cropped from Cell Death Discov by CiteAb, provided under a CC-BY license
Image 1 of 88
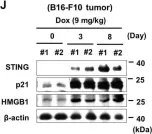
In Cell Death Discov on 8 February 2021 by Lee, J. J., Park, I. H., et al.
Fig.1.E

-
WB
-
Collected and cropped from Cell Death Discov by CiteAb, provided under a CC-BY license
Image 1 of 88
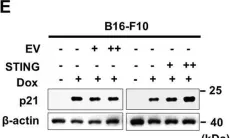
In Cell Death Discov on 8 February 2021 by Lee, J. J., Park, I. H., et al.
Fig.2.J

-
WB
-
Collected and cropped from Cell Death Discov by CiteAb, provided under a CC-BY license
Image 1 of 88
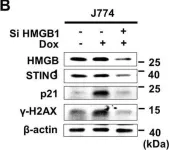
In Cell Death Discov on 8 February 2021 by Lee, J. J., Park, I. H., et al.
Fig.2.B

-
WB
-
Collected and cropped from Cell Death Discov by CiteAb, provided under a CC-BY license
Image 1 of 88
In Cell Death Discov on 8 February 2021 by Lee, J. J., Park, I. H., et al.
Fig.3.K

-
WB
-
Collected and cropped from Cell Death Discov by CiteAb, provided under a CC-BY license
Image 1 of 88