CD4 T cell responses constitute an important component of adaptive immunity and are critical regulators of anti-microbial protection. CD4+ T cells expressing CD32a have been identified as a target for HIV. CD32a is an Fcγ receptor known to be expressed on myeloid cells, granulocytes, B cells and NK cells. Little is known about the biology of CD32+CD4+ T cells. Our goal was to understand the dynamics of CD32+CD4+ T cells in tissues. We analyzed these cells in the blood, lymph nodes, spleen, ileum, jejunum and liver of two nonhuman primate models frequently used in biomedical research: African green monkeys (AGM) and macaques. We studied them in healthy animals and during viral (SIV) infection. We performed phenotypic and transcriptomic analysis at different stages of infection. In addition, we compared CD32+CD4+ T cells in tissues with well-controlled (spleen) and not efficiently controlled (jejunum) SIV replication in AGM. The CD32+CD4+ T cells more frequently expressed markers associated with T cell activation and HIV infection (CCR5, PD-1, CXCR5, CXCR3) and had higher levels of actively transcribed SIV RNA than CD32-CD4+T cells. Furthermore, CD32+CD4+ T cells from lymphoid tissues strongly expressed B-cell-related transcriptomic signatures, and displayed B cell markers at the cell surface, including immunoglobulins CD32+CD4+ T cells were rare in healthy animals and blood but increased strongly in tissues with ongoing viral replication. CD32+CD4+ T cell levels in tissues correlated with viremia. Our results suggest that the tissue environment induced by SIV replication drives the accumulation of these unusual cells with enhanced susceptibility to viral infection.
Copyright © 2021 Huot, Rascle, Planchais, Contreras, Passaes, Le Grand, Beignon, Kornobis, Legendre, Varet, Saez-Cirion, Mouquet, Jacquelin and Müller-Trutwin.
Product Citations: 50
In Frontiers in Immunology on 6 July 2021 by Huot, N., Rascle, P., et al.
-
Immunology and Microbiology
In The Journal of Immunology on 15 June 2020 by Adam, L., Tchitchek, N., et al.
DNA vaccines delivered with electroporation (EP) have shown promising results in preclinical models and are evaluated in clinical trials. In this study, we aim to characterize early mechanisms occurring in the skin after intradermal injection and EP of the auxoGTUmultiSIV DNA vaccine in nonhuman primates. First, we show that EP acts as an adjuvant by enhancing local inflammation, notably via granulocytes, monocytes/macrophages, and CD1aint-expressing cell recruitment. EP also induced Langerhans cell maturation, illustrated by CD86, CD83, and HLA-DR upregulation and their migration out of the epidermis. Second, we demonstrate the crucial role of the DNA vaccine in soluble factors release, such as MCP-1 or IL-15. Transcriptomic analysis showed that EP played a major role in gene expression changes postvaccination. However, the DNA vaccine is required to strongly upregulate several genes involved in inflammatory responses (e.g., Saa4), cell migration (e.g., Ccl3, Ccl5, or Cxcl10), APC activation (e.g., Cd86), and IFN-inducible genes (e.g., Ifit3, Ifit5, Irf7, Isg15, orMx1), illustrating an antiviral response signature. Also, AIM-2, a cytosolic DNA sensor, appeared to be strongly upregulated only in the presence of the DNA vaccine and trends to positively correlate with several IFN-inducible genes, suggesting the potential role of AIM-2 in vaccine sensing and the subsequent innate response activation leading to strong adaptive T cell responses. Overall, these results demonstrate that a combined stimulation of the immune response, in which EP and the auxoGTUmultiSIV vaccine triggered different components of the innate immunity, led to strong and persistent cellular recall responses.
Copyright © 2020 by The American Association of Immunologists, Inc.
-
Genetics
-
Immunology and Microbiology
TIGIT-Fc Promote Immune Tolerance at the Feto-maternal Interface
Preprint on BioRxiv : the Preprint Server for Biology on 28 October 2019 by Fu, W., Ma, Z., et al.
The perfect synchronization of maternal immune-endocrine mechanisms and those of the foetus is necessary for a successful pregnancy. In this report, decidual immune cells at the maternal-foetal interface were detected that expressed TIGIT (T cell immunoreceptor with Ig and ITIM domains), which is a co-inhibitory receptor that triggers immunological tolerance. We generated recombinant TIGIT-Fc fusion proteins by linking the extracellular domain of TIGIT and silent Fc fragments. The treatment with TIGIT-Fc of human decidual dendritic cells (dDCs) increased the production of interleukin 10 and induced the dDCs to powerfully polarize the decidual CD4 + T cells towards a classic T H 2 phenotype. The administration of TIGIT-Fc to CBA/J pregnant mice at preimplantation induced CD4 + forkhead box P3 + (Foxp3 + ) regulatory T cells and tolerogenic dendritic cells and increased pregnancy rates in a mouse model of abortive stress. Moreover, we proposed that progesterone play a direct role in the transcriptional regulation of the TIGIT gene in decidual immune cell subsets. The results suggested the therapeutic potential of the TIGIT-Fc fusion protein in reinstating immune tolerance in failing pregnancies.
-
Immunology and Microbiology
In Scientific Reports on 8 March 2019 by Gross, D. A., Leborgne, C., et al.
The discovery of tumor-associated antigens recognized by T lymphocytes opens the possibility of vaccinating cancer patients with defined antigens. However, one of the major limitation of peptide-based vaccines is the low immunogenicity of antigenic peptides. Interestingly, if these epitopes are directly delivered into the cytoplasm of antigen presenting cells, they can be efficiently presented via the direct MHC class I presentation pathway. To improve antigen entry, one promising approach is the use of cell penetrating peptides (CPPs). However, most studies use a covalent binding of the CPP with the antigen. In the present study, we focused on the C-terminal domain of Vpr which was previously demonstrated to efficiently deliver plasmid DNA into cells. We provide evidence that the peptides Vpr55-91 and Vpr55-82 possess the capacity of delivering proteins and epitopes into cell lines as well as into human primary dendritic cells, without the necessicity for a chemical linkage. Moreover, immunization of HLA-A2 transgenic mice with Vpr55-91 as the sole adjuvant is able to induce antigen-specific cytotoxic T lymphocytes against multiple tumor epitopes.
-
Cancer Research
-
Immunology and Microbiology
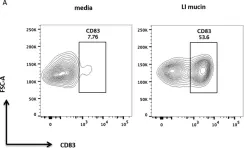
Intestinal mucin activates human dendritic cells and IL-8 production in a glycan-specific manner.
In The Journal of Biological Chemistry on 1 June 2018 by Melo-González, F., Fenton, T. M., et al.
Cross-talk between different components of the intestinal barrier and the immune system may be important in maintaining gut homeostasis. A crucial part of the gut barrier is the mucus layer, a cross-linked gel on top of the intestinal epithelium that consists predominantly of the mucin glycoprotein MUC2. However, whether the mucin layer actively regulates intestinal immune cell responses is not clear. Because recent evidence suggests that intestinal dendritic cells (DCs) may be regulated by the mucus layer, we purified intestinal mucin, incubated it with human DCs, and determined the functional effects. Here we show that expression of the chemokine IL-8 and co-stimulatory DC markers CD86 and CD83 are significantly up-regulated on human DCs in the presence of intestinal mucins. Additionally, mucin-exposed DCs promoted neutrophil migration in an IL-8-dependent manner. The stimulatory effects of mucins on DCs were not due to mucin sample contaminants such as lipopolysaccharide, DNA, or contaminant proteins. Instead, mucin glycans are important for the pro-inflammatory effects on DCs. Thus, intestinal mucins are capable of inducing important pro-inflammatory functions in DCs, which could be important in driving inflammatory responses upon intestinal barrier damage.
© 2018 by The American Society for Biochemistry and Molecular Biology, Inc.
-
FC/FACS
-
Biochemistry and Molecular biology
-
Immunology and Microbiology
In J Biol Chem on 1 June 2018 by Melo-González, F., Fenton, T. M., et al.
Fig.2.A

-
FC/FACS
-
Collected and cropped from J Biol Chem by CiteAb, provided under a CC-BY license
Image 1 of 2
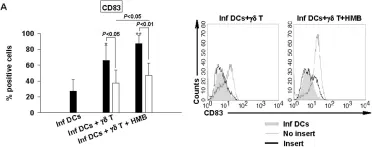
In PLoS One on 29 August 2012 by Ni, M., Martire, D., et al.
Fig.3.A

-
FC/FACS
-
Collected and cropped from PLoS One by CiteAb, provided under a CC-BY license
Image 1 of 2