Patients with acute myeloid leukemia (AML) often carry the same gene mutations. Neoantigens encoded by these mutations are attractive targets for immunotherapy.
We searched for public human leukocyte antigen (HLA) class II-restricted neoantigens on AML using an in vitro T cell stimulation method. Peptides from 26 recurrent genetic aberrations were assessed for predicted HLA class II binding, and 24 long neopeptides encoded by 10 recurrent mutations were synthesized. Naive CD4 T cells from healthy individuals were cocultured with autologous dendritic cells pulsed with neopeptides.
Multiple CD4 T cell clones were isolated that recognized neopeptides encoded by 5 different genetic aberrations. Two of these peptides, one from the well-known DNMT3A-R882H hotspot mutation and one from a long alternative reading frame created by frameshift mutations in RUNX1, were recognized by CD4 T cell clones after endogenous processing and presentation on cell lines transduced or CRISPR-Cas9-edited with the mutation of interest. The T cell clone for DNMT3A-R882H was also activated upon stimulation with primary AML samples from HLA-DQB1*06:02 or -DQB1*06:03 positive patients with the mutation.
We here identified a public HLA class II-restricted neoantigen encoded by a driver mutation occurring in 10% of patients with AML that could become an important target for immunotherapy to treat patients with DNMT3A-R882H-mutated AML.
Copyright © 2025 van der Lee, Argiro, Laan, Honders, de Jong, Struckman, Falkenburg and Griffioen.
Product Citations: 52
Mutated DNMT3A creates a public HLADQ- binding neoantigen on acute myeloid leukemia.
In Frontiers in Immunology on 28 March 2025 by van der Lee, D. I., Argiro, E. M., et al.
-
FC/FACS
-
Homo sapiens (Human)
-
Cancer Research
-
Immunology and Microbiology
In Cellular and Molecular Life Sciences : CMLS on 11 July 2024 by Kushida, Y., Oguma, Y., et al.
Muse cells, identified as cells positive for the pluripotent surface marker SSEA-3, are pluripotent-like endogenous stem cells located in the bone marrow (BM), peripheral blood, and organ connective tissues. The detailed characteristics of SSEA-3(+) cells in extraembryonic tissue, however, are unknown. Here, we demonstrated that similar to human-adult tissue-Muse cells collected from the BM, adipose tissue, and dermis as SSEA-3(+), human-umbilical cord (UC)-SSEA-3(+) cells express pluripotency markers, differentiate into triploblastic-lineage cells at a single cell level, migrate to damaged tissue, and exhibit low telomerase activity and non-tumorigenicity. Notably, ~ 20% of human-UC-SSEA-3(+) cells were negative for X-inactive specific transcript (XIST), a naïve pluripotent stem cell characteristic, whereas all human adult tissue-Muse cells are XIST-positive. Single-cell RNA sequencing revealed that the gene expression profile of human-UC-SSEA-3(+) cells was more similar to that of human post-implantation blastocysts than human-adult tissue-Muse cells. The DNA methylation level showed the same trend, and notably, the methylation levels in genes particularly related to differentiation were lower in human-UC-SSEA-3(+) cells than in human-adult tissue-Muse cells. Furthermore, human-UC-SSEA-3(+) cells newly express markers specific to extraembryonic-, germline-, and hematopoietic-lineages after differentiation induction in vitro whereas human-adult tissue-Muse cells respond only partially to the induction. Among various stem/progenitor cells in living bodies, those that exhibit properties similar to post-implantation blastocysts in a naïve state have not yet been found in humans. Easily accessible human-UC-SSEA-3(+) cells may be a valuable tool for studying early-stage human development and human reproductive medicine.
© 2024. The Author(s).
-
Biochemistry and Molecular biology
Preprint on Research Square on 20 June 2024 by Kushida, Y., Oguma, Y., et al.
Abstract Muse cells, identified as cells positive for the pluripotent surface marker SSEA-3, are pluripotent-like endogenous stem cells located in the bone marrow (BM), peripheral blood, and organ connective tissues. The detailed characteristics of SSEA-3(+) cells in extraembryonic tissue, however, are unknown. Here, we demonstrated that similar to human-adult tissue-Muse cells collected from the BM, adipose tissue, and dermis as SSEA-3(+), human-umbilical cord (UC)-SSEA-3(+) cells express pluripotency markers, differentiate into triploblastic-lineage cells at a single cell level, migrate to damaged tissue, and exhibit low telomerase activity and non-tumorigenicity. Notably, ~ 20% of human-UC-SSEA-3(+) cells were negative for X-inactive specific transcript (XIST), a naïve pluripotent stem cell characteristic, whereas all human adult tissue-Muse cells are XIST-positive. Single-cell RNA sequencing revealed that the gene expression profile of human-UC-SSEA-3(+) cells was more similar to that of human post-implantation blastocysts than human-adult tissue-Muse cells. The DNA methylation level showed the same trend, and notably, the methylation levels in genes particularly related to differentiation were lower in human-UC-SSEA-3(+) cells than in human-adult tissue-Muse cells. Furthermore, human-UC-SSEA-3(+) cells newly express markers specific to extraembryonic-, germline-, and hematopoietic-lineages after differentiation induction in vitro whereas human-adult tissue-Muse cells respond only partially to the induction. Among various stem/progenitor cells in living bodies, those that exhibit properties similar to post-implantation blastocysts in a naïve state have not yet been found in humans. Easily accessible human-UC-SSEA-3(+) cells may be a valuable tool for studying early-stage human development and human reproductive medicine.
In Regenerative Therapy on 1 June 2024 by Takahashi, K., Aritomi, S., et al.
Neural crest cells (NCCs) are cell populations that originate during the formation of neural crest in developmental stages. They are characterized by their multipotency, self-renewal and migration potential. Given their ability to differentiate into various types of cells such as neurons and Schwann cells, NCCs hold promise for cell therapy applications. The conventional method for obtaining NCCs involves inducing them from stem cells like induced pluripotent stem cells (iPSCs), followed by a long-term passage or purification using fluorescence-activated cell sorting (FACS). Although FACS allows high purity induced neural crest cells (iNCCs) to be obtained quickly, it is complex and costly. Therefore, there is a need for a simpler, cost-effective and less time-consuming method for cell therapy application.
To select differentiated iNCCs from heterogeneous cell populations quickly without using FACS, we adopted the use of scaffold material full-length laminin 211 (LN211), a recombinant, xeno-free protein suitable for cell therapy. After fist passage on LN211, iNCCs characterization was performed using polymerase chain reaction and flow cytometry. Additionally, proliferation and multipotency to various cells were evaluated.
The iNCCs obtained using our new method expressed cranial NCC- related genes and exhibited stable proliferation ability for at least 57 days, while maintaining high expression level of the NCCs marker CD271. They demonstrated differentiation ability into several cell types: neurons, astrocytes, melanocytes, smooth muscle cells, osteoblasts, adipocytes and chondrocytes. Furthermore, they could be induced to differentiate into induced mesenchymal stem cells (iMSCs) which retain the essential functions of somatic MSCs.
In this study, we have developed novel method for obtaining high purity iNCCs differentiated from iPSCs in a short time using LN211 under xeno-free condition. Compared with traditional methods, like FACS or long-term passage, this approach enables the acquisition of a large amount of cells at a lower cost and labor, and it is expected to contribute to stable supply of large scale iNCCs for future cell therapy applications.
© 2024 The Author(s).
-
Stem Cells and Developmental Biology
The immunopathological landscape of human pre-TCRα deficiency: From rare to common variants.
In Science on 1 March 2024 by Materna, M., Delmonte, O. M., et al.
We describe humans with rare biallelic loss-of-function PTCRA variants impairing pre-α T cell receptor (pre-TCRα) expression. Low circulating naive αβ T cell counts at birth persisted over time, with normal memory αβ and high γδ T cell counts. Their TCRα repertoire was biased, which suggests that noncanonical thymic differentiation pathways can rescue αβ T cell development. Only a minority of these individuals were sick, with infection, lymphoproliferation, and/or autoimmunity. We also report that 1 in 4000 individuals from the Middle East and South Asia are homozygous for a common hypomorphic PTCRA variant. They had normal circulating naive αβ T cell counts but high γδ T cell counts. Although residual pre-TCRα expression drove the differentiation of more αβ T cells, autoimmune conditions were more frequent in these patients compared with the general population.
-
FC/FACS
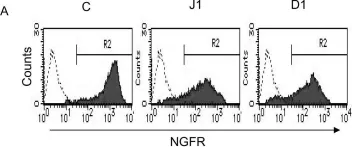
In PLoS One on 16 November 2016 by Ishige-Wada, M., Kwon, S. M., et al.
Fig.1.A

-
FC/FACS
-
Collected and cropped from PLoS One by CiteAb, provided under a CC-BY license
Image 1 of 1