The small intestine harbors a rich array of intestinal intraepithelial lymphocytes (IELs) that interact with structural cells to collectively sustain gut immune homeostasis. Dysregulation of gut immune homeostasis was implicated in the pathogenesis of multiple autoimmune diseases, however, whether this homeostasis is disrupted in a lupus autoimmune background remains unclear.
We performed single-cell RNA sequencing (scRNA-seq) analyses to elucidate immune and structural milieu in the intestinal epithelium of MRL/Lpr lupus mice (Lpr mice) and MRL/Mpj control mice (Mpj mice). Comprehensive analyses including unsupervised clustering, trajectories, and cellular communication were performed. The primary findings from scRNA-seq were further validated by quantitative polymerase chain reaction (qPCR), flow cytometry, and in vivo experiments including selenium supplementation.
We observed a significant reduction in CD8αα + IELs, accompanied by a marked increase in CD8αβ + IELs in Lpr mice. Additionally, subsets of CD8 + IELs exhibiting significantly enhanced effector functions were found to be markedly enriched in Lpr mice. Intercellular communication patterns within intestinal epithelial immune and structural cells were found to be specifically altered in Lpr mice. Moreover, scRNA-seq revealed significantly decreased intestinal TCRγδ T cells (γδT) associated with reduced aryl-hydrocarbon receptor repressor (AHRR) expression and subsequent oxidative stress and ferroptosis in Lpr mice. Antioxidant selenium effectively reversed the loss of γδT in Lpr mice, improved the gut barrier, and alleviated lupus symptoms.
Our high-resolution single-cell atlas enhances the understanding of the immune and structural milieu of intestinal epithelium in lupus and provides new insights into lupus pathogenesis mediated by intestinal immune dysregulation.
© 2025. The Author(s).
Product Citations: 48
In Journal of Translational Medicine on 27 January 2025 by Wang, Q., Wu, Y., et al.
-
Mus musculus (House mouse)
-
Immunology and Microbiology
The causative effect of CXCR7 on experimental autoimmune prostatitis injury and fibrosis.
In International Immunopharmacology on 10 January 2025 by Zhang, Y., Feng, R., et al.
Chronic prostatitis and Pelvic Pain syndrome (CP/CPPS) is an autoimmune inflammatory disease characterized by pelvic or perineal pain and infiltration of inflammatory cells in the prostate. C-X-C chemokine receptor type 7 (CXCR7) is an atypical chemokine receptor that has been shown to play a key role in inflammatory processes in prostate cancer. However, the role of CXCR7 in autoimmune prostate and immune regulation in CP/CPPS along with the mechanism of action for CXCR7 remains unclear. In this study, a mouse model of experimental autoimmune prostatitis (EAP) was constructed by subcutaneous injection of antigen, and CXCR7 agonist was administered to investigate the effects of CXCR7 on the proportion of immune cells and fibrosis in CP/CPPS. Western blotting, immunohistochemical staining and immunofluorescence, flow cytometry, and masson's trichrome staining were used to study the regulatory mechanisms of CXCR7 in immune regulation. CXCR7 agonists can significantly reduce pain and prostatic inflammation, and in vivo flow cytometry studies showed that the antagonists restored the imbalance of the Th17/Treg cell ratio. To elucidate the potential mechanisms by which CXCR7 influences the pathogenesis of CP/CPPS, we conducted simultaneous RNA-seq and non-targeted metabolome sequencing. Our findings suggest that CXCR7 agonists alleviate fibrosis in autoimmune prostatitis by inhibiting the TGFβ/SMAD pathway. This study provides the foundation to target the immunological function of CXCR7 as a novel therapy for CP/CPPS.
Copyright © 2024. Published by Elsevier B.V.
-
FC/FACS
-
Mus musculus (House mouse)
-
Immunology and Microbiology
Effects of PACAP Deficiency on Immune Dysfunction and Peyer's Patch Integrity in Adult Mice.
In International Journal of Molecular Sciences on 3 October 2024 by Sparks, J., Meggyes, M., et al.
PACAP (pituitary adenylate cyclase activating polypeptide) is a widespread neuropeptide with cytoprotective and anti-inflammatory effects. It plays a role in innate and adaptive immunity, but data are limited about gut-associated lymphoid tissue. We aimed to reveal differences in Peyer's patches between wild-type (WT) and PACAP-deficient (KO) mice. Peyer's patch morphology from young (3-months-old) and aging (12-15-months-old) mice was examined, along with flow cytometry to assess immune cell populations, expression of checkpoint molecules (PD-1, PD-L1, TIM-3, Gal-9) and functional markers (CD69, granzyme B, perforin) in CD3+, CD4+, and CD8+ T cells. We found slight differences between aging, but not in young, WT, and KO mice. In WT mice, aging reduced CD8+ T cell numbers frequency and altered checkpoint molecule expression (higher TIM-3, granzyme B; lower Gal-9, CD69). CD4+ T cell frequency was higher with similar checkpoint alterations, indicating a regulatory shift. In PACAP KO mice, aging did not change cell population frequencies but led to higher TIM-3, granzyme B and lower PD-1, PD-L1, Gal-9, and CD69 expression in CD4+ and CD8+ T cells, with reduced overall T cell activity. Thus, PACAP deficiency impacts immune dysfunction by altering checkpoint molecules and T cell functionality, particularly in CD8+ T cells, suggesting complex immune responses by PACAP, highlighting its role in intestinal homeostasis and potential implications for inflammatory bowel diseases.
-
Mus musculus (House mouse)
-
Immunology and Microbiology
In Advanced Science (Weinheim, Baden-Wurttemberg, Germany) on 1 October 2024 by Chen, J., Zhao, D., et al.
Mortality rates due to lung cancer are high worldwide. Although PD-1 and PD-L1 immune checkpoint inhibitors boost the survival of patients with non-small-cell lung cancer (NSCLC), resistance often arises. The Warburg Effect, which causes lactate build-up and potential lysine-lactylation (Kla), links immune dysfunction to tumor metabolism. The role of non-histone Kla in tumor immune microenvironment and immunotherapy remains to be clarified. Here, global lactylome profiling and metabolomic analyses of samples from patients with NSCLC is conducted. By combining multi-omics analysis with in vitro and in vivo validation, that intracellular lactate promotes extracellular lipolysis through lactyl-APOC2 is revealed. Mechanistically, lactate enhances APOC2 lactylation at K70, stabilizing it and resulting in FFA release, regulatory T cell accumulation, immunotherapy resistance, and metastasis. Moreover, the anti-APOC2K70-lac antibody that sensitized anti-PD-1 therapy in vivo is developed. This findings highlight the potential of anti lactyl-APOC2-K70 approach as a new combination therapy for sensitizing immunotherapeutic responses.
© 2024 The Author(s). Advanced Science published by Wiley‐VCH GmbH.
-
Immunology and Microbiology
In BioFactors (Oxford, England) on 4 August 2024 by Das, A., Mitra, A., et al.
Groundwater arsenic is a notorious toxicant and exposure to environmentally relevant concentrations persists as a healthcare burden across the world. Arsenic has been reported to jeopardize the normal functioning of the immune system, but there are still gaps in the understanding of thymic T cell biology. Immunotoxic influence of arsenic in thymic integrity demands a potent restorative molecule. The objectives of this study were to examine key signaling cross-talks associated with arsenic-induced immune alterations in the thymus and propose melatonin as a potential candidate against immunological complications arising from arsenic exposure. Swiss albino mice were exposed to sodium arsenite (0.05 mg/L; in drinking water) and melatonin (IP:10 mg/kg BW) for 28 days. Melatonin successfully protected thymus from arsenic-mediated tissue degeneration and maintained immune homeostasis including T cell maturation and proliferation by mitigating oxidative stress through Nrf2 upregulation. Additionally, melatonin exerted ameliorative effect against arsenic-induced apoptosis and inflammation by inhibiting p53-mediated mitochondrial cell death pathway and NF-κB-p65/STAT3-mediated proinflammatory pathway, respectively. For the first time, we showed that arsenic-induced profibrotic changes were inhibited by melatonin through targeting of inflammation-associated EMT. Our findings clearly demonstrate that melatonin can be a viable and promising candidate in combating arsenic-induced immune toxicity with no collateral damage, making it an important research target.
© 2024 International Union of Biochemistry and Molecular Biology.
-
Immunology and Microbiology
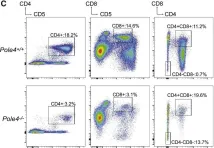
In Mol Cell on 17 May 2018 by Bellelli, R., Borel, V., et al.
Fig.2.C

-
FC/FACS
-
Mus musculus (House mouse)
Collected and cropped from Mol Cell by CiteAb, provided under a CC-BY license
Image 1 of 2
In Mol Cell on 17 May 2018 by Bellelli, R., Borel, V., et al.
Fig.6.D

-
FC/FACS
-
Mus musculus (House mouse)
Collected and cropped from Mol Cell by CiteAb, provided under a CC-BY license
Image 1 of 2