Product Citations: 49
The astrocyte marker ALDH1L1 also identifies a stromal cell population in the lymph node.
In Scientific Reports on 9 February 2026 by Smith, B. C., Nasrallah, M. J., et al.
-
IHC-IF
-
Mus musculus (House mouse)
-
Neuroscience
Pathogenicity of a novel orthonairovirus, Iwanai Valley virus, in mice.
In The Journal of Veterinary Medical Science / the Japanese Society of Veterinary Science on 1 February 2026 by Nishino, A., Park, E. S., et al.
Iwanai Valley virus (IWVV) is a novel tick-borne orthonairovirus classified into the Tamdy genogroup. Since several orthonairoviruses in the Tamdy and closely related Sulina genogroups have been associated with human diseases, evaluating the pathogenic potential of IWVV is an important public health priority. Despite the increase in reports of orthonairoviruses belonging to the Tamdy and Sulina genogroups in East Asia, studies on their pathogenicity remain limited. In this study, we established a mouse model of IWVV infection using type I interferon receptor knockout (IFNAR1-/-) mice and analyzed the pathogenicity of IWVV infection. IWVV infection was lethal in IFNAR1-/-mice but did not cause any significant symptoms in BALB/c mice. IWVV replicated well in the liver and spleen of IFNAR1-/-mice, leading to acute necrotizing hepatitis and damage to the spleen, characterized by lymphocyte depletion. These pathological features are consistent with those observed in immunocompromised mice infected with other human-pathogenic orthonairoviruses, such as Crimean-Congo hemorrhagic fever virus and Yezo virus. Although human infections with IWVV have not been reported, the observed virulence in animal models suggests that further evaluation of its zoonotic potential is warranted. Our findings highlight the importance of continued surveillance of emerging tick-borne IWVV to assess potential risks to public health.
-
IHC
-
Immunology and Microbiology
-
Veterinary Research
In Signal Transduction and Targeted Therapy on 15 December 2025 by Mikawa, T., Kameda, M., et al.
Cellular senescence is deeply involved in physiological homeostasis, development, tissue repair, aging, and diseases. Senescent cells (SnCs) accumulate in aged tissues and exert deleterious effects by secreting proinflammatory molecules that contribute to chronic inflammation and aging-related diseases. We revealed that an aberrant interaction between glycolytic PGAM1 and Chk1 kinase is augmented in SnCs associated with increased glycolysis, whose byproduct, lactate, promotes this binding in a noncell autonomous manner. The pseudo-Warburg effect of SnCs with enhanced PPP (pentose phosphate pathway) activity is maintained by HIF-2α phosphorylation by Chk1 and subsequent upregulation of glycolytic enzymes, creating a vicious cycle reprogramming the glycolytic pathway in SnCs. HIF-2α also activates FoxM1 expression, which transcriptionally suppresses proapoptotic profiles, including BIM, and upregulates DNA repair machineries in SnCs. FoxM1 thus supports the genomic integrity and survival capacity of SnCs during their glycolytic changes. Chemical abrogation of PGAM1-Chk1 binding reverts these phenotypes and eliminates SnCs through senolysis. Inhibition of the PGAM1-Chk1 interaction improves physiological parameters during aging and inhibits lung fibrosis in mouse models. Our study highlights a novel pathway contributing to the metabolic reprogramming of SnCs and how the use of a new senolytic molecule that targets the PGAM-Chk1 interaction creates a specific vulnerability of those cells to potentially fight age-related diseases.
© 2025. The Author(s).
-
IHC
-
Mus musculus (House mouse)
In EMBO Molecular Medicine on 1 April 2025 by Lu, Z., Stencel, O., et al.
Viral infections pose a significant global burden. Host susceptibility to pathogens is determined by many factors including genetic variation that can lead to immunodeficient or dysregulated antiviral immune responses. Pax5 heterozygosity (Pax5-/+), resulting in reduced PAX5 levels in mice, mimics germline or somatic PAX5 dysregulation contributing to diseases such as childhood B-cell precursor acute lymphoblastic leukemia (B-ALL). In contrast to the well-characterized roles of PAX5 during early B-cell development, little is known about how Pax5 heterozygosity impacts antiviral responses. We infected Pax5-/+ mice with the noncytopathic Lymphocytic Choriomeningitis Virus (LCMV) and found that infection with the chronic Docile strain resulted in decreased survival of Pax5-/+ mice. While early adaptive CD8+ T-cell (CTL) immunity was robust in Pax5-/+ mice, LCMV-specific neutralizing antibody production was compromised leading to impaired long-term viral clearance and a pro-inflammatory milieu in the bone marrow (BM). Here we show that survival outcomes were improved upon prophylactic treatment with the β-glucan immune trainer through induction of heterologous protection against chronic infection. β-Glucan enhanced viral clearance, CTL immunity, neutralizing antibody production and reduced monocyte immunosuppression in multiple LCMV-resident host organs. New insight from this study will help design effective prophylactic treatment strategies against chronic viral infections, particularly in genetically predisposed susceptible hosts.
© 2025. The Author(s).
-
Biochemistry and Molecular biology
-
Immunology and Microbiology
In Cellular and Molecular Life Sciences : CMLS on 26 March 2025 by Montilla, A., Zabala, A., et al.
Interferon regulatory factor 5 (IRF5) is a transcription factor that plays a role in orchestrating innate immune responses, particularly in response to viral infections. Notably, IRF5 has been identified as a microglia risk gene linked to multiple sclerosis (MS), but its specific role in MS pathogenesis remains unclear. Through the use of Irf5-/- mice, our study uncovers a non-canonical function of IRF5 in MS recovery. Irf5-/- mice exhibited increased damage in an experimental autoimmune encephalomyelitis (EAE) model and demonstrated impaired oligodendrocyte recruitment into the lesion core following lysolecithin-induced demyelination. Transcriptomic and lipidomic analyses revealed that IRF5 has a role in microglia-mediated myelin phagocytosis, lipid metabolism, and cholesterol homeostasis. Indeed, Irf5-/- microglia phagocytose myelin, but myelin debris is not adequately degraded, leading to an accumulation of lipid droplets, cholesterol esters, and cholesterol crystals within demyelinating lesions. This abnormal buildup can hinder remyelination processes. Importantly, treatments that promote cholesterol transport were found to reduce lipid droplet accumulation and mitigate the exacerbated damage in Irf5-/- mice with EAE. Altogether, our study identified the antiviral transcription factor IRF5 as a key transcriptional regulator of lipid degradation and cholesterol homeostasis and suggest that loss of IRF5 function leads to pathogenic lipid accumulation in microglia, thereby obstructing remyelination. These data and the fact that Irf5 polymorphisms are significantly associated with MS, highlight IRF5 as a potential therapeutic target to promote regenerative responses.
© 2025. The Author(s).
-
Mus musculus (House mouse)
-
Biochemistry and Molecular biology
-
Cell Biology
-
Neuroscience
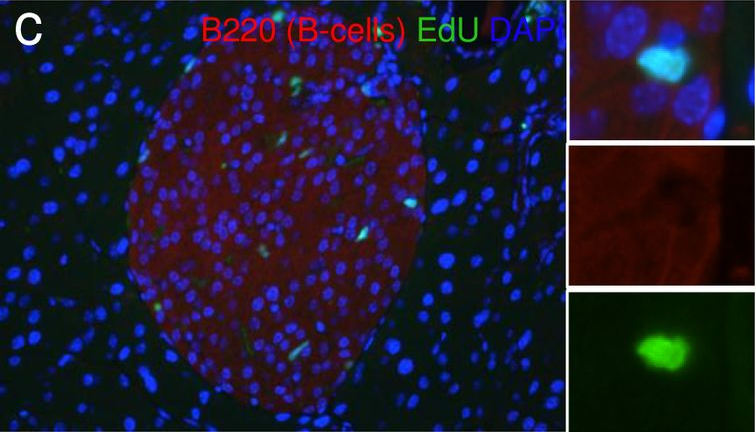
In PLoS One on 15 July 2016 by Cox, A. R., Barrandon, O., et al.
Fig.9.C
-
WB
-
Mus musculus (House mouse)
Collected and cropped from PLoS ONE by CiteAb, provided under a CC-BY license
Image 1 of 1