Extensive studies have demonstrated the relationship between metabolic reprogramming and the tumor microenvironment. Here, we characterized the head and neck squamous cell carcinoma (HNSCC) evolutionary landscape using spatial metabolomics/transcriptomics, single-cell transcriptomics, and bulk multi-omics. Metabolic heterogeneity during HNSCC malignant transformation was identified, with significant enrichment in the purine metabolism. Integrating single-cell and bulk data, we developed a robust ligand-receptor-based signature (LRS) linked to NT5E, a key upstream regulator of purine metabolism, which served as an independent prognostic indicator. The low LRS subtype was associated with a high proportion of immune cell infiltration and improved response to immunotherapy. Notably, in vitro and in vivo experiments demonstrated that AMIGO2, a core molecule within the LRS, regulates tumor-associated purine metabolism, and that its downregulation suppresses tumor cell invasion and migration, inhibits myofibroblast differentiation, and promotes immune effector cell infiltration. Moreover, combining AMIGO2 targeting with anti-PD-1 therapy yielded superior efficacy. Consistent validation was also obtained in a clinical cohort of HNSCC and premalignancy patients.
© 2025. The Author(s).
Product Citations: 71
In NPJ Precision Oncology on 18 November 2025 by Liu, G., Yao, X., et al.
-
FC/FACS
-
Mus musculus (House mouse)
-
Cancer Research
Branched actin networks mediate macrophage-dependent host-microbiota homeostasis.
In Science on 13 November 2025 by Vasconcellos, L. R. C., Huang, S., et al.
Branched actin networks formed by the Arp2/3 complex are essential for immune system function. Patients with loss-of-function mutations in the ARPC5 subunit of the Arp2/3 complex develop inflammation and immunodeficiency after birth, leading to early mortality. The basis for these phenotypes remains obscure. We found that loss of ARPC5, but not the ARPC5L isoform, in the mouse hematopoietic system caused early-onset intestinal inflammation after weaning. This condition was initiated by microbiota breaching the ileal mucosa and led to systemic inflammation. ARPC5-deficient macrophages and neutrophils infiltrated the ileum but failed to restrict microbial invasion. Specifically, macrophages that lack ARPC5 struggled to phagocytose and kill intracellular bacteria. Our results highlight the indispensable role of ARPC5-containing, but not ARPC5L-containing, Arp2/3 complexes in mononuclear phagocyte function and host-microbiota homeostasis.
-
Cell Biology
-
Immunology and Microbiology
A novel dual DYRK1A/B inhibitor for the treatment of type 1 diabetes.
In Frontiers in Pharmacology on 29 October 2025 by Tumas, S., Mingaila, J., et al.
Type 1 diabetes (T1D) is an autoimmune disease that leads to the progressive destruction of pancreatic β cells, resulting in insulin deficiency and hyperglycemia. Current treatments focus on insulin replacement, but novel therapeutic approaches targeting β cell regeneration are needed. Dual-specificity tyrosine-phosphorylation-regulated kinases 1A (DYRK1A) and 1B (DYRK1B) play key roles in cell cycle regulation and β cell proliferation.
In this study, FX8474, a novel DYRK1 inhibitor, was evaluated in a streptozotocin (STZ)-induced diabetic mouse model. Mice were treated orally for 7 days, and pharmacokinetics, glucose regulation, and immune cell profiling were assessed.
Pharmacokinetic analysis confirmed the oral bioavailability of FX8474, and treatment was associated with improved fasted glucose levels and glucose tolerance after a 7-day treatment. Immunophenotyping indicated that FX8474 treatment increases CD4+ memory T cell populations while decreasing CD4+ effector cells, as well as restores CD8+ T cell phenotypes to levels observed in healthy mice.
FX8474 has a modest effect on glucose regulation and immune cell composition, warranting further investigation into its potential therapeutic applications.
Copyright © 2025 Tumas, Mingaila, Baranauskas, Baltrukonytė, Orla, Krasko, Pocevičiūtė, Berlina, Belenky, Vilenchik, Vaitkevičienė, Potapova and Burokas.
-
Mus musculus (House mouse)
-
Pharmacology
In Nature Communications on 6 October 2025 by Kwon, K. W., Kang, T. G., et al.
Cancers are a risk factor for active tuberculosis (TB), and anti-cancer drugs can independently cause TB progression. To understand the underlying mechanisms, mice infected with Mycobacterium tuberculosis (Mtb) were treated with gemcitabine (Gem), cisplatin, or paclitaxel. These treatments delay Mtb-specific T cell responses, increase bacterial loads, and cause hyperinflammation with permissive neutrophils in the lungs. However, depleting Mtb-permissive neutrophils reduce bacterial levels and G-CSF production, thereby attenuating lung immunopathology. Additionally, Mtb-specific T cell responses generated by BCG vaccination inhibit bacterial growth and neutrophil infiltration even after Gem treatment. Gem induces granulocyte-biased generation in the bone marrow via G-CSF signaling, which led to lung neutrophil inflammation. However, pre-existing Mtb-specific T cell responses from BCG vaccination normalizes granulopoiesis by restricting G-CSF production. These findings show the mechanism of anti-cancer drug-induced neutrophilic lung inflammation in TB and highlight the role of Mtb-specific T cell responses in maintaining balanced hematopoiesis against Gem-induced TB immunopathogenesis.
© 2025. The Author(s).
-
Cancer Research
-
Immunology and Microbiology
Apolipoprotein E reduces the number and activation of non-invariant Natural Killer T cells
Preprint on BioRxiv : the Preprint Server for Biology on 7 July 2025 by Chakrabarti, R., Duddu, S., et al.
Natural Killer T (NKT) cells, which modulate atherosclerosis, include two groups – invariant (iNKT) and variant (vNKT). These subsets differentially regulate the disease progression. Yet, the role of vNKTs in atherosclerosis remains unclear. We induced atherosclerosis by feeding high-fat diet (HFD) to Apoe -/- and analyzed the vNKTs in the liver and spleen. The vNKTs were termed non-iNKTs since they were negatively selected within the NKT population. Available literature suggests NKTs as lipid-recognizing cells; however, to our surprise, the non-iNKT numbers and phenotype remained unchanged between HFD-fed and chow-fed Apoe -/- . This was a blindsiding and unexpected outcome of the non-iNKTs being unaltered and unaffected with or without HFD, indicating no observable impact of atherosclerosis on these subsets. Albeit remaining unperturbed by atherosclerosis, these non-iNKTs demonstrated an identical but unique increase and upregulated activation in both the chow and HFD-fed Apoe -/- . These results instigated an investigation of the baseline correlation of the non-iNKTs between young C57BL/6 (WT) and Apoe -/- . Previously unknown and confounding results revealed upregulated activation and increased non-iNKT numbers but decreased IL-4 + non-iNKTs in the Apoe -/- compared to WT. Furthermore, HFD-fed WT that developed dyslipidemia, elucidated increased hepatic non-iNKTs and splenic IFN-γ + non-iNKTs compared to chow-fed WT controls. These results were not perceived in chow and HFD-fed Apoe -/- . Although lipid-responsive, non-iNKTs in Apoe -/- mice failed to respond to lipid stress, unlike those in dyslipidemic WT mice. These findings reveal that loss of Apoe , rather than atherosclerosis itself, drives altered non-iNKT biology. Thus, Apoe deficiency intrinsically dysregulates non-iNKTs, masking disease-associated immune changes. Apoe loss alters non-iNKT number and function, independent of atherosclerosis, and challenges the interpretation of immune responses by NKT subsets in Apoe -/- models. Therefore, this study warrants the use of Apoe null mice in studying NKT cells.
-
FC/FACS
-
Immunology and Microbiology
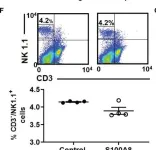
In Front Immunol on 4 June 2022 by Wong, S. W., McCarroll, J., et al.
Fig.6.F
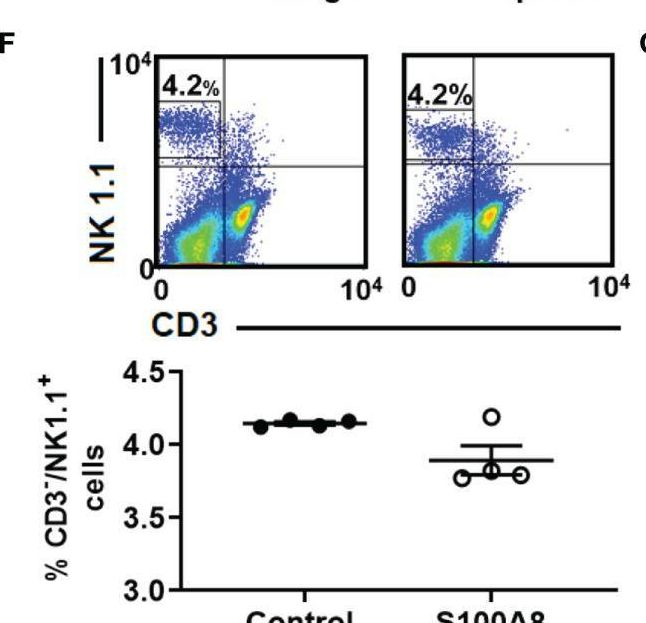
-
FC/FACS
-
Collected and cropped from Frontiers in Immunology by CiteAb, provided under a CC-BY license
Image 1 of 3
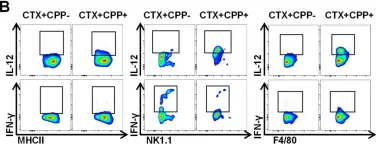
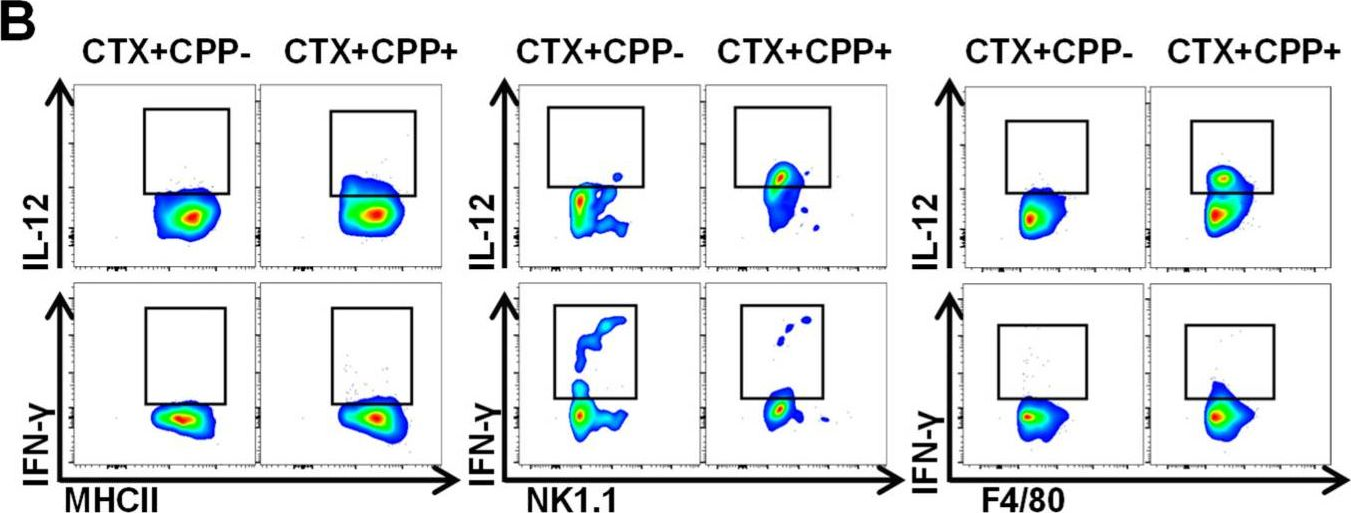
In Nutrients on 9 September 2020 by Sun, P., Kim, Y., et al.
Fig.3.B
-
FC/FACS
-
Mus musculus (House mouse)
Collected and cropped from Nutrients by CiteAb, provided under a CC-BY license
Image 1 of 3
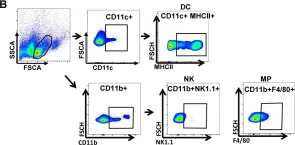
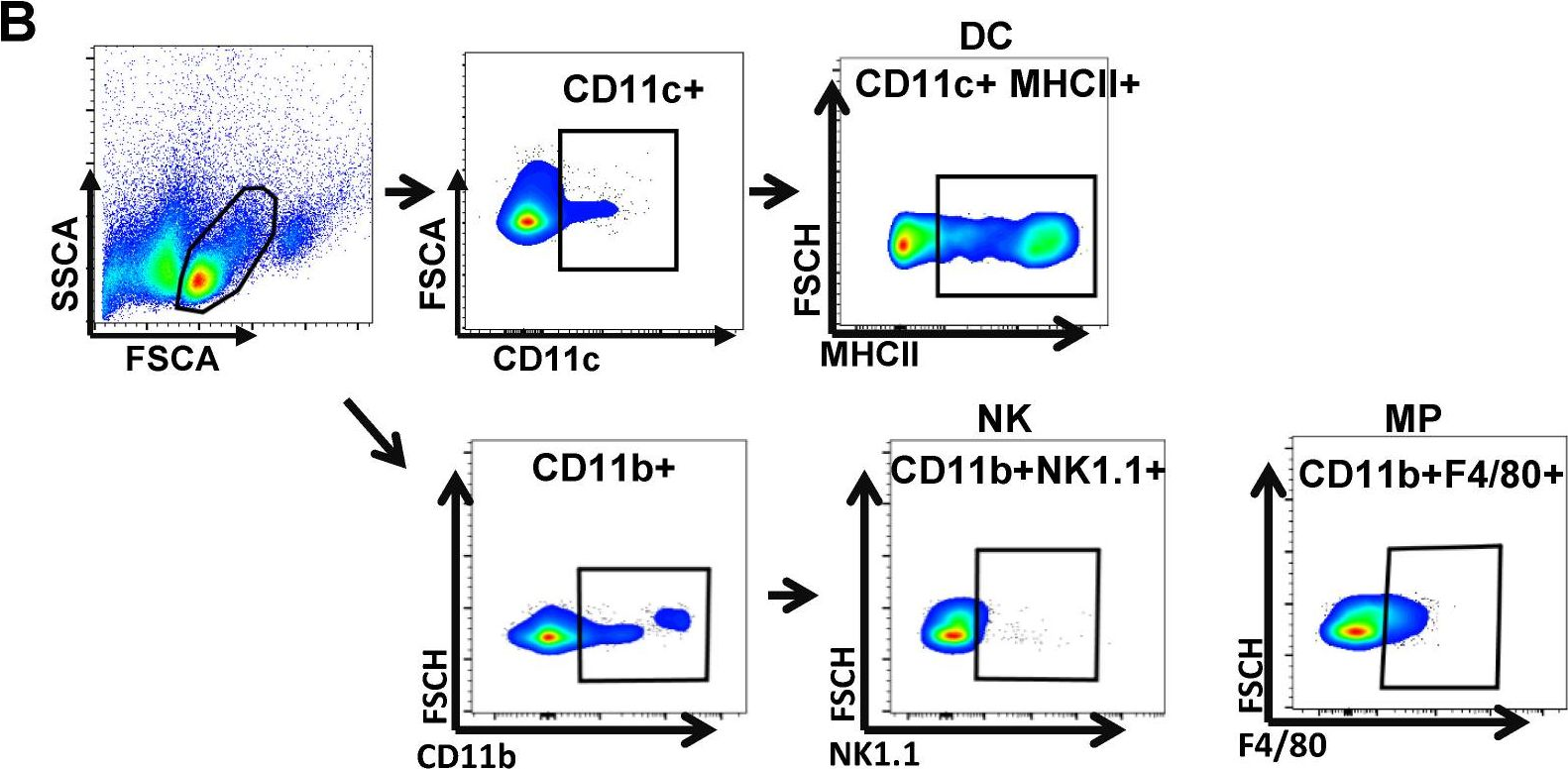
In Nutrients on 9 September 2020 by Sun, P., Kim, Y., et al.
Fig.2.B
-
FC/FACS
-
Mus musculus (House mouse)
Collected and cropped from Nutrients by CiteAb, provided under a CC-BY license
Image 1 of 3