The concept of intravascular immunity is reshaping our understanding of immune surveillance, challenging the traditional view that immune responses are confined to either tissue parenchyma or the bloodstream. Our pilot study utilized intravascular staining (ivs) in rhesus macaques (RhM) to spatially distinguish pulmonary vasculature-associated (ivs+) CD8+ T cells from interstitial (ivs-) T cells. Single-cell RNA sequencing and flow cytometry revealed that ivs+ T cells are not passive blood contaminants but rather a distinct, "resident-like" population enriched in cytotoxic effectors, with elevated expression of transcripts associated with tissue residency, cell adhesion, and vascular/platelet interactions, while ivs- T cells exhibited a classic TRM signature. The presence of SIV-specific tetramer+ ivs+ CD8+ T cells in infected RhM suggests a role in antiviral defense within the lung vasculature. These findings provide new insight into intravascular immunity, highlighting a unique population of CD8+ T cells as potential pulmonary vascular sentinels poised for rapid immune responses.
© 2025 The Author(s).
Product Citations: 109
In IScience on 20 June 2025 by Melton, A., Potter, E. L., et al.
-
Cardiovascular biology
-
Immunology and Microbiology
In Nature Communications on 10 May 2025 by Chen, J. L., Wang, B., et al.
In 2022, a global mpox outbreak occurred, and remains a concern today. The T cell memory response to MPXV (monkeypox virus) infection has not been fully investigated. In this study, we evaluate this response in convalescent and MVA-BN (Modified Vaccinia Ankara - Bavarian Nordic) vaccinated individuals using VACV-infected cells. Strong CD8+ and CD4+ T cell responses are observed, and T cell responses are biased towards viral early expressed proteins. We identify seven immunodominant HLA-A*02:01 restricted MPXV-specific epitopes and focus our detailed phenotypic and scRNAseq analysis on the immunodominant HLA-A*02:01-G5R18-26-specific CD8+ T cell response. While tetramer+CD8+ T cells share similar differentiation and activation phenotypes, T cells from convalescent individuals show greater cytotoxicity, migratory potential to site of infection and TCR clonal expansion. Our data suggest that effective functional profiles of MPXV-specific memory T cells induced by Mpox infection may have an implication on the long-term protective responses to future infection.
© 2025. The Author(s).
-
ICC
-
Homo sapiens (Human)
-
Immunology and Microbiology
In IScience on 18 April 2025 by Karaliota, S., Moussa, M., et al.
A SARS-CoV-2 spike DNA vaccine formulated with a cationic nanoparticle emulsion (LION) was tested in Rhesus macaques. It induced robust, long-lasting (>2 years) cellular and humoral immunity, including increased neutralization breadth. T cell responses were predominantly CD8+, in contrast to other DNA vaccines. A rapid transient cytokine/chemokine response was associated with expansion and trafficking of myeloid cells and lymphocytes. Increased proliferation and dynamic changes between blood and lymph node (LN) were found for monocyte-derived cells, dendritic cells, and B and T cells, resulting in activation of LN and expansion of germinal centers (GCs), likely critical in shaping long-lasting adaptive immunity. Significant GC expansion of B, CD4-, and CD8- cells, including the Tfc3 subset, reflects a balanced immune response, including antibody (Ab) development. DNA/LION vaccination activates myeloid and lymphoid cells in blood and LN and promotes effective antigen presentation, resulting in sustained antigen-specific cellular and humoral responses, emerging as an effective DNA vaccine delivery platform.
-
Genetics
-
Immunology and Microbiology
Long-term immune responses to SARS-CoV-2 Omicron BA.4/5 mRNA booster in people living with HIV.
In Commun Med (Lond) on 27 March 2025 by Augello, M., Bono, V., et al.
Variant-adapted vaccines are recommended in vulnerable populations to address the waning immunity and the emergence of immune-escaping SARS-CoV-2 variants, yet data about immune responses to such vaccines in people living with HIV (PLWH) are limited. We therefore aimed to assess long-term immune responses to an original-BA.4/5 mRNA booster in this population.
In this prospective longitudinal study, PLWH receiving either an original-BA.4/5 bivalent booster or an original monovalent booster and HIV-negative healthcare workers (HCWs) receiving a bivalent booster were enrolled and sampled before (T0), 1 month (T1), and 4-9 months (T2) after the vaccine administration. SARS-CoV-2-specific T and B cells, RBD-binding antibodies, and RBD-blocking antibodies against both wild type (WT) and omicron BA.4/5 virus were determined.
The bivalent booster is able to transiently increase both humoral and polyfunctional T cell responses in PLWH, with humoral responses comparable to those observed in HCWs. While T cell responses are cross-reactive against viral variants and stable over time, humoral immunity is imprinted to the ancestral virus and wanes quickly. Furthermore, whilst previous SARS-CoV-2 infection does not affect the trajectory of vaccine-elicited immune responses, markers of HIV-related T cell dysfunction are associated with lower antibody peak responses and higher antibody waning. Lastly, the bivalent booster was superior to the monovalent one in inducing BA.4/5-reactive RBD-blocking antibodies.
The original-BA.4/5 bivalent booster is highly immunogenic in PLWH and superior to the monovalent one in inducing humoral responses against the BA.4/5 virus, although HIV-related T cell dysfunction markers are associated with blunted and less durable antibody immunity.
© 2025. The Author(s).
-
SARS Coronavirus (SARS-CoV)
-
COVID-19
-
Genetics
-
Immunology and Microbiology
In IScience on 21 March 2025 by Wei, Q., Foyn, H., et al.
FoxP3+ regulatory T cells (Tregs) are responsible for immune homeostasis by suppressing excessive anti-self-immunity. Tregs facilitate tumor growth by inhibiting anti-tumor immunity. Here, we explored the targeting of FoxP3 as a basis for new immunotherapies. In a high-throughput phenotypic screening of a drug repurposing library using human primary T cells, we identified quinacrine as a FoxP3 downregulator. In silico searches based on the structure of quinacrine, testing of sub-libraries of analogs in vitro, and validation identified a subset of 9-amino-acridines that selectively abrogated Treg suppressive functions. Mechanistically, these acridines interfered with the DNA-binding activity of FoxP3 and inhibited FoxP3-regulated downstream gene regulation. Release from Treg suppression by 9-amino-acridines increased anti-tumor immune responses both in cancer patient samples and in mice in a syngeneic tumor model. Our study highlights the feasibility of screening for small molecular inhibitors of FoxP3 as an approach to pursuing Treg-based immunotherapy.
© 2025 The Author(s).
-
Immunology and Microbiology
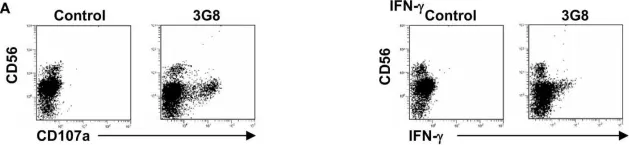
In Front Immunol on 7 December 2017 by Lajoie, L., Congy-Jolivet, N., et al.
Fig.2.A

-
FC/FACS
-
Collected and cropped from Front Immunol by CiteAb, provided under a CC-BY license
Image 1 of 3
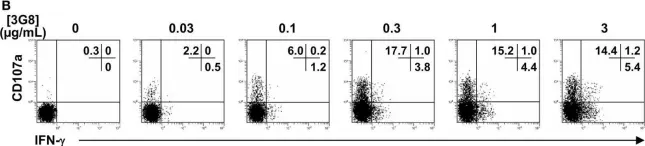
In Front Immunol on 7 December 2017 by Lajoie, L., Congy-Jolivet, N., et al.
Fig.2.B

-
FC/FACS
-
Collected and cropped from Front Immunol by CiteAb, provided under a CC-BY license
Image 1 of 3
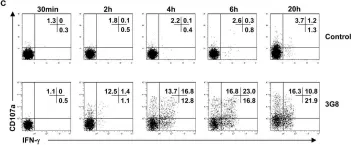
In Front Immunol on 7 December 2017 by Lajoie, L., Congy-Jolivet, N., et al.
Fig.2.C

-
FC/FACS
-
Collected and cropped from Front Immunol by CiteAb, provided under a CC-BY license
Image 1 of 3