Naive T cell activation requires both TCR and CD28 signals. The CARMIL2 cytosolic protein enables CD28-dependent activation of the NF-κB transcription factor via its ability to link CD28 to the CARD11 adaptor protein. Here, we developed mice expressing a mutation named Carmil2QE and mimicking a mutation found in human T cell malignancies. Naive T cells from Carmil2QE mice contained preformed CARMIL2QE-CARD11 complexes in numbers comparable to those assembling in wild-type T cells after CD28 engagement. Such ready-made CARMIL2QE-CARD11 complexes also formed in CD28-deficient mice where they unexpectedly induced most of the functions that normally result from CD28 engagement in a manner that remains antigen-dependent. In turn, tumor-specific T cells expressing Carmil2QE do not require CD28 engagement and thereby escape to both PD-1 and CTLA-4 inhibition. In conclusion, we uncovered the overarching role played by CARMIL2-CARD11 signals among those triggered by CD28 and exploited them to induce potent solid tumor-specific T cell responses in the absence of CD28 ligands and immune checkpoint inhibitors.
© 2025 Zhang et al.
Product Citations: 19
A CARMIL2 gain-of-function mutation suffices to trigger most CD28 costimulatory functions in vivo.
In The Journal of Experimental Medicine on 4 August 2025 by Zhang, F., Celis-Gutierrez, J., et al.
Generation of antitumor chimeric antigen receptors incorporating T cell signaling motifs.
In Science Signaling on 23 July 2024 by Balagopalan, L., Moreno, T., et al.
Chimeric antigen receptor (CAR) T cells have been used to successfully treat various blood cancers, but adverse effects have limited their potential. Here, we developed chimeric adaptor proteins (CAPs) and CAR tyrosine kinases (CAR-TKs) in which the intracellular ζ T cell receptor (TCRζ) chain was replaced with intracellular protein domains to stimulate signaling downstream of the TCRζ chain. CAPs contain adaptor domains and the kinase domain of ZAP70, whereas CAR-TKs contain only ZAP70 domains. We hypothesized that CAPs and CAR-TKs would be more potent than CARs because they would bypass both the steps that define the signaling threshold of TCRζ and the inhibitory regulation of upstream molecules. CAPs were too potent and exhibited high tonic signaling in vitro. In contrast, CAR-TKs exhibited high antitumor efficacy and significantly enhanced long-term tumor clearance in leukemia-bearing NSG mice as compared with the conventional CD19-28ζ-CAR-T cells. CAR-TKs were activated in a manner independent of the kinase Lck and displayed slower phosphorylation kinetics and prolonged signaling compared with the 28ζ-CAR. Lck inhibition attenuated CAR-TK cell exhaustion and improved long-term function. The distinct signaling properties of CAR-TKs may therefore be harnessed to improve the in vivo efficacy of T cells engineered to express an antitumor chimeric receptor.
-
WB
-
Biochemistry and Molecular biology
-
Immunology and Microbiology
Allosteric inhibition of the T cell receptor by a designed membrane ligand.
In eLife on 5 October 2023 by Ye, Y., Morita, S., et al.
The T cell receptor (TCR) is a complex molecular machine that directs the activation of T cells, allowing the immune system to fight pathogens and cancer cells. Despite decades of investigation, the molecular mechanism of TCR activation is still controversial. One of the leading activation hypotheses is the allosteric model. This model posits that binding of pMHC at the extracellular domain triggers a dynamic change in the transmembrane (TM) domain of the TCR subunits, which leads to signaling at the cytoplasmic side. We sought to test this hypothesis by creating a TM ligand for TCR. Previously we described a method to create a soluble peptide capable of inserting into membranes and binding to the TM domain of the receptor tyrosine kinase EphA2 (Alves et al., eLife, 2018). Here, we show that the approach is generalizable to complex membrane receptors, by designing a TM ligand for TCR. We observed that the designed peptide caused a reduction of Lck phosphorylation of TCR at the CD3ζ subunit in T cells. As a result, in the presence of this peptide inhibitor of TCR (PITCR), the proximal signaling cascade downstream of TCR activation was significantly dampened. Co-localization and co-immunoprecipitation in diisobutylene maleic acid (DIBMA) native nanodiscs confirmed that PITCR was able to bind to the TCR. AlphaFold-Multimer predicted that PITCR binds to the TM region of TCR, where it interacts with the two CD3ζ subunits. Our results additionally indicate that PITCR disrupts the allosteric changes in the compactness of the TM bundle that occur upon TCR activation, lending support to the allosteric TCR activation model. The TCR inhibition achieved by PITCR might be useful to treat inflammatory and autoimmune diseases and to prevent organ transplant rejection, as in these conditions aberrant activation of TCR contributes to disease.
© 2023, Ye et al.
-
WB
-
Immunology and Microbiology
Cell-targeted PD-1 agonists that mimic PD-L1 are potent T cell inhibitors.
In JCI Insight on 22 October 2021 by Curnock, A. P., Bossi, G., et al.
The PD-1/PD-L1 pathway is a key immune checkpoint that regulates T cell activation. There is strong rationale to develop PD-1 agonists as therapeutics against autoimmunity, but progress in this area has been limited. Here, we generated T cell receptor (TCR) targeting, PD-1 agonist bispecifics called ImmTAAI molecules that mimic the ability of PD-L1 to facilitate the colocalization of PD-1 with the TCR complex at the target cell-T cell interface. PD-1 agonist ImmTAAI molecules specifically bound to target cells and were highly effective in activating the PD-1 receptor on interacting T cells to achieve immune suppression. Potent PD-1 antibody ImmTAAI molecules closely mimicked the mechanism of action of endogenously expressed PD-L1 in their localization to the target cell-T cell interface, inhibition of proximal TCR signaling events, and suppression of T cell function. At picomolar concentrations, these bispecifics suppressed cytokine production and inhibited CD8+ T cell-mediated cytotoxicity in vitro. Crucially, in soluble form, the PD-1 ImmTAAI molecules were inactive and, hence, could avoid systemic immunosuppression. This study outlines a promising new route to generate more effective, potent, tissue-targeted PD-1 agonists that can inhibit T cell function locally with the potential to treat autoimmune and chronic inflammatory diseases of high unmet need.
-
WB
-
Homo sapiens (Human)
-
Immunology and Microbiology
In Communications Biology on 22 July 2021 by Dhar, P., Basher, F., et al.
Natural Killer (NK) cell dysfunction is associated with poorer clinical outcome in cancer patients. What regulates NK cell dysfunction in tumor microenvironment is not well understood. Here, we demonstrate that the human tumor-derived NKG2D ligand soluble MIC (sMIC) reprograms NK cell to secrete pro-tumorigenic cytokines with diminished cytotoxicity and polyfunctional potential. Antibody clearing sMIC restores NK cell to a normal cytotoxic effector functional state. We discovered that sMIC selectively activates the CBM-signalosome inflammatory pathways in NK cells. Conversely, tumor cell membrane-bound MIC (mMIC) stimulates NK cell cytotoxicity through activating PLC2γ2/SLP-76/Vav1 pathway. Ultimately, antibody targeting sMIC effectuated the in vivo anti-tumor effect of adoptively transferred NK cells. Our findings uncover an unrecognized mechanism that could instruct NK cell to a dysfunctional state in response to cues in the tumor microenvironment. Our findings provide a rationale for co-targeting sMIC to enhance the efficacy of the ongoing NK cell-based cancer immunotherapy.
© 2021. The Author(s).
-
WB
-
Homo sapiens (Human)
-
Cancer Research
-
Immunology and Microbiology
In Elife on 5 October 2023 by Ye, Y., Morita, S., et al.
Fig.2.A
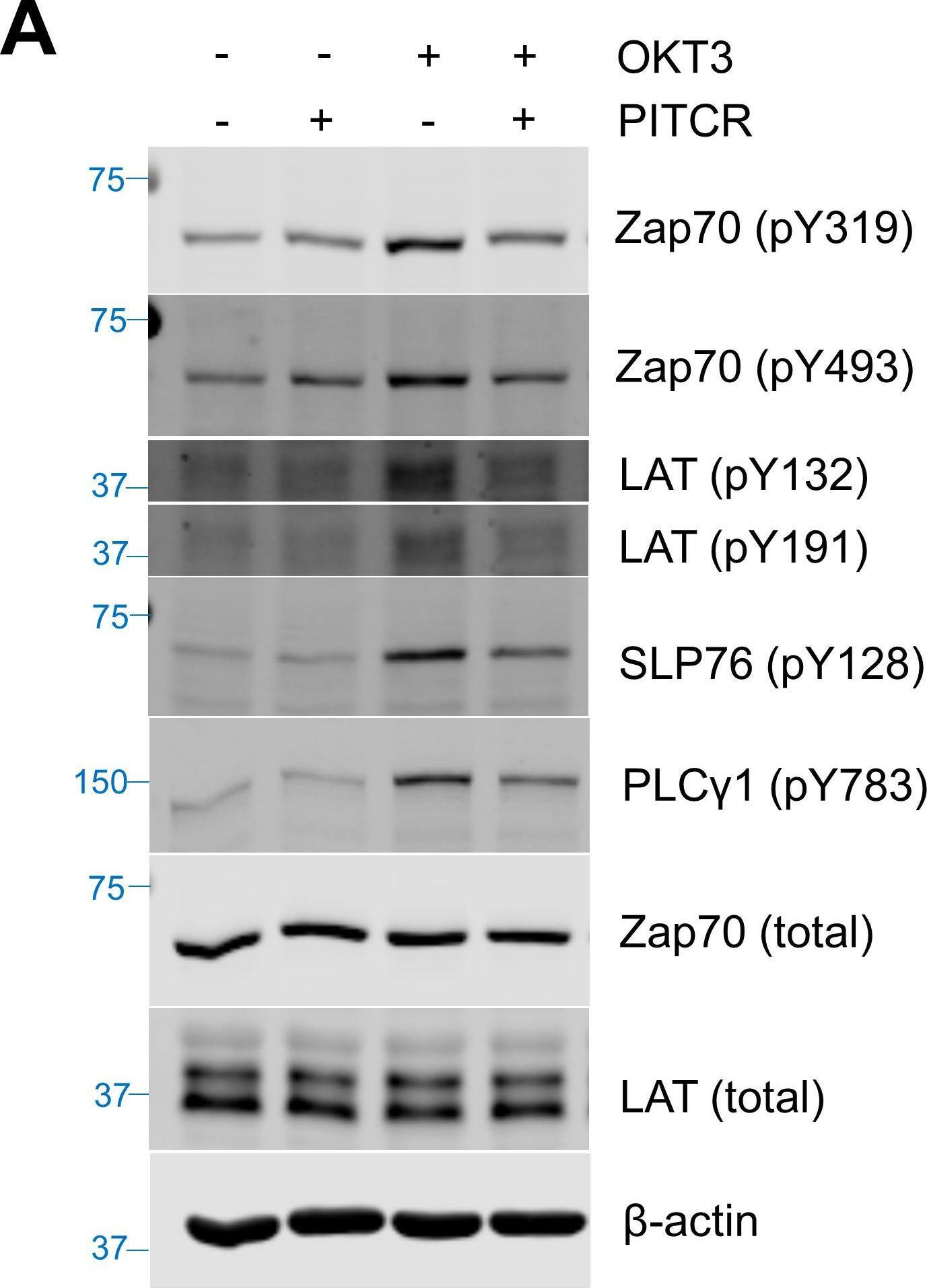
-
WB
-
Collected and cropped from eLife by CiteAb, provided under a CC-BY license
Image 1 of 2
In Cell Rep on 11 June 2019 by Celis-Gutierrez, J., Blattmann, P., et al.
Fig.3.A
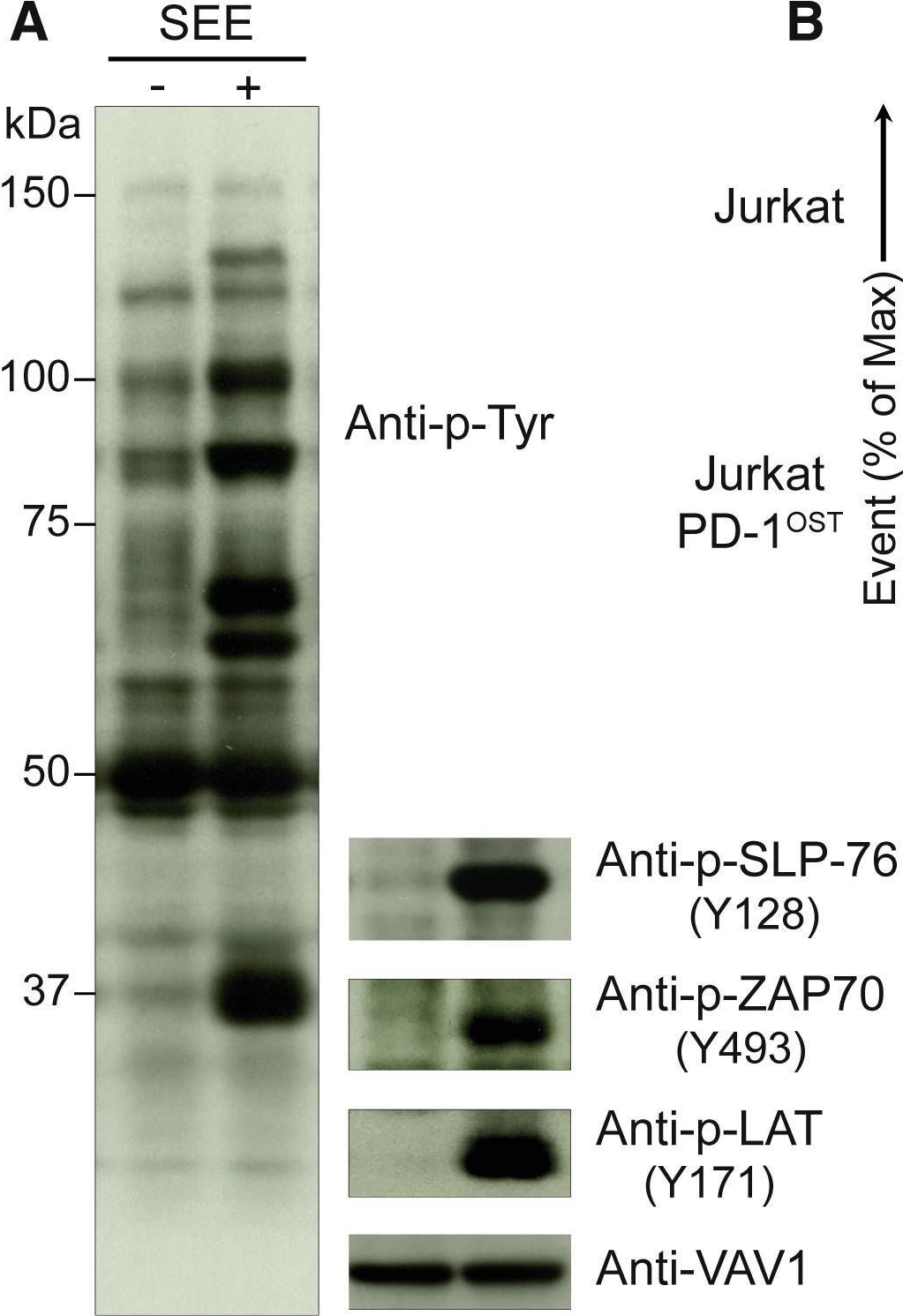
-
WB
-
Collected and cropped from Cell Reports by CiteAb, provided under a CC-BY license
Image 1 of 2