Estrogens regulate eosinophilia in asthma and other inflammatory diseases. Further, peripheral eosinophilia and tumor-associated tissue eosinophilia (TATE) predicts a better response to immune checkpoint blockade (ICB) in breast cancer. However, how and if estrogens affect eosinophil biology in tumors and how this influences ICB efficacy has not been determined. Here, we report that estrogens decrease the number of peripheral eosinophils and TATE, and this contributes to increased tumor growth in validated murine models of breast cancer and melanoma. Moreover, estrogen signaling in healthy female mice also suppressed peripheral eosinophil prevalence by decreasing the proliferation and survival of maturing eosinophils. Inhibiting estrogen receptor (ER) signaling decreased tumor growth in an eosinophil-dependent manner. Further, the efficacy of ICBs was increased when administered in combination with anti-estrogens. These findings highlight the importance of ER signaling as a regulator of eosinophil biology and TATE and highlight the potential near-term clinical application of ER modulators to increase ICB efficacy in multiple tumor types.
Product Citations: 3
Estrogen signaling suppresses tumor-associated tissue eosinophilia to promote breast tumor growth.
In Science Advances on 27 September 2024 by Artham, S., Juras, P. K., et al.
-
Cancer Research
-
Endocrinology and Physiology
In Biology on 12 June 2023 by Lund, M. C., Ellman, D. G., et al.
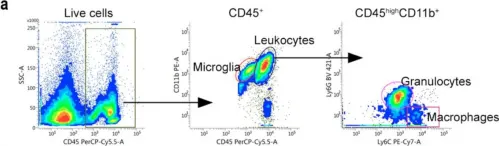
Clinical and animal model studies have implicated inflammation and glial and peripheral immune cell responses in the pathophysiology of spinal cord injury (SCI). A key player in the inflammatory response after SCI is the pleiotropic cytokine tumor necrosis factor (TNF), which exists both in both a transmembrane (tmTNF) and a soluble (solTNF) form. In the present study, we extend our previous findings of a therapeutic effect of topically blocking solTNF signaling after SCI for three consecutive days on lesion size and functional outcome to study the effect on spatio-temporal changes in the inflammatory response after SCI in mice treated with the selective solTNF inhibitor XPro1595 and compared to saline-treated mice. We found that despite comparable TNF and TNF receptor levels between XPro1595- and saline-treated mice, XPro1595 transiently decreased pro-inflammatory interleukin (IL)-1β and IL-6 levels and increased pro-regenerative IL-10 levels in the acute phase after SCI. This was complemented by a decrease in the number of infiltrated leukocytes (macrophages and neutrophils) in the lesioned area of the spinal cord and an increase in the number of microglia in the peri-lesion area 14 days after SCI, followed by a decrease in microglial activation in the peri-lesion area 21 days after SCI. This translated into increased myelin preservation and improved functional outcomes in XPro1595-treated mice 35 days after SCI. Collectively, our data suggest that selective targeting of solTNF time-dependently modulates the neuroinflammatory response by favoring a pro-regenerative environment in the lesioned spinal cord, leading to improved functional outcomes.
-
FC/FACS
-
Mus musculus (House mouse)
-
Cancer Research
-
Neuroscience
The Inflammatory Response after Moderate Contusion Spinal Cord Injury: A Time Study.
In Biology on 20 June 2022 by Lund, M. C., Ellman, D. G., et al.
Spinal cord injury (SCI) initiates detrimental cellular and molecular events that lead to acute and delayed neuroinflammation. Understanding the role of the inflammatory response in SCI requires insight into the temporal and cellular synthesis of inflammatory mediators. We subjected C57BL/6J mice to SCI and investigated inflammatory reactions. We examined activation, recruitment, and polarization of microglia and infiltrating immune cells, focusing specifically on tumor necrosis factor (TNF) and its receptors TNFR1 and TNFR2. In the acute phase, TNF expression increased in glial cells and neuron-like cells, followed by infiltrating immune cells. TNFR1 and TNFR2 levels increased in the delayed phase and were found preferentially on neurons and glial cells, respectively. The acute phase was dominated by the infiltration of granulocytes and macrophages. Microglial/macrophage expression of Arg1 increased from 1-7 days after SCI, followed by an increase in Itgam, Cx3cr1, and P2ry12, which remained elevated throughout the study. By 21 and 28 days after SCI, the lesion core was populated by galectin-3+, CD68+, and CD11b+ microglia/macrophages, surrounded by a glial scar consisting of GFAP+ astrocytes. Findings were verified in postmortem tissue from individuals with SCI. Our findings support the consensus that future neuroprotective immunotherapies should aim to selectively neutralize detrimental immune signaling while sustaining pro-regenerative processes.
-
Immunology and Microbiology
-
Neuroscience
In Biology (Basel) on 12 June 2023 by Lund, M. C., Ellman, D. G., et al.
Fig.5.A

-
FC/FACS
-
Mus musculus (House mouse)
Collected and cropped from Biology (Basel) by CiteAb, provided under a CC-BY license
Image 1 of 1