The iterative bleaching extends multiplexity (IBEX) Knowledge-Base is a central portal for researchers adopting IBEX and related 2D and 3D immunofluorescence imaging methods. The design of the Knowledge-Base is modeled after efforts in the open-source software community and includes three facets: a development platform (GitHub), static website, and service for data archiving. The Knowledge-Base facilitates the practice of open science throughout the research life cycle by providing validation data for recommended and non-recommended reagents, such as primary and secondary antibodies. In addition to reporting negative data, the Knowledge-Base empowers method adoption and evolution by providing a venue for sharing protocols, videos, datasets, software, and publications. A dedicated discussion forum fosters a sense of community among researchers while addressing questions not covered in published manuscripts. Together, scientists from around the world are advancing scientific discovery at a faster pace, reducing wasted time and effort, and instilling greater confidence in the resulting data.
Product Citations: 70
In eLife on 23 January 2026 by Yaniv, Z., Anidi, I. U., et al.
Spontaneous somatic Pten loss contributes to functional heterogeneity of T cells.
In Scientific Reports on 12 January 2026 by Chen, A. Y. & Rudensky, A. Y.
We found that healthy mice harbor T cells with heritable low Pten expression and that monoallelic Pten loss in CD4 T cells causes a bias in their differentiation toward T follicular helper cells during acute viral infection. These results suggest that somatically induced mono- or biallelic loss of expression of signaling-related genes in T cells can impact the quality of population-level T cell responses-without conspicuous pathological sequelae such as autoimmune and inflammatory manifestations or lymphomagenesis.
© 2026. The Author(s).
-
Immunology and Microbiology
In Biomaterials Science on 6 January 2026 by Ganesh, P., Prossnitz, A. N., et al.
Vaccines have been crucial to dramatic improvements in global health in recent decades, yet next-generation vaccine technologies remain out of reach for much of the world. In particular, there are two overarching global needs: (i) develop vaccines eliciting more potent and durable immune responses, especially to reduce incidence of highly communicable diseases, and (ii) enable simple and cost-efficient formulation to maximize global access. Here, we develop an injectable hydrogel depot technology prepared through physical mixing of commercially available, generally recognized as safe (GRAS) polymers that can be formulated with subunit vaccine components to improve immune responses compared to standard vaccine formulations. We demonstrate that these hydrogels are shear-thinning and rapidly self-healing, enabling facile administration via injection, and they exhibit high yield stresses required for robust in vivo depot formation post-injection. These rheological properties prolong release of subunit vaccine cargo over a period of weeks, both in vitro and in vivo, and synchronize release kinetics across physicochemically distinct vaccine components (antigens and adjuvants). When used for formulation of subunit vaccines against wild-type SARS-CoV-2 and H5N1 influenza, these hydrogels enhance potency and durability of immune responses. This vaccine formulation technology can improve protection against current and potential future pandemic pathogens.
-
FC/FACS
-
Mus musculus (House mouse)
-
Immunology and Microbiology
In Frontiers in Immunology on 5 January 2026 by Wolfe, B., Pantha, S., et al.
Obesity is increasing globally, and it negatively impacts influenza vaccine efficacy. Although sex differences in influenza vaccine responses are studied in non-obese hosts, studies investigating sex differences in influenza vaccine-induced immunity and protection during obesity are limited.
Using the C57BL/6J mouse model of high-fat diet (HFD)-induced obesity or low-fat diet controls, we investigated sex differences in influenza vaccine-induced immunity and protection during obesity. Male and female mice with or without obesity were vaccinated intramuscularly twice at a 3-week interval with an inactivated 2009 H1N1 influenza A virus (IAV) vaccine. At 35 days post-vaccination (dpv), antibody responses in plasma and B- and T-cell responses in spleen and bone marrow were quantified. At 42 dpv, mice were intranasally challenged with a drift variant of the H1N1 IAV, and disease severity was assessed by monitoring the change in body mass up to 21 days post-challenge (dpc). Subsets of mice were euthanized at 3 dpc to determine pulmonary virus replication (TCID50 assay), histopathology (H&E staining), and cytokine/chemokine responses (multiplex ELISA).
Female mice, irrespective of diet and obesity status, developed higher antibody responses and were better protected compared to males. Vaccinated males with obesity mounted the poorest antibody responses, experienced a more severe disease, were unable to clear replicating virus from the lungs effectively, and demonstrated heightened pulmonary inflammation. Despite these differences, splenic B- and T-cell frequencies were comparable, suggesting the inefficiency of B cells to produce antibodies in males but not in females with obesity.
Our findings suggest that sex differences are observed in influenza vaccine-induced immunity and protection during obesity, where males are more severely affected. These findings highlight the importance of considering biological sex and obesity status in influenza vaccine design and testing.
Copyright © 2025 Wolfe, Pantha, Vijayakumar, Budha Magar, Aboellail and Dhakal.
-
FC/FACS
-
Mus musculus (House mouse)
-
Immunology and Microbiology
Distinct components of mRNA vaccines cooperate to instruct efficient germinal center responses.
In Cell on 24 December 2025 by Castaño, D., Bettini, E., et al.
Nucleoside-modified messenger RNA (mRNA) vaccines elicit protective antibodies through their ability to promote T follicular helper (Tfh) cell differentiation. The lipid nanoparticles (LNPs) of mRNA vaccines possess inherent adjuvant activity. However, the extent to which the nucleoside-modified mRNA is sensed and contributes to Tfh cell responses remains undefined. Herein, we deconvolute the signals induced by LNPs and mRNA that instruct dendritic cells (DCs) to promote Tfh cell differentiation. We demonstrate that the mRNA drives the production of type I interferons, which act on DCs to enhance their maturation and Tfh cell differentiation, and favors plasma cells and memory B cell responses. In parallel, LNPs, which allow for mRNA uptake by DCs within the draining lymph node, also modulate Tfh cell responses by shaping the localization of CD25+ DCs. Our work unravels distinct adjuvant features of mRNA and LNPs necessary for the induction of Tfh cells, with implications for rational vaccine design.
Copyright © 2025 The Authors. Published by Elsevier Inc. All rights reserved.
-
Genetics
-
Immunology and Microbiology
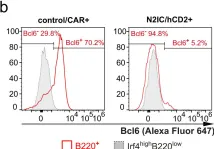
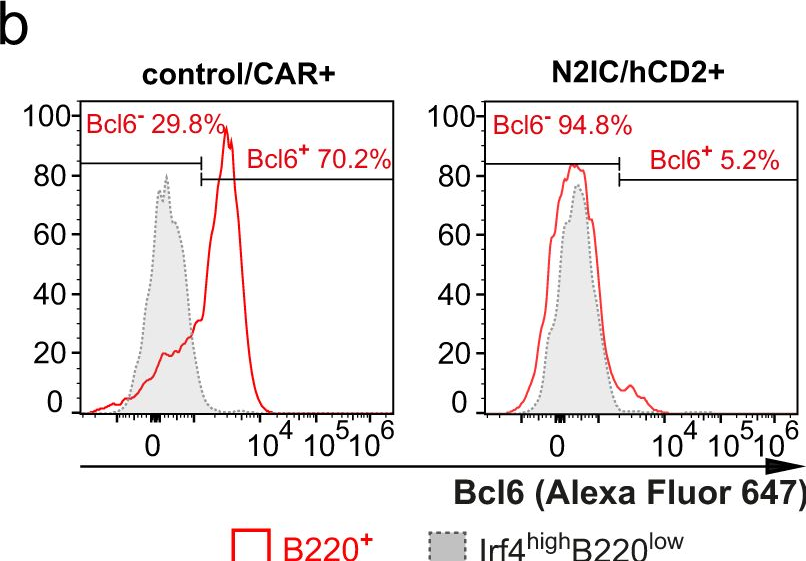
In Nat Commun on 4 March 2024 by Babushku, T., Lechner, M., et al.
Fig.6.B
-
FC/FACS
-
Mus musculus (House mouse)
Collected and cropped from Nature Communications by CiteAb, provided under a CC-BY license
Image 1 of 5
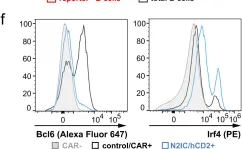
In Nat Commun on 4 March 2024 by Babushku, T., Lechner, M., et al.
Fig.4.F
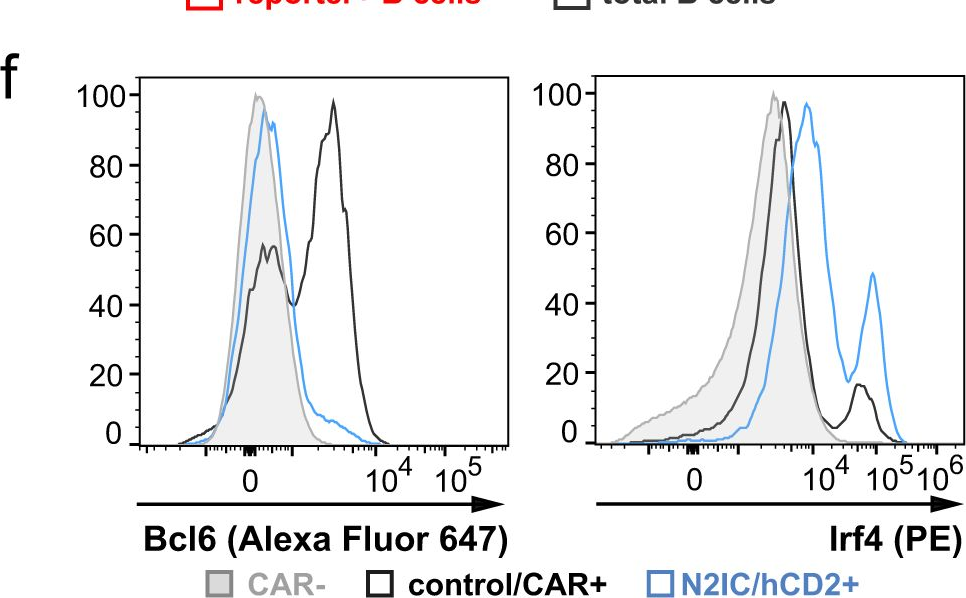
-
FC/FACS
-
Collected and cropped from Nature Communications by CiteAb, provided under a CC-BY license
Image 1 of 5
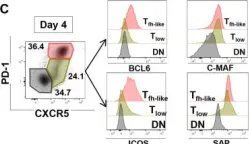
In J Cell Sci on 1 November 2022 by Korniotis, S., Saichi, M., et al.
Fig.5.A
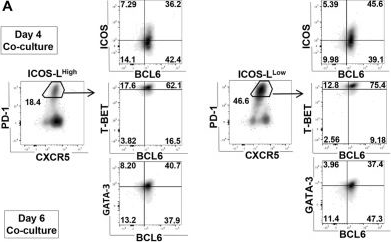
-
FC/FACS
-
Collected and cropped from Journal of Cell Science by CiteAb, provided under a CC-BY license
Image 1 of 5
In J Cell Sci on 1 November 2022 by Korniotis, S., Saichi, M., et al.
Fig.3.E
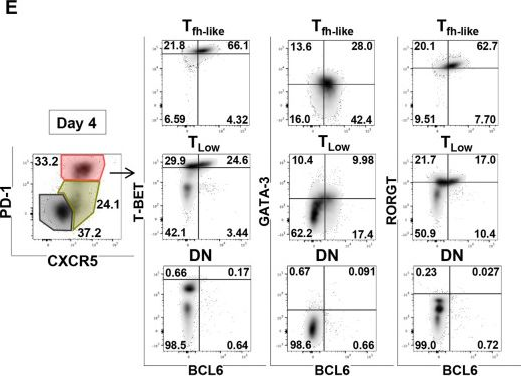
-
FC/FACS
-
Collected and cropped from Journal of Cell Science by CiteAb, provided under a CC-BY license
Image 1 of 5
In J Cell Sci on 1 November 2022 by Korniotis, S., Saichi, M., et al.
Fig.3.C
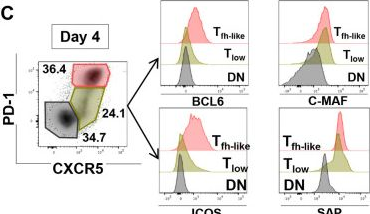
-
FC/FACS
-
Collected and cropped from Journal of Cell Science by CiteAb, provided under a CC-BY license
Image 1 of 5