The Ezrin-Radixin-Moesin (ERM) family member moesin (MSN) plays a crucial role in reversibly linking F-actin to the cell membrane. Patients carrying MSN gene mutations consistently exhibit immunodeficiencies. However, due to the scarce number of reported cases worldwide, the mechanism by which MSN mutation leads to immune function defects remains unclear. This study aims to profile the immunological features in MSN mutant patients elaborately.
In this article, we present a case study of a patient with c.511 C > T, p.Arg171Trp (p.R171W) mutation on the MSN gene. We analyzed abnormalities in peripheral immune cell subsets by quantitative analysis, morphological examination, and functional molecule assessment during various infection states. Using total internal reflection fluorescence microscopy (TIRFm), we visualized BCR clusters and F-actin dynamics in B cells, revealing valuable insights into B cell activation and the link between F-actin aggregation and BCR signaling in MSN mutant patients.
The results suggest that the MSN c.511 C > T, p.Arg171Trp (p.R171W) mutation affects the proliferation, differentiation, metabolism, and adhesion functions in peripheral immune cells, as well as the maturation process in bone marrow cells. Additionally, we elucidate the impact of MSN mutation on B cell and T cell metabolism and propose a potential diagnostic indicator for patients with MSN gene mutations.
Our findings support the diagnosis of primary immunodeficiency and provide detailed insights into changes occurring in immune cells, especially B cells. Overall, our study adds to the diagnosis and pathogenesis of X-linked moesin-associated immunodeficiency (X-MAID).
© 2025. The Author(s).
Product Citations: 20
In Journal of Clinical Immunology on 11 August 2025 by Liu, Q., Zhang, A., et al.
-
FC/FACS
-
Homo sapiens (Human)
-
Immunology and Microbiology
In Nature Immunology on 1 August 2025 by Beumer, N., Imbusch, C. D., et al.
CD4+ regulatory T (Treg) cells in tissues play crucial immunoregulatory and regenerative roles. Despite their importance, the epigenetics and differentiation of human tissue Treg cells are incompletely understood. Here, we performed genome-wide DNA methylation analysis of human Treg cells from skin and blood and integrated these data into a multiomic framework, including chromatin accessibility and gene expression. This analysis identified programs that governed the tissue adaptation of skin Treg cells. We found that subfamilies of transposable elements represented a major constituent of the hypomethylated landscape in tissue Treg cells. Based on T cell antigen receptor sequence and DNA hypomethylation homologies, our data indicate that blood CCR8+ Treg cells contain recirculating human skin Treg cells. Conversely, differences in chromatin accessibility and gene expression suggest a certain reversal of the tissue adaptation program during recirculation. Our findings provide insights into the biology of human tissue Treg cells, which may help harness these cells for therapeutic purposes.
© 2025. The Author(s).
-
Cardiovascular biology
-
Genetics
-
Immunology and Microbiology
The Human Bone Marrow May Offer an IL-15-Dependent Survival Niche for EOMES+ Tr1-Like Cells.
In European Journal of Immunology on 1 May 2025 by Pulvirenti, N., Vasco, C., et al.
Maintenance of memory T-cells in the bone marrow and systemically depends on the homeostatic cytokines IL-7 and IL-15. An immunological memory may also exist for regulatory T-cells. EOMES+type-1 regulatory (Tr1)-like cells have a rapid in vivo turnover, but whether they are short-lived effector cells or are maintained long-term has not been investigated. EOMES+Tr1-like cells expressing GzmK were enriched among CD69+Ki67-T-cells in the bone marrow of healthy donors, suggesting that they became quiescent and bone marrow-resident. Conversely, CD4+GzmB+ effector T-cells were excluded from the bone marrow-resident fraction. The dichotomy between GzmK+ and GzmB+T-cells was observed both in healthy individuals and in multiple sclerosis patients, and also among CD8+T-cells. Intriguingly, bone marrow-resident CD4+ memory T-cells expressed increased levels of IL-7Rα, while EOMES+Tr1-like cells were consistently IL-7Rαlo. However, EOMES+Tr1-like cells expressed the IL-2/15Rβ chain, and the latter was induced upon forced expression of EOMES in primary human CD4+ T-cells. Finally, IL-15 rescued EOMES+Tr1-enriched populations from death by neglect but was not required for CD4+ memory T-cell survival. These findings suggest that the bone marrow may provide a survival niche for EOMES+Tr1-like cells. The different IL-7 and IL-15 receptor expression patterns of CD4+ memory T-cells and EOMES+Tr1-like cells suggest furthermore that they compete for different homeostatic niches.
© 2025 The Author(s). European Journal of Immunology published by Wiley‐VCH GmbH.
-
Immunology and Microbiology
CXCR4-enriched T regulatory cells preferentially home to bone marrow and resolve inflammation.
In IScience on 20 September 2024 by Huang, M., Ke, Z., et al.
CXCR4 cell surface expression is critical for the homing of T regulatory (Treg) cells to the bone marrow (BM). We hypothesize that CXCR4 enrichment on Tregs cell surface may abbreviate their transit time to reach BM. Umbilical cord-blood CD25+ Tregs underwent CXCR4 dual enrichment and ex vivo expansion using the CRANE process to generate CXCR4-enriched Tregs (TregCXCR4) cells, which showed a faster migration across the Transwell membrane toward CXCL12/stromal cell-derived factor 1α (SDF1α) at 15, 30, and 60 min, when compared to unmanipulated Tregcontrol cells (p < 0.0001). TregCXCR4 exhibited preferential homing to BM in vivo at 12 and 24 h. Metacluster analysis of BM showed a decrease in CD8+ and an increase in CD39 and CD73 and CXCR5 when compared to Tregcontrol. TregCXCR4 decreased plasma TGF-β1/β2 and IFN-γ levels. When compared to control, TregCXCR4 cells decreased in CD8+ T cell, IFN-γ, and TNF-α expression in BM. We conclude that TregCXCR4 show enhanced migration toward CXCL12/SDF1α and a preferential homing to BM resulting in resolution of inflammation.
© 2024 The Author(s).
-
Immunology and Microbiology
In Advanced Healthcare Materials on 1 June 2024 by Lin, H. K., Uricoli, B., et al.
Despite the remarkable clinical efficacy of chimeric antigen receptor (CAR) T cells in hematological malignancies, only a subset of patients achieves a durable complete response (dCR). DCR has been correlated with CAR T cell products enriched with T cells memory phenotypes. Therefore, reagents that consistently promote memory phenotypes during the manufacturing of CAR T cells have the potential to significantly improve clinical outcomes. A novel modular multi-cytokine particle (MCP) platform is developed that combines the signals necessary for activation, costimulation, and cytokine support into a single "all-in-one" stimulation reagent for CAR T cell manufacturing. This platform allows for the assembly and screening of compositionally diverse MCP libraries to identify formulations tailored to promote specific phenotypes with a high degree of flexibility. The approach is leveraged to identify unique MCP formulations that manufacture CAR T cell products from diffuse large B cell patients with increased proportions of memory-like phenotypes MCP-manufactured CAR T cells demonstrate superior anti-tumor efficacy in mouse models of lymphoma and ovarian cancer through enhanced persistence. These findings serve as a proof-of-principle of the powerful utility of the MCP platform to identify "all-in-one" stimulation reagents that can improve the effectiveness of cell therapy products through optimal manufacturing.
© 2024 Wiley‐VCH GmbH.
-
Cancer Research
-
Immunology and Microbiology
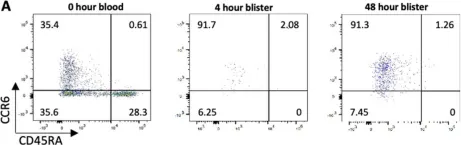
In Sci Rep on 7 March 2022 by Szylar, G., Wysoczanski, R., et al.
Fig.4.A
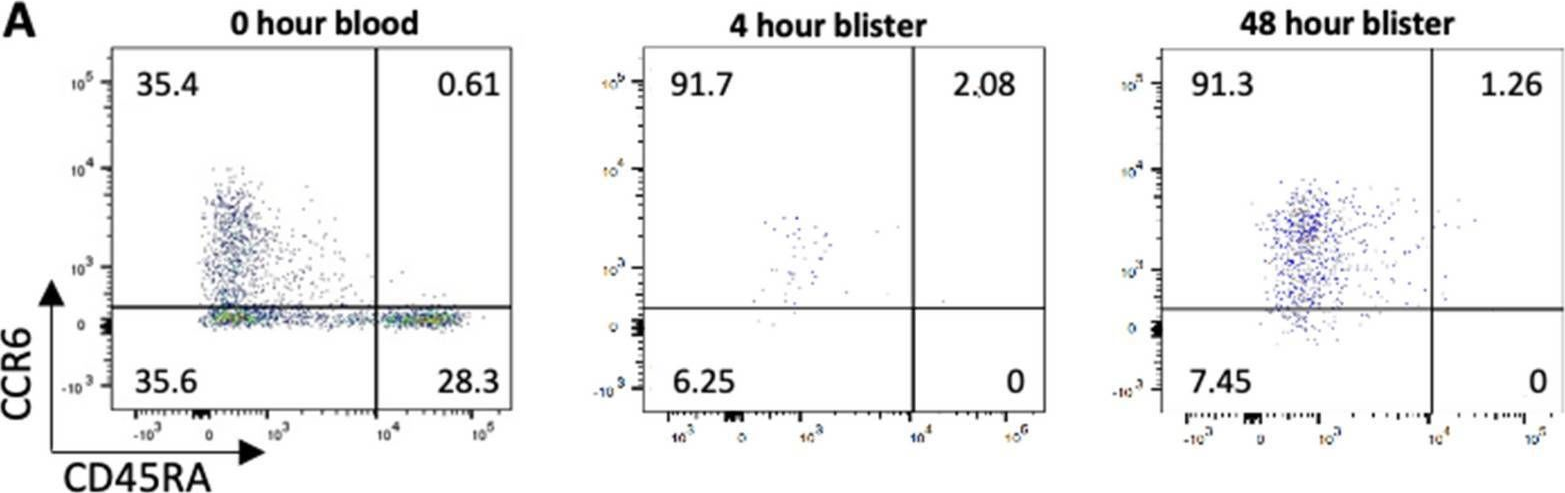
-
FC/FACS
-
Collected and cropped from Scientific Reports by CiteAb, provided under a CC-BY license
Image 1 of 1