Biologics such as proteins, peptides, and oligonucleotides are powerful ligands to modulate challenging drug targets that lack readily accessible and "ligandable" pockets. However, the limited membrane permeance of biologics severely restricts their intracellular applications. Moreover, different cell types may exhibit varying levels of impermeability, and some delivery vehicles might be more sensitive to this variance. Erythroid lineage cells are especially challenging to deliver cargo to because of their unique cytoskeleton and the absence of endocytosis in mature erythrocytes. We recently employed a cell permeant miniature protein to deliver bioPROTACs to human umbilical cord blood derived erythroid progenitor cells (HUDEP-2) and primary hematopoietic stem (CD34+) cells (Shen et al., ACS Cent. Sci.2022, 8, 1695-1703). While successful, the low efficiency of delivery and lack of cell-type specificity limit use of bioPROTACs in vivo. In this work, we thoroughly evaluated the performance of various recently reported cell penetrating peptides (CPPs), CPP additives, bacterial toxins, and contractile injection systems for their ability to deliver cargo to erythroid precursor cells. We also explored how targeting receptors enriched on the erythroid cell surface might improve the efficiencies and specificities of these delivery vehicles. Our results reveal that certain vehicles exhibit improved efficiencies when directed to cell surface receptors while others do not benefit from this targeting strategy. Together, these findings advance our understanding of protein delivery to challenging cell types and illustrate some of the intricacies of cell-surface receptor targeting.
© 2025 The Authors. Published by American Chemical Society.
Product Citations: 45
Engineering Cell-Specific Protein Delivery Vehicles for Erythroid Lineage Cells.
In ACS Bio Med Chem Au on 16 April 2025 by Setegne, M. T., Cabral, A. T., et al.
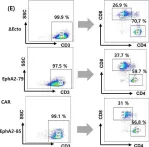
Developing CAR-T/NK cells that target EphA2 for non-small cell lung cancer treatment.
In Frontiers in Immunology on 4 April 2025 by Kim, S. M., Lee, S. Y., et al.
Chimeric antigen receptor (CAR) immunotherapy has revolutionized anticancer therapy, as it accurately targets cancer cells by recognizing specific antigens expressed in cancer cells. This innovative therapeutic strategy has attracted considerable attention. However, few therapeutics are available for treating non-small cell lung cancer (NSCLC), which accounts for most lung cancer cases and is one of the deadliest cancers with low survival rates.
In this study, we developed a new antibody targeting erythropoietin-producing hepatocellular carcinoma A2 (EphA2), which is highly expressed in NSCLC, and established CAR-T/ natural killer (NK) immune cells to verify its potential for immune cell therapy. The killing capacity, cytokine secretion and solid tumor growth inhibition of EphA2 CAR-T/NK cells were compared to normal T/NK cells.
EphA2 CAR-T cells demonstrated superior killing capacity, enhanced cytokine secretion, and significant solid tumor growth inhibition. Additionally, they exhibited improved tumor infiltration in lung cancer models compared to normal T cells. The anticancer efficacy of the developed EphA2 CAR-NK cells was also confirmed, showcasing their potential as robust candidates for immune cell therapy.
The findings of this study highlight the potential of CAR-T/NK cell therapy targeting EphA2 as an effective treatment for lung cancer, particularly NSCLC with high EphA2 expression. By leveraging the specific targeting capabilities of CAR-T cells and the unique properties of CAR-NK cells, this approach provides a promising therapeutic strategy to address the unmet needs in NSCLC treatment.
Copyright © 2025 Kim, Lee, Kim, Bae, Hong, Jo, Kim, Chung and Kim.
-
FC/FACS
-
Homo sapiens (Human)
-
Cancer Research
-
Immunology and Microbiology
In International Journal of Molecular Sciences on 26 June 2024 by Magne, B., Ferland, K., et al.
The immunogenicity of allogeneic skin fibroblasts in transplantation has been controversial. Whether this controversy comes from a natural heterogeneity among fibroblast subsets or species-specific differences between human and mouse remains to be addressed. In this study, we sought to investigate whether fibroblasts derived from either adult or neonatal human skin tissues could induce different immune responses toward phagocytosis and T cell activation using in vitro co-culture models. Our results indicate that both phagocytosis and T cell proliferation are reduced in the presence of neonatal skin fibroblasts compared to adult skin fibroblasts. We also show that neonatal skin fibroblasts secrete paracrine factors that are responsible for reduced T cell proliferation. In addition, we show that neonatal skin fibroblasts express less class II human leukocyte antigen (HLA) molecules than adult skin fibroblasts after interferon gamma priming, which might also contribute to reduced T cell proliferation. In conclusion, this study supports the use of allogeneic neonatal skin fibroblasts as a readily available cell source for tissue production and transplantation to treat patients with severe injuries.
-
Homo sapiens (Human)
-
Immunology and Microbiology
CD8+ T-cell responses towards conserved influenza B virus epitopes across anatomical sites and age.
In Nature Communications on 29 April 2024 by Menon, T., Illing, P. T., et al.
Influenza B viruses (IBVs) cause substantive morbidity and mortality, and yet immunity towards IBVs remains understudied. CD8+ T-cells provide broadly cross-reactive immunity and alleviate disease severity by recognizing conserved epitopes. Despite the IBV burden, only 18 IBV-specific T-cell epitopes restricted by 5 HLAs have been identified currently. A broader array of conserved IBV T-cell epitopes is needed to develop effective cross-reactive T-cell based IBV vaccines. Here we identify 9 highly conserved IBV CD8+ T-cell epitopes restricted to HLA-B*07:02, HLA-B*08:01 and HLA-B*35:01. Memory IBV-specific tetramer+CD8+ T-cells are present within blood and tissues. Frequencies of IBV-specific CD8+ T-cells decline with age, but maintain a central memory phenotype. HLA-B*07:02 and HLA-B*08:01-restricted NP30-38 epitope-specific T-cells have distinct T-cell receptor repertoires. We provide structural basis for the IBV HLA-B*07:02-restricted NS1196-206 (11-mer) and HLA-B*07:02-restricted NP30-38 epitope presentation. Our study increases the number of IBV CD8+ T-cell epitopes, and defines IBV-specific CD8+ T-cells at cellular and molecular levels, across tissues and age.
© 2024. The Author(s).
-
Immunology and Microbiology
synNotch-programmed iPSC-derived NK cells usurp TIGIT and CD73 activities for glioblastoma therapy.
In Nature Communications on 1 March 2024 by Lupo, K. B., Yao, X., et al.
Severe heterogeneity within glioblastoma has spurred the notion that disrupting the interplay between multiple elements on immunosuppression is at the core of meaningful anti-tumor responses. T cell immunoreceptor with Ig and ITIM domains (TIGIT) and its glioblastoma-associated antigen, CD155, form a highly immunosuppressive axis in glioblastoma and other solid tumors, yet targeting of TIGIT, a functionally heterogeneous receptor on tumor-infiltrating immune cells, has largely been ineffective as monotherapy, suggesting that disruption of its inhibitory network might be necessary for measurable responses. It is within this context that we show that the usurpation of the TIGIT - CD155 axis via engineered synNotch-mediated activation of induced pluripotent stem cell-derived natural killer (NK) cells promotes transcription factor-mediated activation of a downstream signaling cascade that results in the controlled, localized blockade of CD73 to disrupt purinergic activity otherwise resulting in the production and accumulation of immunosuppressive extracellular adenosine. Such "decoy" receptor engages CD155 binding to TIGIT, but tilts inhibitory TIGIT/CD155 interactions toward activation via downstream synNotch signaling. Usurping activities of TIGIT and CD73 promotes the function of adoptively transferred NK cells into intracranial patient-derived models of glioblastoma and enhances their natural cytolytic functions against this tumor to result in complete tumor eradication. In addition, targeting both receptors, in turn, reprograms the glioblastoma microenvironment via the recruitment of T cells and the downregulation of M2 macrophages. This study demonstrates that TIGIT/CD155 and CD73 are targetable receptor partners in glioblastoma. Our data show that synNotch-engineered pluripotent stem cell-derived NK cells are not only effective mediators of anti-glioblastoma responses within the setting of CD73 and TIGIT/CD155 co-targeting, but represent a powerful allogeneic treatment option for this tumor.
© 2024. The Author(s).
-
Stem Cells and Developmental Biology
In Front Immunol on 4 April 2025 by Kim, S. M., Lee, S. Y., et al.
Fig.1.E

-
FC/FACS
-
Collected and cropped from Front Immunol by CiteAb, provided under a CC-BY license
Image 1 of 2
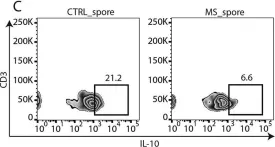
In mSystems on 13 November 2018 by Cekanaviciute, E., Pröbstel, A. K., et al.
Fig.2.C

-
FC/FACS
-
Homo sapiens (Human)
Collected and cropped from mSystems by CiteAb, provided under a CC-BY license
Image 1 of 2