Areca nut is the fourth most addictive substance worldwide and is a known Group 1 carcinogen. Chronic areca nut chewing has been strongly associated with the development of oral squamous cell carcinoma (OSCC), although the underlying carcinogenic mechanisms remain unclear.
Employing spatial multiomics technology, we recently mapped the immune microenvironment of areca nut-associated OSCC and identified a marked upregulation of the immune checkpoint molecule CD155 in these tumors. Further quantitative reverse transcription-PCR, western blotting, immunofluorescence, animal experiments, etc, explored and verified the reasons for the upregulation of CD155 caused by arecoline and the results of immune escape in OSCC.
The investigation revealed that arecoline, a bioactive component of areca nut, induces CD155 expression in OSCC cells. Through interaction with its receptor, TIGIT, CD155 suppresses CD8+ T cell function and activity, facilitating immune evasion in OSCC. Mechanistically, arecoline upregulates CD155 via activation of muscarinic acetylcholine receptors on the surface of OSCC cells, leading to RhoA-mediated inhibition of YAP phosphorylation. This promotes YAP nuclear translocation and phase separation, driving CD155 transcription. In both in vitro and in vivo models, blockade of the CD155-TIGIT signaling axis significantly enhanced the efficacy of immunotherapy in OSCC.
This study unveils a novel mechanism by which arecoline promotes immune evasion in OSCC and highlights promising immune therapeutic targets and strategies for the treatment of areca nut-associated OSCC.
© Author(s) (or their employer(s)) 2025. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ Group.
Product Citations: 24
Arecoline upregulates CD155 expression to facilitate immune evasion in oral squamous cell carcinoma.
In Journal for Immunotherapy of Cancer on 9 December 2025 by Wang, Q., Meng, Y., et al.
-
Cancer Research
-
Immunology and Microbiology
In The Journal of Clinical Investigation on 2 September 2025 by Pellegrini, J. M., Keriel, A., et al.
-
Immunology and Microbiology
In IScience on 16 May 2025 by Wang, S., Sun, X., et al.
Wilson disease (WD) is caused by mutations of the copper-transporting gene, ATP7B, leading to abnormal copper metabolism. A better characterization of WD is essential in understanding the effects of excess copper and how it disrupts immune regulation and hematopoietic development. Furthermore, the exploration of the relationship between copper-mediated proliferation or cuproptosis and immune regulation is critical for developing new immune therapies. Therefore, we performed single-cell RNA sequencing (scRNA-seq) on peripheral blood mononuclear cells (PBMCs) to develop an atlas of the immune landscape. Cells were clustered into several immune subsets, and cuproptosis-associated genes were assessed. Differential expression analysis was performed to identify WD-specific signatures by comparing transcriptome profiles of patients with WD with HDs. Excess copper impaired immune homeostasis and hematopoietic development. Then, we developed a map of the immune landscape of patients with WD. Excess copper is involved in the metabolic reprogramming of immune cells, such as glycolysis in CD14+ monocytes. We found that the antigen processing-related pathway is dysregulated in immune cells of patients with WD. Our study revealed that abnormal copper concentration influences the expression of HLA-I and HLA-II molecules. It is noteworthy that a high concentration of intracellular copper differs significantly from the high concentration of extracellular copper. We have also identified a gene set of neurologic abnormalities, which were dysregulated in PBMCs of patients with WD. We also observed abnormal expression of cuproptosis-associated genes in proliferating or malignant cells, providing new insights into the application of cuproptosis in cancer treatment.
© 2025 The Author(s).
-
Immunology and Microbiology
Dual function of DOT1L suppresses tumor intrinsic immunogenicity in hepatocellular carcinoma
Preprint on BioRxiv : the Preprint Server for Biology on 2 May 2025 by Xu, S., Gong, R., et al.
Immune checkpoint inhibitor (ICB) therapy for many cancers remains limited in patients’ overall response rate. Discovery and development of more effective combinatorial approaches is urgent. Here, through CRISPR/Cas9 genetic screens, we identify DOT1L as a versatile epigenetic factor that functions to suppress tumor-intrinsic immunity through a dual mechanism. Depletion of DOT1L induces the expression of transposable elements and subsequent type I interferon (IFN) response, and meanwhile lowers ZEB1 levels to further unleash the expression of immune-related genes. In turn, we demonstrate that DOT1L loss or treatment with the clinical stage inhibitor EPZ-5676 sensitizes tumors to ICB with increased immune infiltration in mice. More importantly, EPZ-5676 treatment alone is sufficient to enhance antitumor immunity in humanized mice. TCGA data analysis reveals an inverse correlation between DOT1L expression and IFN signatures across multiple cancer types. These findings provide a rationale for targeting DOT1L to improve tumor immunogenicity and overcome immunotherapy resistance. One Sentence Summary CRISPR genetic screens identified DOT1L as a potential suppressor of tumor intrinsic immunogenicity
-
FC/FACS
-
Mus musculus (House mouse)
-
Cancer Research
-
Immunology and Microbiology
In The Journal of Clinical Investigation on 15 April 2025 by Pellegrini, J. M., Keriel, A., et al.
Plasmacytoid dendritic cells (pDCs), professional type I IFN-producing cells, have been implicated in host responses against bacterial infections. However, their role in host defense is debated, and the operating molecular mechanisms are unknown. Certain signaling lymphocyte activation molecule family (SLAMF) members act as microbial sensors and modulate immune functions in response to infection. Here, human blood transcriptomic analyses reveal the involvement of SLAMF7 and SLAMF8 in many infectious diseases, with elevated levels associated with type I IFN responses in salmonellosis and brucellosis patients. We further identify SLAMF7 and SLAMF8 as key regulators of human pDC function. They activate pDC maturation and cytokine production during infection with bacteria that induce acute (Salmonella) or chronic (Brucella) inflammation. SLAMF7 and SLAMF8 signal through NF-κB, IRF7, and STAT-1, and limit mitochondrial ROS accumulation upon Salmonella infection. Remarkably, this SLAMF7/8-dependent control of mitochondrial ROS levels favors bacterial persistence and NF-κB activation. Overall, our results unravel essential shared multifaceted roles of SLAMF7 and SLAMF8 in finely tuning human pDC responses to intracellular bacterial infections with potential for future diagnostic and therapeutic applications.
-
Immunology and Microbiology
In Front Immunol on 14 August 2019 by Chen, E. W., Tay, N. Q., et al.
Fig.5.A
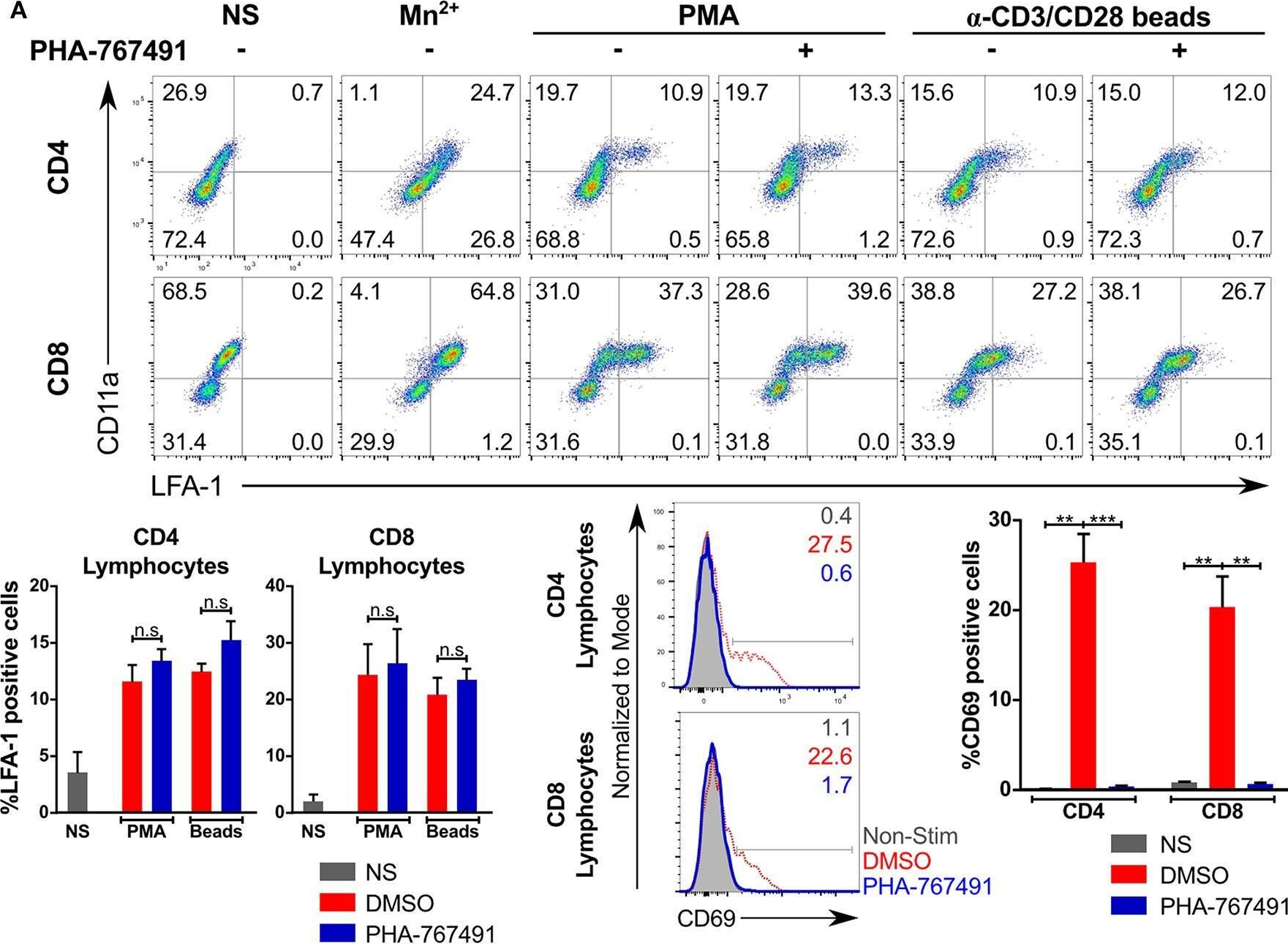
-
FC/FACS
-
Collected and cropped from Frontiers in Immunology by CiteAb, provided under a CC-BY license
Image 1 of 1