Adoptive immunotherapy with third-party virus-specific T lymphocytes (VSTs) is effective against refractory viral infections. However, its long-term efficacy and persistence must be enhanced. T memory stem cells (TSCMs) with superior self-renewal and multilineage differentiation potential may enhance VSTs durability, although their antiviral capacity is underexplored. Cytomegalovirus (CMV)-and Epstein-Barr virus (EBV)-specific T cells are enriched with CD8⁺ TSCM through cytokine and peptide stimulation. Comprehensive preclinical evaluations show that purified TSCM-VSTs exhibit reduced exhaustion, enhanced expansion, and stronger antiviral activity than central or effector memory VSTs (TCM or TEM). Transcriptomic and epigenetic analyses show significant enrichment of the MAPK and Wnt signaling pathways, consistent with stem-like characteristics. In a murine model, CD8⁺ TSCM VSTs provide more effective protection against Raji-pp65 tumors than TCM or TEM VSTs. In a phase I clinical trial, 10 patients with refractory CMV or EBV infections post-transplant who received third-party, off-the-shelf TSCM-enriched VSTs show a 100% overall response rate and 70% complete response, with persistence up to 12 weeks and no severe adverse events. These findings support TSCM-enriched VSTs as a potent, scalable antiviral immunotherapy and highlight TSCM proportion as a critical determinant of VSTs efficacy.
© 2025 The Author(s). Advanced Science published by Wiley‐VCH GmbH.
Product Citations: 65
In Advanced Science (Weinheim, Baden-Wurttemberg, Germany) on 1 February 2026 by Cao, X. H., Pei, X. Y., et al.
-
Immunology and Microbiology
-
Stem Cells and Developmental Biology
In Archivum Immunologiae et Therapiae Experimentalis on 1 January 2026 by Jablonska, E., Iwaniuk, A., et al.
In light of the increasing incidence of head and neck cancer and the widespread occurrence of methylparabens (MeP) in the human environment, which mimic the action of endogenous estrogens, we investigated the effect of this compound on head and neck squamous cell carcinoma (HNSCC) cells. We took into account autophagy and apoptosis, as well as the ability of HNSCC cell lines FaDu and Detroit 562 to proliferate, and the effect of flavonoids on the determined parameters. The obtained results revealed that MeP inhibits autophagy, indicated by downregulation of autophagy-related proteins such as beclin-1, LC3β, and APG5 expression in both cancer cell lines. Moreover, MeP, by changing the expression of mitochondrial proteins Bcl-2 and Bax, may be responsible for the reduction of apoptosis and the increased proliferation of cancer cells associated with high expression of caspase-3. The applied flavonoids restored the values of the tested parameters to the level observed in cells not exposed to MeP. In conclusion, our studies have shown for the first time that MeP, in addition to well-documented exposure factors, may facilitate the development of HNSCCs. We suggest that the adverse effects of MeP can be reduced by the use of flavonoids, which may lead to the inhibition of HNSCC cells growth.
© 2026 Ewa Jablonska et al., published by Hirszfeld Institute of Immunology and Experimental Therapy.
-
Cancer Research
-
Cell Biology
Single-Cell RNA Sequencing of Thyroid Tissues Reveals Pathogenesis of Graves' Disease.
In Advanced Science (Weinheim, Baden-Wurttemberg, Germany) on 1 January 2026 by Zhou, X., Cong, J., et al.
Graves' disease (GD) is an autoimmune disorder primarily targeting the thyroid tissue. While major histocompatibility complex (MHC)-dependent B cell activation and thyroid-stimulating hormone receptor (TSHR) autoantibody production are central to GD, the intrathyroidal immune landscape remains largely unexplored. Through single-cell RNA sequencing (scRNA-seq), this work constructed a comprehensive immune cell atlas, revealing dominant IFN-γ-secreting CD4+ T cells, expanded T peripheral helper (Tph) cells, CD11c+ atypical B cells, and CD8+ effector T cells. Notably, stress-surveilling γδ T/NK cells are enriched in GD. Thyroid follicular cells (TFCs) in GD exhibited a stressed phenotype, and in vitro functional assays showed that they promote γδ T cell activation and proliferation. γδ T cells may recruit conventional type 1 dendritic cells (cDC1) via XCL1/XCL2, suggesting a potential link to adaptive immune reorganization. These findings suggest an additional MHC-independent pathway linking TFC stress to autoimmune activation via γδ T cells in GD pathogenesis.
© 2025 The Author(s). Advanced Science published by Wiley‐VCH GmbH.
-
Endocrinology and Physiology
-
Genetics
CD8+HLA-DR+CD27+ T cells define a population of naturally occurring regulatory precursors in humans.
In Science Advances on 12 September 2025 by Guo, H., Wang, B., et al.
Regulatory Treg cells are essential for immune homeostasis. While CD4 Treg cells are well characterized, CD8 Treg cells remain less understood and are primarily observed in pathological or experimental contexts. Here, we identify a naturally occurring CD8 regulatory precursor Trp cell at the steady state, defined by a CD8+HLA-DR+CD27+ phenotype and a transcriptome resembling CD4 Treg cells. Multiomics analyses reveal activation of TCF7 and costimulatory and co-inhibitory molecules in CD8 Trp cells. CD8 Trp cells suppress T cell expansion in vitro and in vivo. In a humanized xenogeneic graft-versus-host disease (GVHD) model, they dampen T cell activation, alleviate GVHD pathology, and prolong survival without impairing antileukemia activity. Mechanistically, CD8 Trp cells promote immune regulation by inducing FOXP3 expression in both CD4 Treg cells and themselves. Their expansion also correlates with immune homeostasis restoration post-allogeneic stem cell transplantation. These findings establish CD8 Trp cells as a naturally occurring regulatory precursor population that promotes transplantation tolerance.
-
Immunology and Microbiology
In Cells on 9 September 2025 by Zhang, J., Wei, L., et al.
Donor-specific anti-HLA antibodies (DSAs) bind to donor vascular endothelial cells and mediate allograft rejection (AMR), but a clinical challenge for which targeted therapeutic options remain limited. We used a multiplexed single-antigen bead (SAB) assay to detect anti-human leukocyte antigen (HLA) antibodies. Based on the antigens which patient's antibodies aganist to, we developed bivalent HLA-Fc fusion proteins composed of HLA-derived antigenic domains and human IgG1-Fc effector regions (rA24-Fc and rB13-Fc). Specific binding and functional activity of the HLA-Fc proteins were further validated by flow cytometry, ELISA, complement-dependent cytotoxicity (CDC) and antibody-dependent cellular cytotoxicity (ADCC) assays. Our findings demonstrate that the fusion proteins rA24-Fc and rB13-Fc significantly reduced HLA-specific antibody reactivity in vitro. Notably, rA24-Fc and rB13-Fc selectively bound to B-cell hybridomas (e.g., mouse W6/32 cells) expressing membrane immunoglobulins (BCR) which bound to the most HLA class I antigens. Importantly, rA24-Fc and rB13-Fc elicited antigen-specific, Fc-dependent elimination of the specific B-cell hybridomas. This study highlights HLA-Fc fusion proteins as a promising therapeutic strategy for the antigen-specific suppression of depletion of alloreactive B cells through dual cytotoxic mechanisms. This precision targeted to BCR of B cells approach is used to apply to the treatment of antibody-mediated rejection.
-
FC/FACS
-
Cell Biology
-
Immunology and Microbiology
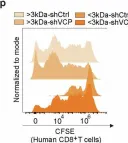
In Signal Transduct Target Ther on 24 January 2025 by Cheng, C., Zha, Q., et al.
Fig.2.P
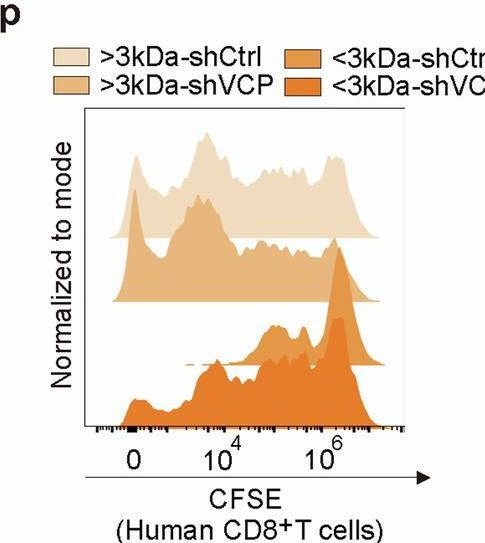
Collected and cropped from Signal Transduction and Targeted Therapy by CiteAb, provided under a CC-BY license
Image 1 of 3
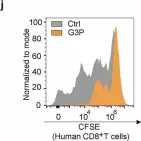
In Signal Transduct Target Ther on 24 January 2025 by Cheng, C., Zha, Q., et al.
Fig.3.I
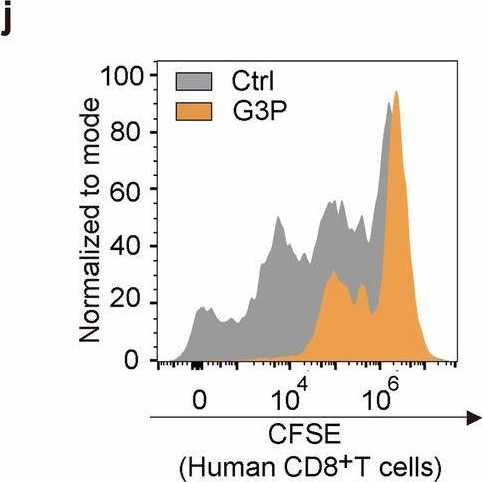
-
FC/FACS
-
Collected and cropped from Signal Transduction and Targeted Therapy by CiteAb, provided under a CC-BY license
Image 1 of 3
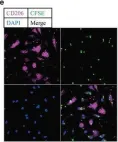
In Adv Sci (Weinh) on 1 July 2023 by Lin, L., Wang, M., et al.
Fig.6.E
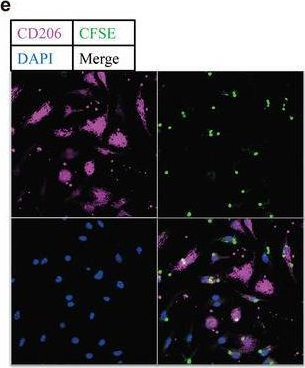
-
ICC-IF
-
Collected and cropped from Advanced Science (Weinheim, Baden-Wurttemberg, Germany) by CiteAb, provided under a CC-BY license
Image 1 of 3