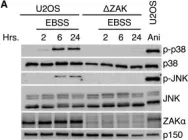
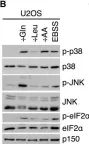
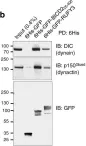
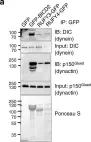
To ensure the correct euploid state of embryos, it is essential that vertebrate oocytes await fertilization arrested at metaphase of meiosis II. This MII arrest is mediated by XErp1/Emi2, which inhibits the ubiquitin ligase APC/C (anaphase-promoting complex/cyclosome). Cyclin B3 in complex with Cdk1 (cyclin-dependent kinase 1) is essential to prevent an untimely arrest of vertebrate oocytes in meiosis I by targeting XErp1/Emi2 for degradation. Yet, the molecular mechanism of XErp1/Emi2 degradation in MI is not well understood. Here, by combining TRIM-Away in oocytes with egg extract and in vitro studies, we demonstrate that a hitherto unknown phosphate-binding pocket in cyclin B3 is essential for efficient XErp1/Emi2 degradation in meiosis I. This pocket enables Cdk1/cyclin B3 to bind pre-phosphorylated XErp1/Emi2 facilitating further phosphorylation events, which ultimately target XErp1/Emi2 for degradation in a Plk1- (Polo-like kinase 1) dependent manner. Key elements of this degradative mechanism are conserved in frog and mouse. Our studies identify a novel, evolutionarily conserved determinant of Cdk/cyclin substrate specificity essential to prevent an untimely oocyte arrest at meiosis I with catastrophic consequences upon fertilization.
© 2025. The Author(s).
Product Citations: 117
A phosphate-binding pocket in cyclin B3 is essential for XErp1/Emi2 degradation in meiosis I.
In EMBO Reports on 1 February 2025 by Schunk, R., Halder, M., et al.
-
Cell Biology
Nesprin-2 coordinates opposing microtubule motors during nuclear migration in neurons.
In The Journal of Cell Biology on 4 November 2024 by Zhou, C., Wu, Y. K., et al.
Nuclear migration is critical for the proper positioning of neurons in the developing brain. It is known that bidirectional microtubule motors are required for nuclear transport, yet the mechanism of the coordination of opposing motors is still under debate. Using mouse cerebellar granule cells, we demonstrate that Nesprin-2 serves as a nucleus-motor adaptor, coordinating the interplay of kinesin-1 and dynein. Nesprin-2 recruits dynein-dynactin-BicD2 independently of the nearby kinesin-binding LEWD motif. Both motor binding sites are required to rescue nuclear migration defects caused by the loss of function of Nesprin-2. In an intracellular cargo transport assay, the Nesprin-2 fragment encompassing the motor binding sites generates persistent movements toward both microtubule minus and plus ends. Nesprin-2 drives bidirectional cargo movements over a prolonged period along perinuclear microtubules, which advance during the migration of neurons. We propose that Nesprin-2 keeps the nucleus mobile by coordinating opposing motors, enabling continuous nuclear transport along advancing microtubules in migrating cells.
© 2024 Zhou et al.
-
WB
-
Cell Biology
-
Neuroscience
A molecular switch for stress-induced activation of retrograde mitochondrial transport
Preprint on BioRxiv : the Preprint Server for Biology on 14 September 2024 by Gladkova, C., Paez-Segala, M. G., et al.
The cellular distribution of mitochondria in response to stress and local energy needs is governed by the relative activities of kinesin and dynein. The mechanism for switching between these two opposite polarity microtubule motors remains unknown. Here, we coupled a novel cellular synthetic cargo transport assay with AlphaFold2-guided mutagenesis to identify a regulatory helix in the mitochondrial adaptor protein (TRAK) that mediates switching between kinesin- and dynein-driven transport. Differences in the helix sequence explain why two near-identical TRAK isoforms transport mitochondria in predominantly opposite directions. Phosphorylation of the regulatory helix by stress-activated kinases causes the activation of dynein and dissociation of kinesin. Our results reveal a molecular mechanism for coordinating the directional transport of mitochondria in response to intracellular signals.
-
Cell Biology
UBAP2L ensures homeostasis of nuclear pore complexes at the intact nuclear envelope.
In The Journal of Cell Biology on 1 July 2024 by Liao, Y., Andronov, L., et al.
Assembly of macromolecular complexes at correct cellular sites is crucial for cell function. Nuclear pore complexes (NPCs) are large cylindrical assemblies with eightfold rotational symmetry, built through hierarchical binding of nucleoporins (Nups) forming distinct subcomplexes. Here, we uncover a role of ubiquitin-associated protein 2-like (UBAP2L) in the assembly and stability of properly organized and functional NPCs at the intact nuclear envelope (NE) in human cells. UBAP2L localizes to the nuclear pores and facilitates the formation of the Y-complex, an essential scaffold component of the NPC, and its localization to the NE. UBAP2L promotes the interaction of the Y-complex with POM121 and Nup153, the critical upstream factors in a well-defined sequential order of Nups assembly onto NE during interphase. Timely localization of the cytoplasmic Nup transport factor fragile X-related protein 1 (FXR1) to the NE and its interaction with the Y-complex are likewise dependent on UBAP2L. Thus, this NPC biogenesis mechanism integrates the cytoplasmic and the nuclear NPC assembly signals and ensures efficient nuclear transport, adaptation to nutrient stress, and cellular proliferative capacity, highlighting the importance of NPC homeostasis at the intact NE.
© 2024 Liao et al.
-
Homo sapiens (Human)
-
Cell Biology
A phosphate-binding pocket in cyclin B3 is essential for XErp1/ Emi2 degradation in meiosis I
Preprint on BioRxiv : the Preprint Server for Biology on 16 May 2024 by Demmig, R., Schäfer, M., et al.
To ensure the correct euploid state of embryos, it is essential that vertebrate oocytes await fertilization arrested at metaphase of meiosis II. This MII arrest is mediated by XErp1/Emi2, which inhibits the ubiquitin ligase APC/C (anaphase-promoting complex/cyclosome). Cyclin B3 in complex with Cdk1 (cyclin-dependent kinase 1) is essential to prevent an untimely arrest of vertebrate oocytes in meiosis I by targeting XErp1/Emi2 for degradation. Yet, the molecular mechanism of XErp1/Emi2 degradation in MI is not well understood. Here, by combining TRIM-Away in oocytes with egg extract and in vitro studies, we demonstrate that a hitherto unknown phosphate-binding pocket in cyclin B3 is essential for efficient XErp1/Emi2 degradation in meiosis I. This pocket enables Cdk1/cyclin B3 to bind pre-phosphorylated XErp1/Emi2 facilitating further phosphorylation events, which ultimately target XErp1/Emi2 for degradation in a Plk1 (Polo-like kinase 1) dependent manner. Key elements of this degradative mechanism are conserved in frog and mouse. Our studies identify a novel, evolutionarily conserved determinant of Cdk/cyclin substrate specificity essential to prevent an untimely oocyte arrest at meiosis I with catastrophic consequences upon fertilization.
-
WB
-
Cell Biology
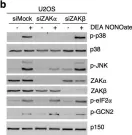
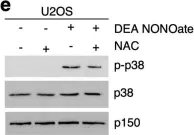
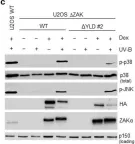
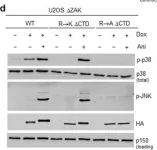
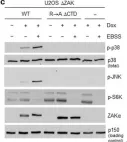
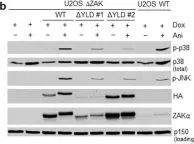
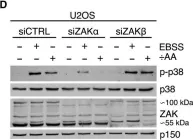
In Cell Death Dis on 26 July 2023 by Ryder, L., Arendrup, F. S., et al.
Fig.2.B

-
WB
-
Collected and cropped from Cell Death Dis by CiteAb, provided under a CC-BY license
Image 1 of 40
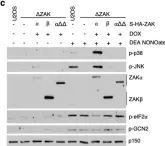
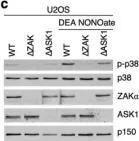
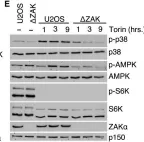
In Cell Death Dis on 26 July 2023 by Ryder, L., Arendrup, F. S., et al.
Fig.2.C

-
WB
-
Collected and cropped from Cell Death Dis by CiteAb, provided under a CC-BY license
Image 1 of 40
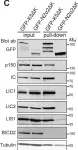
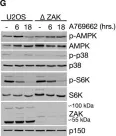
In Cell Death Dis on 26 July 2023 by Ryder, L., Arendrup, F. S., et al.
Fig.2.D

-
WB
-
Collected and cropped from Cell Death Dis by CiteAb, provided under a CC-BY license
Image 1 of 40
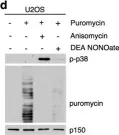
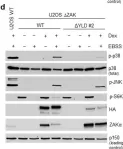
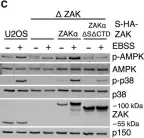
In Cell Death Dis on 26 July 2023 by Ryder, L., Arendrup, F. S., et al.
Fig.1.D

-
WB
-
Collected and cropped from Cell Death Dis by CiteAb, provided under a CC-BY license
Image 1 of 40
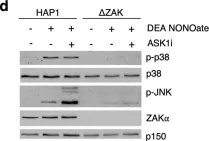
In Cell Death Dis on 26 July 2023 by Ryder, L., Arendrup, F. S., et al.
Fig.1.A

-
WB
-
Collected and cropped from Cell Death Dis by CiteAb, provided under a CC-BY license
Image 1 of 40
In Cell Death Dis on 26 July 2023 by Ryder, L., Arendrup, F. S., et al.
Fig.1.E

-
WB
-
Collected and cropped from Cell Death Dis by CiteAb, provided under a CC-BY license
Image 1 of 40
In Cell Death Dis on 26 July 2023 by Ryder, L., Arendrup, F. S., et al.
Fig.1.C

-
WB
-
Collected and cropped from Cell Death Dis by CiteAb, provided under a CC-BY license
Image 1 of 40
In J Cell Biol on 1 May 2023 by Garner, K. E. L., Salter, A., et al.
Fig.1.C

-
WB
-
Collected and cropped from J Cell Biol by CiteAb, provided under a CC-BY license
Image 1 of 40
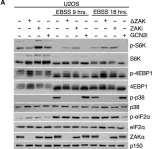
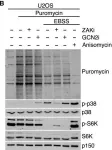
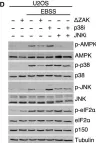
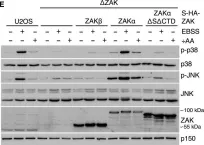
In Cells on 22 March 2023 by Johansen, V. B. I., Snieckute, G., et al.
Fig.5.D

-
WB
-
Homo sapiens (Human)
Collected and cropped from Cells by CiteAb, provided under a CC-BY license
Image 1 of 40
In Cells on 22 March 2023 by Johansen, V. B. I., Snieckute, G., et al.
Fig.5.C

-
WB
-
Homo sapiens (Human)
Collected and cropped from Cells by CiteAb, provided under a CC-BY license
Image 1 of 40
In Cells on 22 March 2023 by Johansen, V. B. I., Snieckute, G., et al.
Fig.1.D

-
WB
-
Homo sapiens (Human)
Collected and cropped from Cells by CiteAb, provided under a CC-BY license
Image 1 of 40
In Cells on 22 March 2023 by Johansen, V. B. I., Snieckute, G., et al.
Fig.1.C

-
WB
-
Homo sapiens (Human)
Collected and cropped from Cells by CiteAb, provided under a CC-BY license
Image 1 of 40
In Cells on 22 March 2023 by Johansen, V. B. I., Snieckute, G., et al.
Fig.5.B

-
WB
-
Homo sapiens (Human)
Collected and cropped from Cells by CiteAb, provided under a CC-BY license
Image 1 of 40
In Cell Metab on 6 December 2022 by Snieckute, G., Genzor, A. V., et al.
Fig.1.D

-
WB
-
Collected and cropped from Cell Metab by CiteAb, provided under a CC-BY license
Image 1 of 40
In Cell Metab on 6 December 2022 by Snieckute, G., Genzor, A. V., et al.
Fig.2.E

-
WB
-
Collected and cropped from Cell Metab by CiteAb, provided under a CC-BY license
Image 1 of 40
In Cell Metab on 6 December 2022 by Snieckute, G., Genzor, A. V., et al.
Fig.2.G

-
WB
-
Collected and cropped from Cell Metab by CiteAb, provided under a CC-BY license
Image 1 of 40
In Cell Metab on 6 December 2022 by Snieckute, G., Genzor, A. V., et al.
Fig.2.C

-
WB
-
Collected and cropped from Cell Metab by CiteAb, provided under a CC-BY license
Image 1 of 40
In Cell Metab on 6 December 2022 by Snieckute, G., Genzor, A. V., et al.
Fig.2.A

-
WB
-
Collected and cropped from Cell Metab by CiteAb, provided under a CC-BY license
Image 1 of 40
In Cell Metab on 6 December 2022 by Snieckute, G., Genzor, A. V., et al.
Fig.2.B

-
WB
-
Collected and cropped from Cell Metab by CiteAb, provided under a CC-BY license
Image 1 of 40
In Cell Metab on 6 December 2022 by Snieckute, G., Genzor, A. V., et al.
Fig.2.D

-
WB
-
Collected and cropped from Cell Metab by CiteAb, provided under a CC-BY license
Image 1 of 40
In Cell Metab on 6 December 2022 by Snieckute, G., Genzor, A. V., et al.
Fig.1.E

-
WB
-
Collected and cropped from Cell Metab by CiteAb, provided under a CC-BY license
Image 1 of 40
In Cell Metab on 6 December 2022 by Snieckute, G., Genzor, A. V., et al.
Fig.1.A

-
WB
-
Collected and cropped from Cell Metab by CiteAb, provided under a CC-BY license
Image 1 of 40
In Cell Metab on 6 December 2022 by Snieckute, G., Genzor, A. V., et al.
Fig.1.B

-
WB
-
Collected and cropped from Cell Metab by CiteAb, provided under a CC-BY license
Image 1 of 40
In Nat Commun on 21 March 2022 by Keren-Kaplan, T., Saric, A., et al.
Fig.7.B

-
WB
-
Homo sapiens (Human)
Collected and cropped from Nat Commun by CiteAb, provided under a CC-BY license
Image 1 of 40
In Nat Commun on 21 March 2022 by Keren-Kaplan, T., Saric, A., et al.
Fig.7.A

-
WB
-
Homo sapiens (Human)
Collected and cropped from Nat Commun by CiteAb, provided under a CC-BY license
Image 1 of 40