The molecular mechanisms regulating the zonal distribution of metabolism in liver are incompletely understood. Here we use single nuclei genomics techniques to examine the spatial transcriptional function of transcription factor 7-like 2 (TCF7L2) in mouse liver, and determine the consequences of TCF7L2 transcriptional inactivation on the metabolic architecture of the liver and the function of zonated metabolic pathways. We report that while Tcf7l2 mRNA expression is ubiquitous across the liver lobule, accessibility of the consensus TCF/LEF DNA binding motif is restricted to pericentral (PC) hepatocytes in zone 3. In mice expressing functionally inactive TCF7L2 in liver, PC hepatocyte-specific gene expression is absent, which we demonstrate promotes hepatic cholesterol accumulation, impaired bile acid synthesis, disruption to glutamine/glutamate homeostasis and pronounced dietary-induced hepatic fibrosis. In summary, TCF7L2 is a key regulator of hepatic zonal gene expression and regulates several zonated metabolic pathways that may contribute to the development of fibrotic liver disease.
© 2025. The Author(s).
Product Citations: 144
In Nature Communications on 10 April 2025 by Ayala, I., Hebbale, S. K., et al.
-
IHC
-
Mus musculus (House mouse)
-
Biochemistry and Molecular biology
-
Cell Biology
In eLife on 6 March 2025 by Taylor, O. B., DeGroff, N., et al.
The purpose of these studies is to investigate how Sphingosine-1-phosphate (S1P) signaling regulates glial phenotype, dedifferentiation of Müller glia (MG), reprogramming into proliferating MG-derived progenitor cells (MGPCs), and neuronal differentiation of the progeny of MGPCs in the chick retina. We found that S1P-related genes are highly expressed by retinal neurons and glia, and levels of expression were dynamically regulated following retinal damage. Drug treatments that activate S1P receptor 1 (S1PR1) or increase levels of S1P suppressed the formation of MGPCs. Conversely, treatments that inhibit S1PR1 or decrease levels of S1P stimulated the formation of MGPCs. Inhibition of S1P receptors or S1P synthesis significantly enhanced the neuronal differentiation of the progeny of MGPCs. We report that S1P-related gene expression in MG is modulated by microglia and inhibition of S1P receptors or S1P synthesis partially rescues the loss of MGPC formation in damaged retinas missing microglia. Finally, we show that TGFβ/Smad3 signaling in the resting retina maintains S1PR1 expression in MG. We conclude that the S1P signaling is dynamically regulated in MG and MGPCs in the chick retina, and activation of S1P signaling depends, in part, on signals produced by reactive microglia.
© 2024, Taylor et al.
-
Neuroscience
Preprint on Research Square on 16 January 2025 by Esperante, I., Banzan, C., et al.
Abstract The Wobbler mouse is a genetic model of familial amyotrophic lateral sclerosis. Wobblers show spinal cord neurodegeneration associated with gliosis, neuroinflammation, and demyelination. Similar to human neurodegenerative diseases, Wobblers show high levels of corticosterone in blood and the nervous system. A role for glucocorticoids in neuropathology is suggested by the observation that pathological signs attenuate with treatment with glucocorticoid receptor (GR) antagonists/modulators. In the present study, we demonstrated in 5-month-old clinically afflicted Wobbler mice that the selective GR modulator CORT125329 decreased motoneuron degeneration, astro- and microgliosis, and levels of pro-inflammatory factors (HMGB1, toll-like receptor 4, tumor necrosis factorFα, and its receptor). In addition, CORT125329 increased the acetylcholine-producing enzyme choline acetyltransferase, the neurotrophin brain-derived neurotrophic factor, and their cellular colocalization. Furthermore, the increased oligodendrocyte number and a healthier myelin ultrastructure are consistent with the enhanced axonal myelination after CORT125329 treatment. Finally, the high expression of immunoreactive protein and mRNA levels of acquaporin4 in Wobblers was decreased by CORT125329 treatment, implying this water channel is a glucocorticoid target involved in neuropathology. The beneficial effects of CORT125329 correlated with enhanced motor behavioral performance and trophic changes of the forelimbs. In conclusion our results support further preclinical and clinical studies with GR modulators in sporadic amyotrophic lateral sclerosis.
-
Neuroscience
In Frontiers in Cell and Developmental Biology on 5 December 2024 by Rumford, J. E., Grieshaber, A., et al.
Inflammation and microglia appear to be key factors influencing the outcome of retinal regeneration following acute retinal damage. Despite such findings, direct connection of microglia-specific inflammatory factors as drivers of regenerative responses in the retina are still not defined, and intracellular pathways activated to stimulate such signals from microglia are currently unknown. We became interested in MyD88 regulation in microglia because transcriptomic datasets suggest myd88 could be regulated temporally in zebrafish microglia responding to damage in the central nervous system. MyD88 is an intracellular molecular adaptor that initiates signaling cascades downstream of several innate immune receptors, and probably most well-known for inducing gene expression of pro-inflammatory factors. Using zebrafish, which spontaneously regenerate retinal neurons after acute retinal damage, we studied the effects of overactivation of MyD88 signaling in microglia and macrophages on the Müller glia-mediated regenerative response. Our results indicate that increased MyD88 signaling in microglia/macrophages impacts the initial response of Müller glia entering a regenerative response after acute, neurotoxin-induced retinal damage to inner retinal neurons. In addition, increased MyD88 signaling in microglia/macrophages resulted in reduced survival of inner retinal neurons in regenerated retinas. This work supports the idea that temporal control of inflammatory signaling is a key component in the production of MG-derived progenitors yet further indicates that such control is important for differentiation and survival of regenerated neurons.
Copyright © 2024 Rumford, Grieshaber, Lewiston, Reed, Long and Mitchell.
-
IHC-IF
-
Neuroscience
In Frontiers in Medicine on 28 November 2024 by Pang, V. Y., Yang, Z., et al.
The elevation of the intraocular and extraocular pressures is associated with various visual conditions, including glaucoma and traumatic retinal injury. The retina expresses mechanosensitive channels (MSCs), but the role of MSCs in retinal physiology and pathologies has been unclear.
Using immunocytochemistry, confocal microscopy, and patch-clamp recording techniques, we studied the co-expression of K+-permeable (K-MSCs) TRAAK and big potassium channel BK with the epithelial sodium channel ENaC and transient receptor potential channel vanilloid TPRV4 and TRPV2 favorably permeable to Ca2+ than Na+ (together named N-MSCs), and TRPV4 activity in the mouse retina.
TRAAK immunoreactivity (IR) was mainly located in Müller cells. Photoreceptor outer segments (OSs) expressed BK and ENaCα intensively and TRAAK, TRPV2, and TRPV4 weakly. Somas and axons of retinal ganglion cells (RGCs) retrograde-identified clearly expressed ENaCα, TRPV4, and TRPV2 but lacked TRAAK and BK. Rod bipolar cells (RBCs) showed TRPV4-IR in somas and BK-IR in axonal globules. Horizontal cells were BK-negative, and some cone BCs lacked TRPV4-IR. TRPV4 agonist depolarized RGCs, enhanced spontaneous spikes and excitatory postsynaptic currents, reduced the visual signal reliability (VSR = 1-noise/signal) by ~50%, and resulted in ATP crisis, which could inactivate voltage-gated sodium channels in RGCs.
Individual neurons co-express hyperpolarizing K-MSCs with depolarizing N-MSCs to counterbalance the pressure-induced excitation, and the level of K-MSCs relative to N-MSCs (RK/N ratio) is balanced in the outer retina but low in RGCs, bringing out novel determinants for the pressure vulnerability of retinal neurons and new targets for clinical interventions.
Copyright © 2024 Pang, Yang, Wu and Pang.
-
Neuroscience
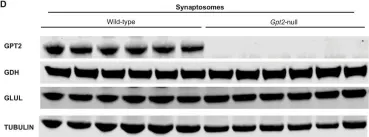
In Mol Brain on 27 November 2024 by Baytas, O., Davidson, S. M., et al.
Fig.6.D

-
WB
-
Mus musculus (House mouse)
Collected and cropped from Mol Brain by CiteAb, provided under a CC-BY license
Image 1 of 5
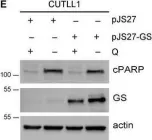
In Mol Oncol on 1 May 2021 by Nguyen, T. L., Nokin, M. J., et al.
Fig.6.E

-
WB
-
Collected and cropped from Mol Oncol by CiteAb, provided under a CC-BY license
Image 1 of 5
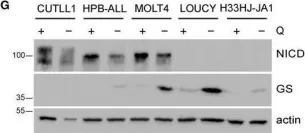
In Mol Oncol on 1 May 2021 by Nguyen, T. L., Nokin, M. J., et al.
Fig.6.G

-
WB
-
Collected and cropped from Mol Oncol by CiteAb, provided under a CC-BY license
Image 1 of 5
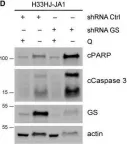
In Mol Oncol on 1 May 2021 by Nguyen, T. L., Nokin, M. J., et al.
Fig.6.D

-
WB
-
Collected and cropped from Mol Oncol by CiteAb, provided under a CC-BY license
Image 1 of 5
In Mol Oncol on 1 May 2021 by Nguyen, T. L., Nokin, M. J., et al.
Fig.6.F

-
WB
-
Collected and cropped from Mol Oncol by CiteAb, provided under a CC-BY license
Image 1 of 5