Lysosomes are membrane-bound organelles critical for maintaining cellular homeostasis. Delivery of biosynthetic lysosomal proteins to lysosomes is crucial to orchestrate proper lysosomal function. However, it remains unknown how the delivery of biosynthetic lysosomal proteins to lysosomes is ensured in neurons, which are highly polarized cells. Here, we developed Protein Origin, Trafficking And Targeting to Organelle Mapping (POTATOMap), by combining trafficking synchronization and proximity-labelling based proteomics, to unravel the trafficking routes and interactome of the biosynthetic lysosomal membrane protein LAMP1 at specified time points. This approach, combined with advanced microscopy, enables us to identify the neuronal domain-specific trafficking machineries of biosynthetic LAMP1. We reveal a role in replenishing axonal lysosomes, in delivery of newly synthesized axonal synaptic proteins, and interactions with RNA granules to facilitate hitchhiking in the axon. POTATOMap offers a robust approach to map out dynamic biosynthetic protein trafficking and interactome from their origin to destination.
© 2025. The Author(s).
Product Citations: 27
In Nature Communications on 30 December 2024 by Li, C. H., Kersten, N., et al.
-
Cell Biology
-
Neuroscience
Spatiotemporal proteomics reveals the biosynthetic lysosomal membrane protein interactome in neurons
Preprint on BioRxiv : the Preprint Server for Biology on 16 May 2024 by Li, C. H., Kersten, N., et al.
Lysosomes are membrane-bound organelles critical for maintaining cellular homeostasis. Delivery of biosynthetic lysosomal proteins to lysosomes is crucial to orchestrate proper lysosomal function. However, it remains unknown how the delivery of biosynthetic lysosomal proteins to lysosomes is ensured in neurons, which are highly polarized cells. Here, we developed Protein Origin, Trafficking And Targeting to Organelle Mapping (POTATOMap), by combining trafficking synchronization and proximity-labelling based proteomics, to unravel the trafficking routes and interactome of the biosynthetic lysosomal membrane protein LAMP1 at specified time points. This approach, combined with advanced microscopy, enabled us to identify the neuronal domain-specific trafficking machineries of biosynthetic LAMP1. We revealed a role in replenishing axonal lysosomes, in delivery of newly synthesized axonal synaptic proteins, and interactions with RNA granules to facilitate hitchhiking in the axon. POTATOMap offers a robust approach to map out dynamic biosynthetic protein trafficking and interactome from their origin to destination.
-
Cell Biology
-
Neuroscience
Rab21 regulates caveolin-1-mediated endocytic trafficking to promote immature neurite pruning.
In EMBO Reports on 6 March 2023 by Shikanai, M., Ito, S., et al.
Transmembrane proteins are internalized by clathrin- and caveolin-dependent endocytosis. Both pathways converge on early endosomes and are thought to share the small GTPase Rab5 as common regulator. In contrast to this notion, we show here that the clathrin- and caveolin-mediated endocytic pathways are differentially regulated. Rab5 and Rab21 localize to distinct populations of early endosomes in cortical neurons and preferentially regulate clathrin- and caveolin-mediated pathways, respectively, suggesting heterogeneity in the early endosomes, rather than a converging point. Suppression of Rab21, but not Rab5, results in decreased plasma membrane localization and total protein levels of caveolin-1, which perturbs immature neurite pruning of cortical neurons, an in vivo-specific step of neuronal maturation. Taken together, our data indicate that clathrin- and caveolin-mediated endocytic pathways run in parallel in early endosomes, which show different molecular regulation and physiological function.
© 2023 The Authors. Published under the terms of the CC BY 4.0 license.
In Frontiers in Neuroanatomy on 18 February 2022 by Ferdos, S., Brockhaus, J., et al.
Communication between neurons through synapses includes the release of neurotransmitter-containing synaptic vesicles (SVs) and of neuromodulator-containing dense-core vesicles (DCVs). Neurexins (Nrxns), a polymorphic family of cell surface molecules encoded by three genes in vertebrates (Nrxn1-3), have been proposed as essential presynaptic organizers and as candidates for cell type-specific or even synapse-specific regulation of synaptic vesicle exocytosis. However, it remains unknown whether Nrxns also regulate DCVs. Here, we report that at least β-neurexins (β-Nrxns), an extracellularly smaller Nrxn variant, are involved in the distribution of presynaptic DCVs. We found that conditional deletion of all three β-Nrxn isoforms in mice by lentivirus-mediated Cre recombinase expression in primary hippocampal neurons reduces the number of ultrastructurally identified DCVs in presynaptic boutons. Consistently, colabeling against marker proteins revealed a diminished population of chromogranin A- (ChrgA-) positive DCVs in synapses and axons of β-Nrxn-deficient neurons. Moreover, we validated the impaired DCV distribution in cerebellar brain tissue from constitutive β-Nrxn knockout (β-TKO) mice, where DCVs are normally abundant and β-Nrxn isoforms are prominently expressed. Finally, we observed that the ultrastructure and marker proteins of the Golgi apparatus, responsible for packaging neuropeptides into DCVs, seem unchanged. In conclusion, based on the validation from the two deletion strategies in conditional and constitutive KO mice, two neuronal populations from the hippocampus and cerebellum, and two experimental protocols in cultured neurons and in the brain tissue, this study presented morphological evidence that the number of DCVs at synapses is altered in the absence of β-Nrxns. Our results therefore point to an unexpected contribution of β-Nrxns to the organization of neuropeptide and neuromodulator function, in addition to their more established role in synaptic vesicle release.
Copyright © 2022 Ferdos, Brockhaus, Missler and Rohlmann.
-
ICC
-
Neuroscience
In The Journal of Biological Chemistry on 1 December 2021 by Jaimon, E., Tripathi, A., et al.
ARL5B, an ARF-like small GTPase localized to the trans-Golgi, is known for regulating endosome-Golgi trafficking and promoting the migration and invasion of breast cancer cells. Although a few interacting partners have been identified, the mechanism of the shuttling of ARL5B between the Golgi membrane and the cytosol is still obscure. Here, using GFP-binding protein (GBP) pull-down followed by mass spectrometry, we identified heat shock cognate protein (HSC70) as an additional interacting partner of ARL5B. Our pull-down and isothermal titration calorimetry (ITC)-based studies suggested that HSC70 binds to ARL5B in an ADP-dependent manner. Additionally, we showed that the N-terminal helix and the nucleotide status of ARL5B contribute to its recognition by HSC70. The confocal microscopy and cell fractionation studies in MDA-MB-231 breast cancer cells revealed that the depletion of HSC70 reduces the localization of ARL5B to the Golgi. Using in vitro reconstitution approach, we provide evidence that HSC70 fine-tunes the association of ARL5B with Golgi membrane. Finally, we demonstrated that the interaction between ARL5B and HSC70 is important for the localization of cation independent mannose-6-phosphate receptor (CIMPR) at Golgi. Collectively, we propose a mechanism by which HSC70, a constitutively expressed chaperone, modulates the Golgi association of ARL5B, which in turn has implications for the Golgi-associated functions of this GTPase.
Copyright © 2021 The Authors. Published by Elsevier Inc. All rights reserved.
-
Homo sapiens (Human)
-
Biochemistry and Molecular biology
-
Cell Biology
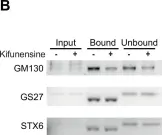
In Elife on 8 June 2016 by Mkhikian, H., Mortales, C. L., et al.
Fig.6.B

-
WB
-
Homo sapiens (Human)
Collected and cropped from Elife by CiteAb, provided under a CC-BY license
Image 1 of 3
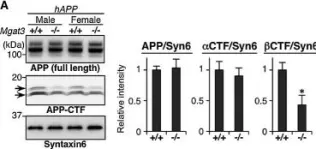
In EMBO Mol Med on 1 February 2015 by Kizuka, Y., Kitazume, S., et al.
Fig.3.A

-
WB
-
Collected and cropped from EMBO Mol Med by CiteAb, provided under a CC-BY license
Image 1 of 3
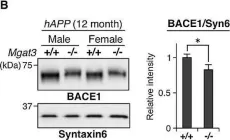
In EMBO Mol Med on 1 February 2015 by Kizuka, Y., Kitazume, S., et al.
Fig.6.B

-
WB
-
Collected and cropped from EMBO Mol Med by CiteAb, provided under a CC-BY license
Image 1 of 3