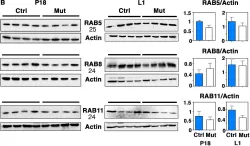
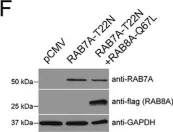
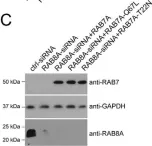
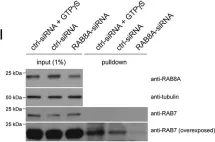
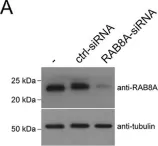
The Rab11-Rab8 cascade mediated by the Rab8 guanine nucleotide exchange factor (GEF), Rabin8, orchestrates multiple membrane transport processes, but Rab membrane loading and exchange dynamics are unclear. Here, we use advanced fluorescence imaging approaches to characterize Rab11, Rab8, and Rabin8 protein dynamics. Using fluorescence ablation and recovery studies (FRAP), we show that Rab8 ciliary trafficking requires Rab11 and Rabin8. Reciprocally, we discover that Rab11 is recruited to cilia during ciliogenesis in association with Rab8. We uncover a requirement for this cascade in Rab8 association with long tubular membranes (LTMs) in human cells and zebrafish embryos. Membrane exchange dynamics of Rab11 on Rab8 LTMs is shown using super-resolution imaging, along with a dependency on Rabin8 GEF activity. Finally, cascade-dependent Rab8 loading onto enlarged Rab11-Rabin8 membrane structures is discussed. This study demonstrates that the Rab11-Rab8 cascade involves membrane conversion and expands our understanding of the cellular multifunctionality of this trafficking pathway.
Published by Elsevier Inc.
Product Citations: 59
Rab11-Rab8 cascade dynamics in primary cilia and membrane tubules.
In Cell Reports on 26 November 2024 by Saha, I., Insinna, C., et al.
In Neuron on 4 September 2024 by Lam, I., Ndayisaba, A., et al.
The heterogeneity of protein-rich inclusions and its significance in neurodegeneration is poorly understood. Standard patient-derived iPSC models develop inclusions neither reproducibly nor in a reasonable time frame. Here, we developed screenable iPSC "inclusionopathy" models utilizing piggyBac or targeted transgenes to rapidly induce CNS cells that express aggregation-prone proteins at brain-like levels. Inclusions and their effects on cell survival were trackable at single-inclusion resolution. Exemplar cortical neuron α-synuclein inclusionopathy models were engineered through transgenic expression of α-synuclein mutant forms or exogenous seeding with fibrils. We identified multiple inclusion classes, including neuroprotective p62-positive inclusions versus dynamic and neurotoxic lipid-rich inclusions, both identified in patient brains. Fusion events between these inclusion subtypes altered neuronal survival. Proteome-scale α-synuclein genetic- and physical-interaction screens pinpointed candidate RNA-processing and actin-cytoskeleton-modulator proteins like RhoA whose sequestration into inclusions could enhance toxicity. These tractable CNS models should prove useful in functional genomic analysis and drug development for proteinopathies.
Copyright © 2024 The Authors. Published by Elsevier Inc. All rights reserved.
-
Neuroscience
-
Stem Cells and Developmental Biology
In Journal of Cell Science on 1 December 2023 by Ray, A., Wen, J., et al.
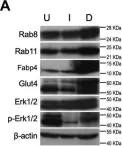
Regulation of glucose transport, which is central for control of whole-body metabolism, is determined by the amount of GLUT4 glucose transporter (also known as SLC2A4) in the plasma membrane (PM) of fat and muscle cells. Physiologic signals [such as activated insulin receptor or AMP-activated protein kinase (AMPK)] increase PM GLUT4. Here, we show that the distribution of GLUT4 between the PM and interior of human muscle cells is dynamically maintained, and that AMPK promotes PM redistribution of GLUT4 by regulating exocytosis and endocytosis. Stimulation of exocytosis by AMPK is mediated by Rab10 and the Rab GTPase-activating protein TBC1D4. APEX2 proximity mapping reveals that GLUT4 traverses both PM-proximal and PM-distal compartments in unstimulated muscle cells, further supporting retention of GLUT4 by a constitutive retrieval mechanism. AMPK-stimulated translocation involves GLUT4 redistribution among the same compartments traversed in unstimulated cells, with a significant recruitment of GLUT4 from the Golgi and trans-Golgi network compartments. Our comprehensive proximal protein mapping provides an integrated, high-density, whole-cell accounting of the localization of GLUT4 at a resolution of ∼20 nm that serves as a structural framework for understanding the molecular mechanisms regulating GLUT4 trafficking downstream of different signaling inputs in a physiologically relevant cell type.
© 2023. Published by The Company of Biologists Ltd.
-
WB
-
Cell Biology
Preprint on BioRxiv : the Preprint Server for Biology on 7 June 2023 by Ray, A., Wen, J., et al.
Regulation of glucose transport into muscle and adipocytes, central for control of whole-body metabolism, is determined by the amount of GLUT4 glucose transporter in the plasma membrane ( PM ). Physiologic signals (activated insulin receptor or AMP kinase [ AMPK ]), acutely increase PM GLUT4 to enhance glucose uptake. Here we show in kinetic studies that intracellular GLUT4 is in equilibrium with the PM in unstimulated cultured human skeletal muscle cells, and that AMPK promotes GLUT4 redistribution to the PM by regulating both exocytosis and endocytosis. AMPK-stimulation of exocytosis requires Rab10 and Rab GTPase activating protein TBC1D4, requirements shared with insulin control of GLUT4 in adipocytes. Using APEX2 proximity mapping, we identify, at high-density and high-resolution, the GLUT4 proximal proteome, revealing GLUT4 traverses both PM proximal and distal compartments in unstimulated muscle cells. These data support intracellular retention of GLUT4 in unstimulated muscle cells by a dynamic mechanism dependent on the rates of internalization and recycling. AMPK promoted GLUT4 translocation to the PM involves redistribution of GLUT4 among the same compartments traversed in unstimulated cells, with a significant redistribution of GLUT4 from the PM distal Trans Golgi Network Golgi compartments. The comprehensive proximal protein mapping provides an integrated, whole cell accounting of GLUT4’s localization at a resolution of ∼20 nm, a structural framework for understanding the molecular mechanisms regulating GLUT4 trafficking downstream of different signaling inputs in physiologically relevant cell type and as such, sheds new light on novel key pathways and molecular components as potential therapeutic approaches to modulate muscle glucose uptake.
-
WB
-
Mus musculus (House mouse)
In The Journal of Biological Chemistry on 1 May 2023 by Mahajan, D., Madugula, V., et al.
Arl13b, an ARF/Arl-family GTPase, is highly enriched in the cilium. Recent studies have established Arl13b as one of the most crucial regulators for ciliary organization, trafficking, and signaling. The ciliary localization of Arl13b is known to require the RVEP motif. However, its cognate ciliary transport adaptor has been elusive. Here, by imaging the ciliary localization of truncation and point mutations, we defined the ciliary targeting sequence (CTS) of Arl13b as a C-terminal stretch of 17 amino acids containing the RVEP motif. We found Rab8-GDP, but not Rab8-GTP, and TNPO1 simultaneously and directly bind to the CTS of Arl13b in pull-down assays using cell lysates or purified recombinant proteins. Furthermore, Rab8-GDP substantially enhances the interaction between TNPO1 and CTS. Additionally, we determined that the RVEP motif is an essential element as its mutation abolishes the interaction of the CTS with Rab8-GDP and TNPO1 in pull-down and TurboID-based proximity ligation assays. Finally, the knockdown of endogenous Rab8 or TNPO1 decreases the ciliary localization of endogenous Arl13b. Therefore, our results suggest Rab8 and TNPO1 might function together as a ciliary transport adaptor for Arl13b by interacting with its RVEP-containing CTS.
Copyright © 2023 The Authors. Published by Elsevier Inc. All rights reserved.
-
WB
-
Biochemistry and Molecular biology
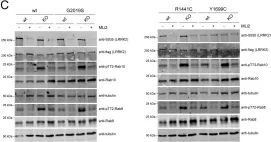
In Biol Open on 15 August 2022 by Lara Ordóñez, A. J., Fasiczka, R., et al.
Fig.2.C

-
WB
-
Homo sapiens (Human)
Collected and cropped from Biol Open by CiteAb, provided under a CC-BY license
Image 1 of 17
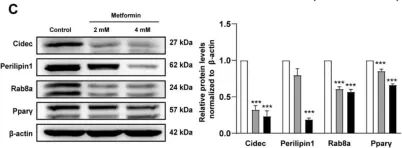
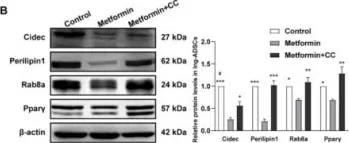
In Int J Mol Sci on 26 May 2022 by Yang, L., Jia, X., et al.
Fig.4.C

-
WB
-
Rattus norvegicus (Rat)
Collected and cropped from Int J Mol Sci by CiteAb, provided under a CC-BY license
Image 1 of 17
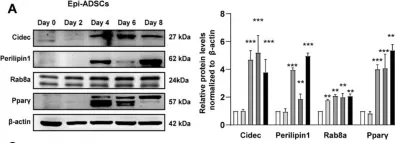
In Int J Mol Sci on 26 May 2022 by Yang, L., Jia, X., et al.
Fig.4.A

-
WB
-
Rattus norvegicus (Rat)
Collected and cropped from Int J Mol Sci by CiteAb, provided under a CC-BY license
Image 1 of 17
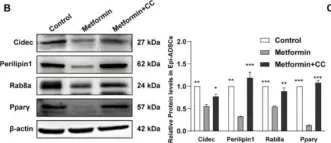
In Int J Mol Sci on 26 May 2022 by Yang, L., Jia, X., et al.
Fig.5.B

-
WB
-
Rattus norvegicus (Rat)
Collected and cropped from Int J Mol Sci by CiteAb, provided under a CC-BY license
Image 1 of 17
In Int J Mol Sci on 26 May 2022 by Yang, L., Jia, X., et al.
Fig.6.B

-
WB
-
Rattus norvegicus (Rat)
Collected and cropped from Int J Mol Sci by CiteAb, provided under a CC-BY license
Image 1 of 17
In Cells on 29 January 2022 by Limone, A., Veneruso, I., et al.
Fig.4.A

-
WB
-
Collected and cropped from Cells by CiteAb, provided under a CC-BY license
Image 1 of 17
In J Biol Chem on 5 March 2021 by Stypulkowski, E., Feng, Q., et al.
Fig.3.A

-
WB
-
Collected and cropped from J Biol Chem by CiteAb, provided under a CC-BY license
Image 1 of 17
In Development on 8 October 2020 by Cayre, S., Faraldo, M. M., et al.
Fig.5.B

-
WB
-
Collected and cropped from Development by CiteAb, provided under a CC-BY license
Image 1 of 17
In EMBO Rep on 1 April 2019 by Takahashi, T., Minami, S., et al.
Fig.4.T

-
WB
-
Collected and cropped from EMBO Rep by CiteAb, provided under a CC-BY license
Image 1 of 17
In EMBO Rep on 1 April 2019 by Takahashi, T., Minami, S., et al.
Fig.4.E

-
WB
-
Collected and cropped from EMBO Rep by CiteAb, provided under a CC-BY license
Image 1 of 17
In EMBO Rep on 1 April 2019 by Takahashi, T., Minami, S., et al.
Fig.1.C

-
WB
-
Collected and cropped from EMBO Rep by CiteAb, provided under a CC-BY license
Image 1 of 17
In J Biol Chem on 29 March 2019 by Rivero-Ríos, P., Romo-Lozano, M., et al.
Fig.5.G

-
WB
-
Homo sapiens (Human)
Collected and cropped from J Biol Chem by CiteAb, provided under a CC-BY license
Image 1 of 17
In J Biol Chem on 29 March 2019 by Rivero-Ríos, P., Romo-Lozano, M., et al.
Fig.10.F

-
WB
-
Homo sapiens (Human)
Collected and cropped from J Biol Chem by CiteAb, provided under a CC-BY license
Image 1 of 17
In J Biol Chem on 29 March 2019 by Rivero-Ríos, P., Romo-Lozano, M., et al.
Fig.8.C

-
WB
-
Homo sapiens (Human)
Collected and cropped from J Biol Chem by CiteAb, provided under a CC-BY license
Image 1 of 17
In J Biol Chem on 29 March 2019 by Rivero-Ríos, P., Romo-Lozano, M., et al.
Fig.5.I

-
WB
-
Homo sapiens (Human)
Collected and cropped from J Biol Chem by CiteAb, provided under a CC-BY license
Image 1 of 17
In J Biol Chem on 29 March 2019 by Rivero-Ríos, P., Romo-Lozano, M., et al.
Fig.5.A

-
WB
-
Homo sapiens (Human)
Collected and cropped from J Biol Chem by CiteAb, provided under a CC-BY license
Image 1 of 17
In PLoS Biol on 1 April 2015 by Zhang, B., Zhang, T., et al.
Fig.3.B

-
WB
-
Collected and cropped from PLoS Biol by CiteAb, provided under a CC-BY license
Image 1 of 17