Subcellular mitochondrial positioning in cells is necessary for localized energy and signaling requirements. Mitochondria are strategically trafficked throughout the cytoplasm via the actin cytoskeleton, microtubule motor proteins, and adaptor proteins. Miro1, an outer mitochondrial membrane adaptor protein, is necessary for attachment of mitochondria to microtubule motor proteins for trafficking. Previous work showed when Miro1 is deleted (Miro1 -/- ) from mouse embryonic fibroblasts (MEFs), the mitochondria become sequestered to the perinuclear space, disrupting subcellular energy and reactive oxygen species gradients. Here, we show that Miro1 -/- MEFs grow slower compared to Miro1 +/+ and Miro1 -/- MEFs stably re-expressing the Myc-Miro1 plasmid. Miro1 -/- MEFs have a have a cell cycle defect with decreased percentage of cells in G1 and increased cells in the S phase of the cell cycle. We conducted the first ever RNA sequencing experiment dependent upon Miro1 expression and found differential expression in cell proliferation and migration genes upon deletion of Miro1, including the MAP Kinase signaling pathway. We find that ERK1/2 phosphorylation is elevated both spatially (cytoplasm and nucleus) and temporally following serum stimulation in Miro1 -/- MEFs. We investigated the expression levels and oxidation of the Dual Specificity Phosphatases (DUSP1-6), ERK1/2 target phosphatases. We found no differences in DUSP1-6 expression and oxidation under asynchronous and synchronized cells. Lastly, we evaluated the oxidation status of ERK1/2 and found an increase in ERK1/2 oxidation in the Miro1 -/- MEFs compared to Miro1 +/+ and Myc-Miro1. These data highlight transcriptional control based off Miro1 expression and demonstrate the highly dynamic regulation of ERK1/2 upon deletion of Miro1 that may support the observed cell cycle and proliferation defects.
Product Citations: 54
Preprint on BioRxiv : the Preprint Server for Biology on 6 November 2024 by Shannon, N., Raymond, C., et al.
-
Mus musculus (House mouse)
In Viruses on 23 September 2024 by Rosenfeld, P., Singh, G., et al.
Our examination of RNA helicases for effects on HIV-1 protein production and particle assembly identified Rocaglamide (RocA), a known modulator of eIF4A1 function, as an inhibitor of HIV-1 replication in primary CD4+ T cells and three cell systems. HIV-1 attenuation by low-nM RocA doses was associated with reduced viral particle formation without a marked decrease in Gag production. Rather, the co-localization of Gag and HIV-1 genomic RNA (gRNA) assemblies was impaired by RocA treatment in a reversible fashion. Ribonucleoprotein (RNP) immunoprecipitation studies recapitulated the loss of Gag-gRNA assemblies upon RocA treatment. Parallel biophysical studies determined that neither RocA nor eIF4A1 independently affected the ability of Gag to interact with viral RNA, but together, they distorted the structure of the HIV-1 RNP visualized by electron microscopy. Taken together, several lines of evidence indicate that RocA induces stable binding of eIF4A1 onto the viral RNA genome in a manner that interferes with the ordered assembly of Gag along Gag-gRNA assemblies required to generate infectious virions.
-
Genetics
Preprint on BioRxiv : the Preprint Server for Biology on 18 August 2024 by Maeda, H. & Sasaki, H.
The epiblast is a pluripotent cell population formed in the late blastocyst stage of preimplantation embryos. During the process of epiblast formation from the inner cell mass (ICM) of the early blastocyst, activation of the Hippo pathway transcription factor TEAD by the nuclear translocation of the coactivator protein YAP is required for the robust expression of pluripotency factors. However, the mechanisms that alter YAP localization during epiblast formation remain unknown. Here, we reveal two such mechanisms. Expansion of the blastocoel promotes nuclear YAP localization by increasing cytoplasmic F-actin and reducing YAP phosphorylation. Additionally, cell differentiation regulates YAP. Expression of the junctional Hippo component, AMOT, gradually decreases during epiblast formation through a tankyrase-mediated degradation. SOX2 expression in the ICM is necessary for the reduction of AMOT and YAP phosphorylation. These two mechanisms function in parallel. Thus, the blastocoel-F-actin and SOX2-AMOT axes cooperatively suppress YAP phosphorylation and promote YAP nuclear localization during epiblast formation. The cooperation of these two distinct mechanisms likely contributes to the robustness of epiblast cell differentiation.
-
Mus musculus (House mouse)
Time-resolved proteomic analyses of senescence highlight metabolic rewiring of mitochondria.
In Life Science Alliance on 1 September 2023 by Kim, J. Y., Atanassov, I., et al.
Mitochondrial dysfunction and cellular senescence are hallmarks of aging. However, the relationship between these two phenomena remains incompletely understood. In this study, we investigated the rewiring of mitochondria upon development of the senescent state in human IMR90 fibroblasts. Determining the bioenergetic activities and abundance of mitochondria, we demonstrate that senescent cells accumulate mitochondria with reduced OXPHOS activity, resulting in an overall increase of mitochondrial activities in senescent cells. Time-resolved proteomic analyses revealed extensive reprogramming of the mitochondrial proteome upon senescence development and allowed the identification of metabolic pathways that are rewired with different kinetics upon establishment of the senescent state. Among the early responding pathways, the degradation of branched-chain amino acid was increased, whereas the one carbon folate metabolism was decreased. Late-responding pathways include lipid metabolism and mitochondrial translation. These signatures were confirmed by metabolic flux analyses, highlighting metabolic rewiring as a central feature of mitochondria in cellular senescence. Together, our data provide a comprehensive view on the changes in mitochondrial proteome in senescent cells and reveal how the mitochondrial metabolism is rewired in senescent cells.
© 2023 Kim et al.
-
WB
-
Biochemistry and Molecular biology
-
Cell Biology
In Frontiers in Oncology on 28 February 2023 by Singh, U., Bindra, D., et al.
Bidirectional nucleo-cytoplasmic transport, regulating several vital cellular processes, is mediated by the Nuclear Pore Complex (NPC) comprising the nucleoporin (Nup) proteins. Nup88, a constituent nucleoporin, is overexpressed in many cancers, and a positive correlation exists between progressive stages of cancer and Nup88 levels. While a significant link of Nup88 overexpression in head and neck cancer exists but mechanistic details of Nup88 roles in tumorigenesis are sparse. Here, we report that Nup88 and Nup62 levels are significantly elevated in head and neck cancer patient samples and cell lines. We demonstrate that the elevated levels of Nup88 or Nup62 impart proliferation and migration advantages to cells. Interestingly, Nup88-Nup62 engage in a strong interaction independent of Nup-glycosylation status and cell-cycle stages. We report that the interaction with Nup62 stabilizes Nup88 by inhibiting the proteasome-mediated degradation of overexpressed Nup88. Overexpressed Nup88 stabilized by interaction with Nup62 can interact with NF-κB (p65) and sequesters p65 partly into nucleus of unstimulated cells. NF-κB targets like Akt, c-myc, IL-6 and BIRC3 promoting proliferation and growth are induced under Nup88 overexpression conditions. In conclusion, our data indicates that simultaneous overexpression of Nup62 and Nup88 in head and neck cancer stabilizes Nup88. Stabilized Nup88 interacts and activates p65 pathway, which perhaps is the underlying mechanism in Nup88 overexpressing tumors.
Copyright © 2023 Singh, Bindra, Samaiya and Mishra.
-
WB
-
Cancer Research
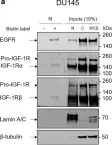
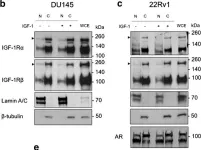
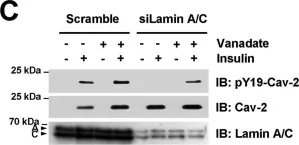
In Discov Oncol on 11 May 2021 by Mills, J. V., Osher, E., et al.
Fig.2.A

-
WB
-
Homo sapiens (Human)
Collected and cropped from Discov Oncol by CiteAb, provided under a CC-BY license
Image 1 of 10
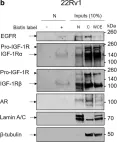
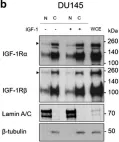
In Discov Oncol on 11 May 2021 by Mills, J. V., Osher, E., et al.
Fig.2.B

-
WB
-
Homo sapiens (Human)
Collected and cropped from Discov Oncol by CiteAb, provided under a CC-BY license
Image 1 of 10
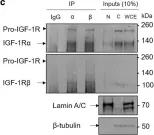
In Discov Oncol on 11 May 2021 by Mills, J. V., Osher, E., et al.
Fig.2.C

-
WB
-
Homo sapiens (Human)
Collected and cropped from Discov Oncol by CiteAb, provided under a CC-BY license
Image 1 of 10
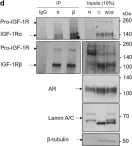
In Discov Oncol on 11 May 2021 by Mills, J. V., Osher, E., et al.
Fig.2.D

-
WB
-
Homo sapiens (Human)
Collected and cropped from Discov Oncol by CiteAb, provided under a CC-BY license
Image 1 of 10
In Discov Oncol on 11 May 2021 by Mills, J. V., Osher, E., et al.
Fig.1.B

-
WB
-
Homo sapiens (Human)
Collected and cropped from Discov Oncol by CiteAb, provided under a CC-BY license
Image 1 of 10
In Discov Oncol on 11 May 2021 by Mills, J. V., Osher, E., et al.
Fig.1.C

-
WB
-
Homo sapiens (Human)
Collected and cropped from Discov Oncol by CiteAb, provided under a CC-BY license
Image 1 of 10
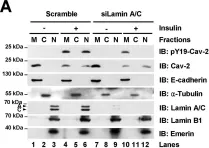
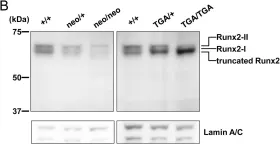
In Oncotarget on 19 July 2016 by Ferralli, J., Chiquet-Ehrismann, R., et al.
Fig.3.C

-
WB
-
Collected and cropped from Oncotarget by CiteAb, provided under a CC-BY license
Image 1 of 10
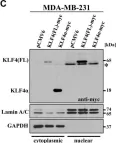
In Nucleic Acids Res on 31 March 2015 by Jeong, K., Kwon, H., et al.
Fig.2.A

-
WB
-
Homo sapiens (Human)
Collected and cropped from Nucleic Acids Res by CiteAb, provided under a CC-BY license
Image 1 of 10
In Nucleic Acids Res on 31 March 2015 by Jeong, K., Kwon, H., et al.
Fig.2.C

-
WB
-
Homo sapiens (Human)
Collected and cropped from Nucleic Acids Res by CiteAb, provided under a CC-BY license
Image 1 of 10
In PLoS One on 23 September 2014 by Okura, H., Sato, S., et al.
Fig.5.B

-
WB
-
Collected and cropped from PLoS One by CiteAb, provided under a CC-BY license
Image 1 of 10