Tissue engineering strategies predominantly rely on the production of living substitutes, whereby implanted cells actively participate in the regenerative process. Beyond cost and delayed graft availability, the patient-specific performance of engineered tissues poses serious concerns on their clinical translation ability. A more exciting paradigm consists in exploiting cell-laid, engineered extracellular matrices (eECMs), which can be used as off-the-shelf materials. Here, the regenerative capacity solely relies on the preservation of the eECM structure and embedded signals to instruct an endogenous repair. We recently described the possibility to exploit custom human stem cell lines for eECM manufacturing. In addition to the conferred standardization, the availability of such cell lines opened avenues for the design of tailored eECMs by applying dedicated genetic tools. In this study, we demonstrated the exploitation of CRISPR/Cas9 as a high precision system for editing the composition and function of eECMs. Human mesenchymal stromal/stem cell (hMSC) lines were modified to knock out vascular endothelial growth factor (VEGF) and Runt-related transcription factor 2 (RUNX2) and assessed for their capacity to generate osteoinductive cartilage matrices. We report the successful editing of hMSCs, subsequently leading to targeted VEGF and RUNX2-knockout cartilage eECMs. Despite the absence of VEGF, eECMs retained full capacity to instruct ectopic endochondral ossification. Conversely, RUNX2-edited eECMs exhibited impaired hypertrophy, reduced ectopic ossification, and superior cartilage repair in a rat osteochondral defect. In summary, our approach can be harnessed to identify the necessary eECM factors driving endogenous repair. Our work paves the road toward the compositional eECMs editing and their exploitation in broad regenerative contexts.
© 2024, Prithiviraj et al.
Product Citations: 243
In eLife on 28 March 2025 by Prithiviraj, S., Garcia Garcia, A., et al.
-
Stem Cells and Developmental Biology
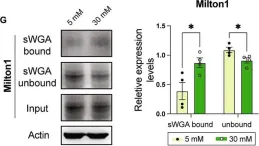
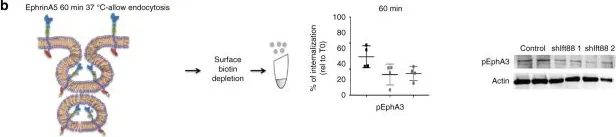
In Nature Communications on 11 March 2025 by Naghdi, S., Mishra, P., et al.
Differences between normal tissues and invading tumors that allow tumor targeting while saving normal tissue are much sought after. Here we show that scarcity of VDAC2, and the consequent lack of Bak recruitment to mitochondria, renders hepatocyte mitochondria resistant to permeabilization by truncated Bid (tBid), a Bcl-2 Homology 3 (BH3)-only, Bcl-2 family protein. Increased VDAC2 and Bak is found in most human liver cancers and mitochondria from tumors and hepatic cancer cell lines exhibit VDAC2- and Bak-dependent tBid sensitivity. Exploring potential therapeutic targeting, we find that combinations of activators of the tBid pathway with inhibitors of the Bcl-2 family proteins that suppress Bak activation enhance VDAC2-dependent death of hepatocarcinoma cells with little effect on normal hepatocytes. Furthermore, in vivo, combination of S63845, a selective Mcl-1 inhibitor, with tumor-nectrosis factor-related, apoptosis-induncing ligand (TRAIL) peptide reduces tumor growth, but only in tumors expressing VDAC2. Thus, we describe mitochondrial molecular fingerprint that discriminates liver from hepatocarcinoma and allows sparing normal tissue while targeting tumors.
© 2025. The Author(s).
-
WB
-
Rattus norvegicus (Rat)
-
Mus musculus (House mouse)
-
Cancer Research
-
Cell Biology
Laminin β4 is required for the development of human peripheral sensory neurons
Preprint on BioRxiv : the Preprint Server for Biology on 24 November 2024 by Saito-Diaz, K., Saini, T., et al.
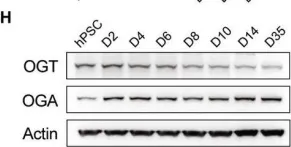
The extracellular matrix (ECM) is a mixture of glycoproteins and fibrous proteins that provide the biophysical properties necessary to maintain cellular homeostasis. ECM integrity is of particular importance during development, where it allows proper migration and cellular differentiation. Laminins are ECM heterotrimeric proteins consisting of α, β, and γ chains. There are five known α chains, four β chains, and three γ chains. Thus, there are 60 potential combinations for laminin trimers, however only 16 laminin trimers have been identified to date. Furthermore, none of them contain laminin β4 and its function is unknown. Here, we sought to characterize the role of LAMB4 (the gene encoding laminin β4) during human embryonic development of the peripheral sensory nervous system. Using human pluripotent stem cells (hPSCs), we found that LAMB4 is expressed in the ectoderm in the early stages of sensory neuron (SN) specification. SNs, part of the peripheral nervous system, are specialized neurons that detect pain, temperature, and touch. Surprisingly, more than 20 million people in the US have some form of peripheral nerve damage (including SNs), however there are very few treatment options available. Learning about the biology of peripheral neurons will uncover potential new therapeutic targets, thus we focused on understanding the effects of LAMB4 in SNs. First, we knocked out LAMB4 in hPSCs, using CRISPR/Cas9, and found that loss of LAMB4 impairs the migration of the SN progenitors neural crest cells (NCCs) and harms SN development and survival. To assess if LAMB4 has clinical relevance, we studied the genetic disorder Familial Dysautonomia (FD), which specifically affects the peripheral nervous system. FD is caused by a mutation in ELP1 (a component of the Elongator complex) leading to developmental and degenerative defects in SNs. A previous report showed that patients with severe FD harbor additional single nucleotide variants in LAMB4. We found that these variants sharply downregulate the expression of LAMB4 and laminin β4 levels in SNs differentiated from induced pluripotent stem cells (iPSCs) reprogramed from patients with severe FD. Moreover, a healthy ECM is sufficient to rescue the developmental phenotypes of FD, further confirming that ECM defects contribute significantly to the etiology of FD. Finally, we found that LAMB4/laminin β4 is necessary for actin filament accumulation and it interacts with laminin α4 and laminin γ3, forming the laminin-443, a previously unreported laminin trimer. Together, these results show that LAMB4 is a critical, but largely unknown gene required for SN development and survival.
-
Neuroscience
Preprint on Research Square on 16 September 2024 by Konishi, H., Sasaki, T., et al.
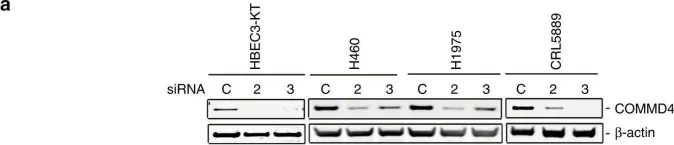
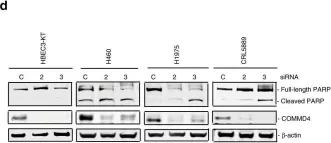
Abstract RNA dysregulation mediated by abnormal RNA binding proteins (RBPs) is associated with tumorigenesis. However, the specific tumorigenic mechanisms of each RBP remained unclear. In this study, we demonstrate that splicing factor 3A1 (SF3A1) interacts to and stabilizes the mRNA of STX12, thereby inhibiting programmed cell death (PCD) in colorectal cancer cells. Downregulation of SF3A1 significantly inhibited cell growth in colorectal cancer cells, with minimal cytotoxicity observed in non-cancerous epithelial cells. We validated the tumor-promoting function of SF3A1 in an HCT116 transplanted mouse model. TUNEL staining and western blotting of PARP revealed SF3A1 inhibits PCD in colorectal cancer cells. A transcriptome analysis, combined with RNA-immunoprecipitation (IP), demonstrated that SF3A1 interact to and stabilized 144 mRNAs. Among these mRNAs, knockdown of STX12 (Syntaxin 12) in colorectal cancer cells inhibited cell growth but had no inhibitory effect on non-cancerous epithelial cells, HCEC-1CT. The mRNA levels of STX12 were significantly reduced upon downregulation of SF3A1, contributing to the inhibition of PCD in colorectal cancer cells. Therefore, SF3A1, which mediates STX12 mRNA stabilization, represents a promising therapeutic target for the treatment of colorectal cancer with fewer side effects.
-
Cancer Research
In Biochemistry and Biophysics Reports on 1 September 2024 by Sotozono, A., Namekata, K., et al.
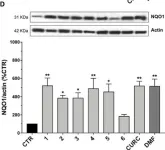
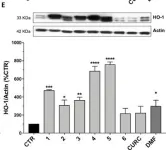
Neurodegenerative diseases including glaucoma affect insulin signaling, and insulin treatment has been shown to reverse the neurodegenerative loss of dendritic complexity in retinal ganglion cells. Therefore, strategies for enhancing or maintaining insulin signaling are worth pursuing to establish new therapies for these diseases. In the present study, we generated constitutively active insulin receptor (F-iIR) and insulin-like growth factor-1 receptor (F-iIGF1R) using a system that forces membrane localization of the intracellular domains of these receptors by farnesylation. Immunohistochemistry and Western blot analysis revealed that F-iIR and F-iIGF1R caused the activation of ERK and AKT in the absence of ligands in vitro. Our results suggest that in vivo effects of F-iIR and F-iIGF1R on the progression of neurodegenerative diseases should be investigated in the future.
© 2024 The Authors.
-
Endocrinology and Physiology
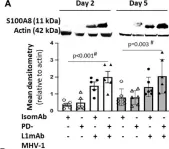
In Front Immunol on 23 January 2024 by Curran, C. S., Cui, X., et al.
Fig.7.A

-
WB
-
Collected and cropped from Front Immunol by CiteAb, provided under a CC-BY license
Image 1 of 86
In Front Neurosci on 25 May 2023 by Wu, H. F., Huang, C. W., et al.
Fig.1.H

-
WB
-
Collected and cropped from Front Neurosci by CiteAb, provided under a CC-BY license
Image 1 of 86
In Front Neurosci on 25 May 2023 by Wu, H. F., Huang, C. W., et al.
Fig.5.G

-
WB
-
Collected and cropped from Front Neurosci by CiteAb, provided under a CC-BY license
Image 1 of 86
In Front Oncol on 28 February 2023 by Singh, U., Bindra, D., et al.
Fig.5.C

-
WB
-
Collected and cropped from Front Oncol by CiteAb, provided under a CC-BY license
Image 1 of 86
In Front Oncol on 28 February 2023 by Singh, U., Bindra, D., et al.
Fig.5.B

-
WB
-
Collected and cropped from Front Oncol by CiteAb, provided under a CC-BY license
Image 1 of 86
In Front Oncol on 28 February 2023 by Singh, U., Bindra, D., et al.
Fig.5.E

-
WB
-
Collected and cropped from Front Oncol by CiteAb, provided under a CC-BY license
Image 1 of 86
In Int J Mol Sci on 16 November 2022 by Cialdai, F., Bacci, S., et al.
Fig.10.A

-
WB
-
Collected and cropped from Int J Mol Sci by CiteAb, provided under a CC-BY license
Image 1 of 86
In Int J Mol Sci on 16 November 2022 by Cialdai, F., Bacci, S., et al.
Fig.10.C

-
WB
-
Collected and cropped from Int J Mol Sci by CiteAb, provided under a CC-BY license
Image 1 of 86
In Cancers (Basel) on 1 January 2022 by Johnson, K. S., Hussein, S., et al.
Fig.6.H

-
WB
-
Homo sapiens (Human)
Collected and cropped from Cancers (Basel) by CiteAb, provided under a CC-BY license
Image 1 of 86
In Cancers (Basel) on 1 January 2022 by Johnson, K. S., Hussein, S., et al.
Fig.5.D

-
WB
-
Collected and cropped from Cancers (Basel) by CiteAb, provided under a CC-BY license
Image 1 of 86
In Cancers (Basel) on 1 January 2022 by Johnson, K. S., Hussein, S., et al.
Fig.6.D

-
WB
-
Collected and cropped from Cancers (Basel) by CiteAb, provided under a CC-BY license
Image 1 of 86
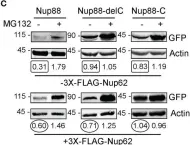
In Commun Biol on 19 April 2021 by Suraweera, A., Gandhi, N. S., et al.
Fig.4.A

-
WB
-
Homo sapiens (Human)
Collected and cropped from Commun Biol by CiteAb, provided under a CC-BY license
Image 1 of 86
In Oncogenesis on 11 December 2020 by Masi, M., Garattini, E., et al.
Fig.5.A

-
WB
-
Homo sapiens (Human)
Collected and cropped from Oncogenesis by CiteAb, provided under a CC-BY license
Image 1 of 86
In Front Cell Dev Biol on 28 August 2020 by Assar, E. A. & Tumbarello, D. A.
Fig.1.B

-
WB
-
Homo sapiens (Human)
Collected and cropped from Front Cell Dev Biol by CiteAb, provided under a CC-BY license
Image 1 of 86
In Front Cell Dev Biol on 28 August 2020 by Assar, E. A. & Tumbarello, D. A.
Fig.2.A

-
WB
-
Homo sapiens (Human)
Collected and cropped from Front Cell Dev Biol by CiteAb, provided under a CC-BY license
Image 1 of 86
In Front Cell Dev Biol on 28 August 2020 by Assar, E. A. & Tumbarello, D. A.
Fig.4.A

-
WB
-
Homo sapiens (Human)
Collected and cropped from Front Cell Dev Biol by CiteAb, provided under a CC-BY license
Image 1 of 86
In Front Cell Dev Biol on 28 August 2020 by Assar, E. A. & Tumbarello, D. A.
Fig.4.D

-
WB
-
Homo sapiens (Human)
Collected and cropped from Front Cell Dev Biol by CiteAb, provided under a CC-BY license
Image 1 of 86
In Front Cell Dev Biol on 28 August 2020 by Assar, E. A. & Tumbarello, D. A.
Fig.5.A

-
WB
-
Homo sapiens (Human)
Collected and cropped from Front Cell Dev Biol by CiteAb, provided under a CC-BY license
Image 1 of 86
In Front Cell Dev Biol on 28 August 2020 by Assar, E. A. & Tumbarello, D. A.
Fig.7.A

-
WB
-
Homo sapiens (Human)
Collected and cropped from Front Cell Dev Biol by CiteAb, provided under a CC-BY license
Image 1 of 86
In Front Cell Dev Biol on 28 August 2020 by Assar, E. A. & Tumbarello, D. A.
Fig.7.E

-
WB
-
Homo sapiens (Human)
Collected and cropped from Front Cell Dev Biol by CiteAb, provided under a CC-BY license
Image 1 of 86
In Br J Cancer on 1 August 2020 by Suraweera, A., Duff, A., et al.
Fig.3.A

-
WB
-
Collected and cropped from Br J Cancer by CiteAb, provided under a CC-BY license
Image 1 of 86
In Br J Cancer on 1 August 2020 by Suraweera, A., Duff, A., et al.
Fig.6.D

-
WB
-
Collected and cropped from Br J Cancer by CiteAb, provided under a CC-BY license
Image 1 of 86
In Front Pharmacol on 13 February 2020 by Serafini, M. M., Catanzaro, M., et al.
Fig.3.D

-
WB
-
Homo sapiens (Human)
Collected and cropped from Front Pharmacol by CiteAb, provided under a CC-BY license
Image 1 of 86
In Front Pharmacol on 13 February 2020 by Serafini, M. M., Catanzaro, M., et al.
Fig.3.E

-
WB
-
Homo sapiens (Human)
Collected and cropped from Front Pharmacol by CiteAb, provided under a CC-BY license
Image 1 of 86
In Nat Commun on 12 December 2019 by Volta, F., Scerbo, M. J., et al.
Fig.4.B

-
WB
-
Mus musculus (House mouse)
Collected and cropped from Nat Commun by CiteAb, provided under a CC-BY license
Image 1 of 86