Viral protein genome-linked (VPg) is a small protein encoded by several non-enveloped positive-sense RNA viruses, including Cardiovirus. Although VPg is widely recognized as essential for viral RNA synthesis, its additional roles remain largely unexplored. Here, we report an alternative function of Cardiovirus VPg as an interferon-γ receptor agonist. During encephalomyocarditis virus (EMCV) infection, a member of the Cardiovirus genus, we uncovered that VPg from EMCV (VPgEMCV) is released extracellularly from host cells. VPgEMCV is then sensed by uninfected host cells, activating the interferon-γ signaling cascade and ultimately producing nitric oxide (NO) through inducible NO synthase induction. NO accumulation is crucial for triggering potent cell death in cooperation with tumor necrosis factor α (TNF-α) signaling, with TNF-α also being induced as an antiviral host response during EMCV infection. We demonstrate that VPgEMCV, comprising merely 20 amino acids, can exhibit cytokine-mimicry activity, with the synergistic interplay between VPgEMCV and TNF-α signaling regulating viral replication and disease pathogenesis.
Copyright © 2025 The Author(s). Published by Elsevier Inc. All rights reserved.
In Cell Reports on 27 January 2026 by Shirasaka, Y., Takeuchi, F., et al.
In International Journal of Molecular Sciences on 25 July 2025 by Lu, Z., Zhou, X., et al.
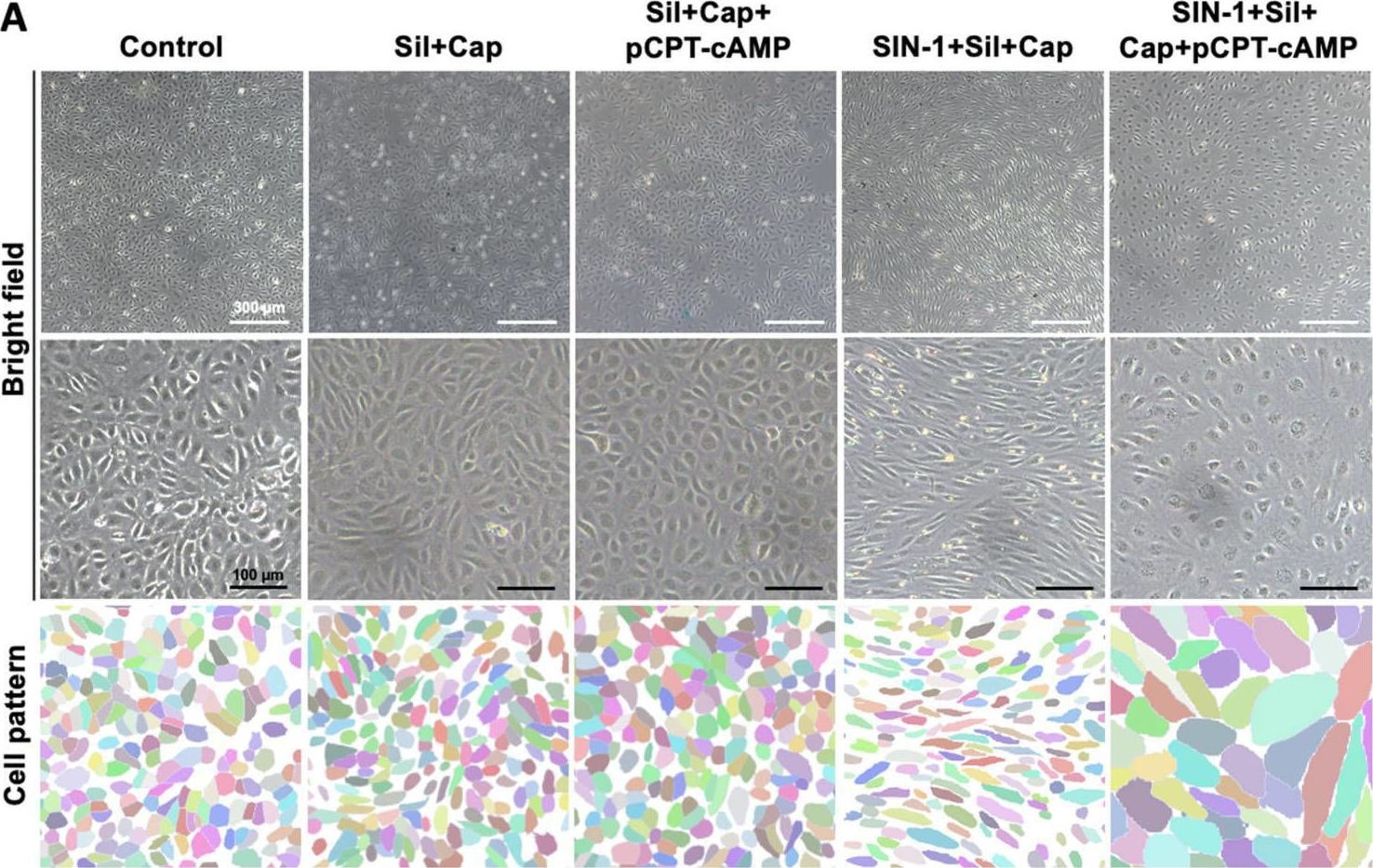
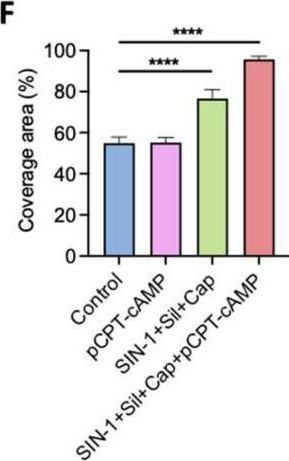
The development of functional endothelial monolayers on synthetic vascular grafts remains challenging, particularly for small-diameter vessels (<6 mm) prone to thrombosis. Here, we present a pharmacological strategy combining 8-(4-chlorophenylthio) adenosine 3',5'-cyclic monophosphate sodium salt (pCPT-cAMP, a tight junction promoter) with nitric oxide/cGMP pathway agonists 3-morpholinosydnonimine (SIN-1), captopril, and sildenafil) to enhance endothelialization. In human umbilical vein endothelial cells (HUVECs), this four-agent cocktail induced a flat, extended phenotype with a 3-fold increased cell area and 57.5% fewer cells required for surface coverage compared to controls. Immunofluorescence analysis revealed enhanced ZO-1 expression and continuous tight junction formation, while sustained nitric oxide (NO) production (3.9-fold increase) and restored prostacyclin (PGI2) secretion demonstrated preserved endothelial functionality. Anticoagulation assays confirmed a significant reduction in thrombus formation (p < 0.01) via dual inhibition of platelet activation and thrombin binding. These findings establish a synergistic drug combination that promotes rapid endothelialization while maintaining antithrombogenic activity, offering a promising solution for small-diameter vascular grafts. Further studies should validate long-term stability and translational potential in preclinical models.
Stable oxidative posttranslational modifications alter the gating properties of RyR1.
In The Journal of General Physiology on 2 December 2024 by Steinz, M. M., Beard, N. A., et al.
The ryanodine receptor type 1 (RyR1) is a Ca2+ release channel that regulates skeletal muscle contraction by controlling Ca2+ release from the sarcoplasmic reticulum (SR). Posttranslational modifications (PTMs) of RyR1, such as phosphorylation, S-nitrosylation, and carbonylation are known to increase RyR1 open probability (Po), contributing to SR Ca2+ leak and skeletal muscle dysfunction. PTMs on RyR1 have been linked to muscle dysfunction in diseases like breast cancer, rheumatoid arthritis, Duchenne muscle dystrophy, and aging. While reactive oxygen species (ROS) and oxidative stress induce PTMs, the impact of stable oxidative modifications like 3-nitrotyrosine (3-NT) and malondialdehyde adducts (MDA) on RyR1 gating remains unclear. Mass spectrometry and single-channel recordings were used to study how 3-NT and MDA modify RyR1 and affect Po. Both modifications increased Po in a dose-dependent manner, with mass spectrometry identifying 30 modified residues out of 5035 amino acids per RyR1 monomer. Key modifications were found in domains critical for protein interaction and channel activation, including Y808/3NT in SPRY1, Y1081/3NT and H1254/MDA in SPRY2&3, and Q2107/MDA and Y2128/3NT in JSol, near the binding site of FKBP12. Though these modifications did not directly overlap with FKBP12 binding residues, they promoted FKBP12 dissociation from RyR1. These findings provide detailed insights into how stable oxidative PTMs on RyR1 residues alter channel gating, advancing our understanding of RyR1-mediated Ca2+ release in conditions associated with oxidative stress and muscle weakness.
© 2024 Steinz et al.
Inhibition of Sirtuin Deacylase Activity by Peroxynitrite.
In Biochemistry on 1 October 2024 by Bohl, K., Wynia-Smith, S. L., et al.
Sirtuins are a class of enzymes that deacylate protein lysine residues using NAD+ as a cosubstrate. Sirtuin deacylase activity has been historically regarded as protective; loss of sirtuin deacylase activity potentially increases susceptibility to aging-related disease development. However, which factors may inhibit sirtuins during aging or disease is largely unknown. Increased oxidant and inflammatory byproduct production damages cellular proteins. Previously, we and others found that sirtuin deacylase activity is inhibited by the nitric oxide (NO)-derived cysteine post-translational modification S-nitrosation. However, the comparative ability of the NO-derived oxidant peroxynitrite (ONOO-) to affect human sirtuin activity had not yet been assessed under uniform conditions. Here, we compare the ability of ONOO- (donated from SIN-1) to post-translationally modify and inhibit SIRT1, SIRT2, SIRT3, SIRT5, and SIRT6 deacylase activity. In response to SIN-1 treatment, inhibition of SIRT1, SIRT2, SIRT3, SIRT5, and SIRT6 deacylase activity correlated with increased tyrosine nitration. Mass spectrometry identified multiple novel tyrosine nitration sites in SIRT1, SIRT3, SIRT5, and SIRT6. As each sirtuin isoform has at least one tyrosine nitration site within the catalytic core, nitration may result in sirtuin inhibition. ONOO- can also react with cysteine residues, resulting in sulfenylation; however, only SIRT1 showed detectable peroxynitrite-mediated cysteine sulfenylation. While SIRT2, SIRT3, SIRT5, and SIRT6 showed no detectable sulfenylation, SIRT6 likely undergoes transient sulfenylation, quickly resolving into an intermolecular disulfide bond. These results suggest that the aging-related oxidant peroxynitrite can post-translationally modify and inhibit sirtuins, contributing to susceptibility to aging-related disease.
In IScience on 20 October 2023 by Suhail, H., Nematullah, M., et al.
Metabolism and energy processes governing oligodendrocyte function during neuroinflammatory disease are of great interest. However, how varied cellular environments affect oligodendrocyte activity during neuroinflammation is unknown. We demonstrate that activated microglial energy metabolism controls oligodendrocyte mitochondrial respiration and activity. Lipopolysaccharide/interferon gamma promote glycolysis and decrease mitochondrial respiration and myelin protein synthesis in rat brain glial cells. Enriched microglia showed an early burst in glycolysis. In microglia-conditioned medium, oligodendrocytes did not respire and expressed less myelin. SCENITH revealed metabolic derangement in microglia and O4-positive oligodendrocytes in endotoxemia and experimental autoimmune encephalitogenic models. The early burst of glycolysis in microglia was mediated by PDPK1 and protein kinase B/AKT signaling. We found that microglia-produced NO and itaconate, a tricarboxylic acid bifurcated metabolite, reduced mitochondrial respiration in oligodendrocytes. During inflammation, we discovered a signaling pathway in microglia that could be used as a therapeutic target to restore mitochondrial function in oligodendrocytes and induce remyelination.
© 2023 The Author(s).
In Int J Mol Sci on 25 July 2025 by Lu, Z., Zhou, X., et al.
Fig.1.B
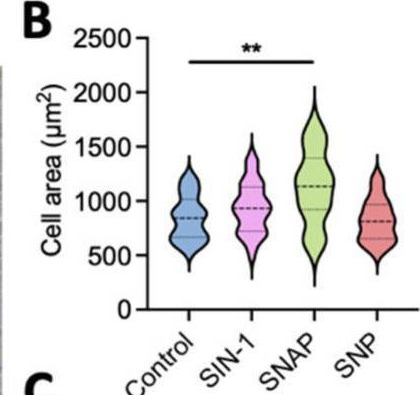
Collected and cropped from International Journal of Molecular Sciences by CiteAb, provided under a CC-BY license
Image 1 of 17
In Int J Mol Sci on 25 July 2025 by Lu, Z., Zhou, X., et al.
Fig.3.A
-
IHC
-
Collected and cropped from International Journal of Molecular Sciences by CiteAb, provided under a CC-BY license
Image 1 of 17
In Int J Mol Sci on 25 July 2025 by Lu, Z., Zhou, X., et al.
Fig.3.C
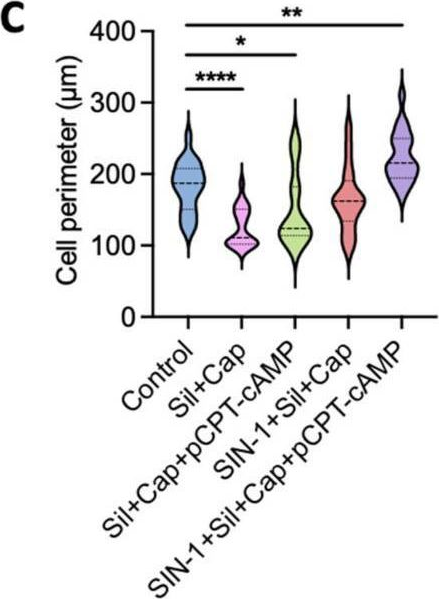
Collected and cropped from International Journal of Molecular Sciences by CiteAb, provided under a CC-BY license
Image 1 of 17
In Int J Mol Sci on 25 July 2025 by Lu, Z., Zhou, X., et al.
Fig.3.B
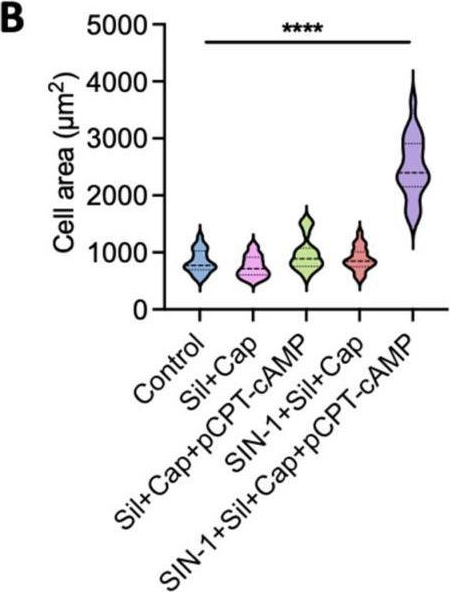
Collected and cropped from International Journal of Molecular Sciences by CiteAb, provided under a CC-BY license
Image 1 of 17
In Int J Mol Sci on 25 July 2025 by Lu, Z., Zhou, X., et al.
Fig.3.D
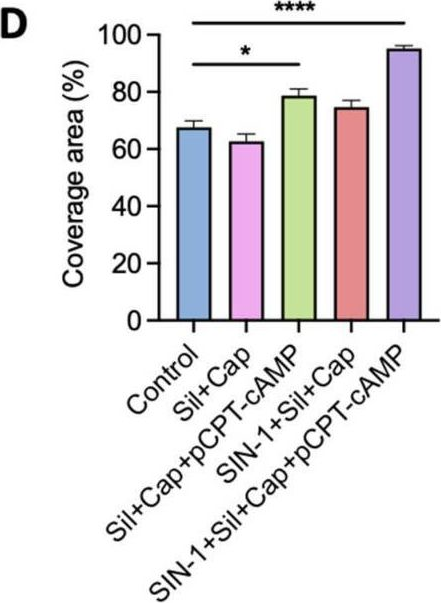
Collected and cropped from International Journal of Molecular Sciences by CiteAb, provided under a CC-BY license
Image 1 of 17
In Int J Mol Sci on 25 July 2025 by Lu, Z., Zhou, X., et al.
Fig.5.B
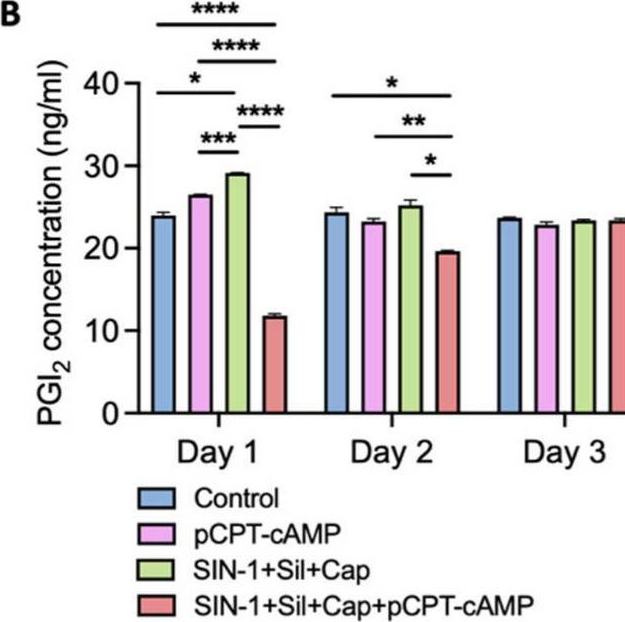
Collected and cropped from International Journal of Molecular Sciences by CiteAb, provided under a CC-BY license
Image 1 of 17
In Int J Mol Sci on 25 July 2025 by Lu, Z., Zhou, X., et al.
Fig.5.E
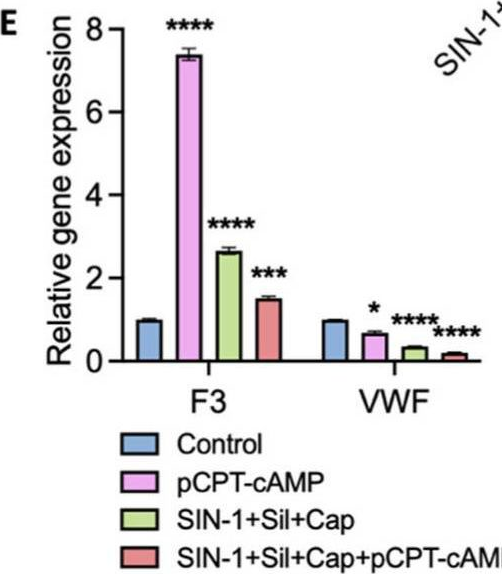
Collected and cropped from International Journal of Molecular Sciences by CiteAb, provided under a CC-BY license
Image 1 of 17
In Int J Mol Sci on 25 July 2025 by Lu, Z., Zhou, X., et al.
Fig.5.D
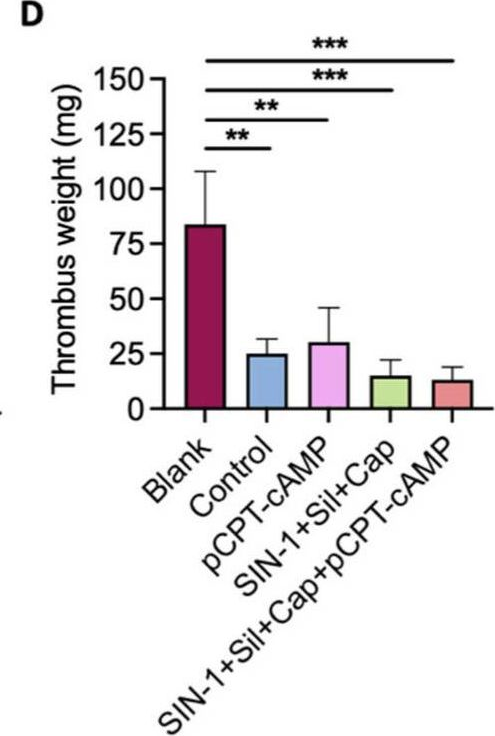
Collected and cropped from International Journal of Molecular Sciences by CiteAb, provided under a CC-BY license
Image 1 of 17
In Int J Mol Sci on 25 July 2025 by Lu, Z., Zhou, X., et al.
Fig.5.A
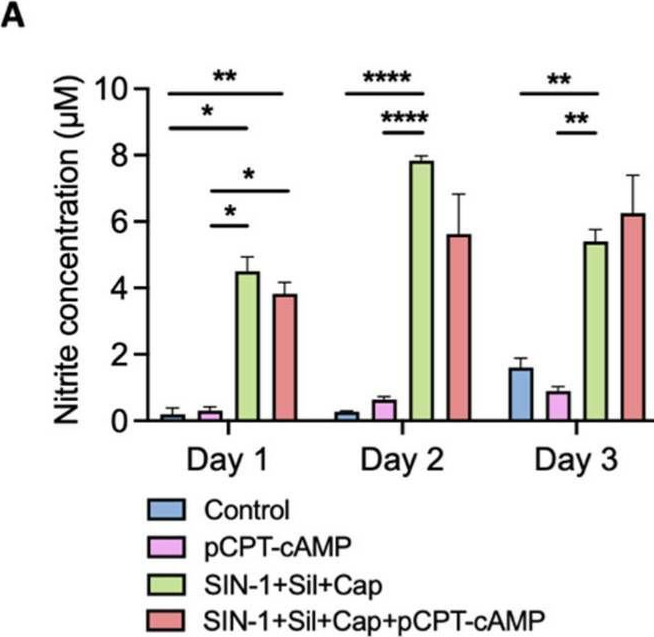
Collected and cropped from International Journal of Molecular Sciences by CiteAb, provided under a CC-BY license
Image 1 of 17
In Int J Mol Sci on 25 July 2025 by Lu, Z., Zhou, X., et al.
Fig.4.D
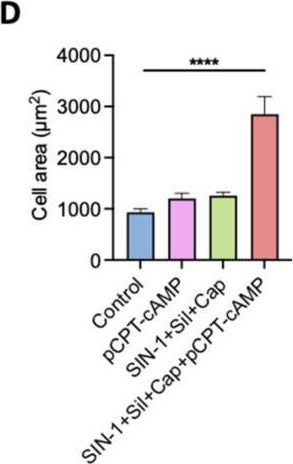
Collected and cropped from International Journal of Molecular Sciences by CiteAb, provided under a CC-BY license
Image 1 of 17
In Int J Mol Sci on 25 July 2025 by Lu, Z., Zhou, X., et al.
Fig.1.A
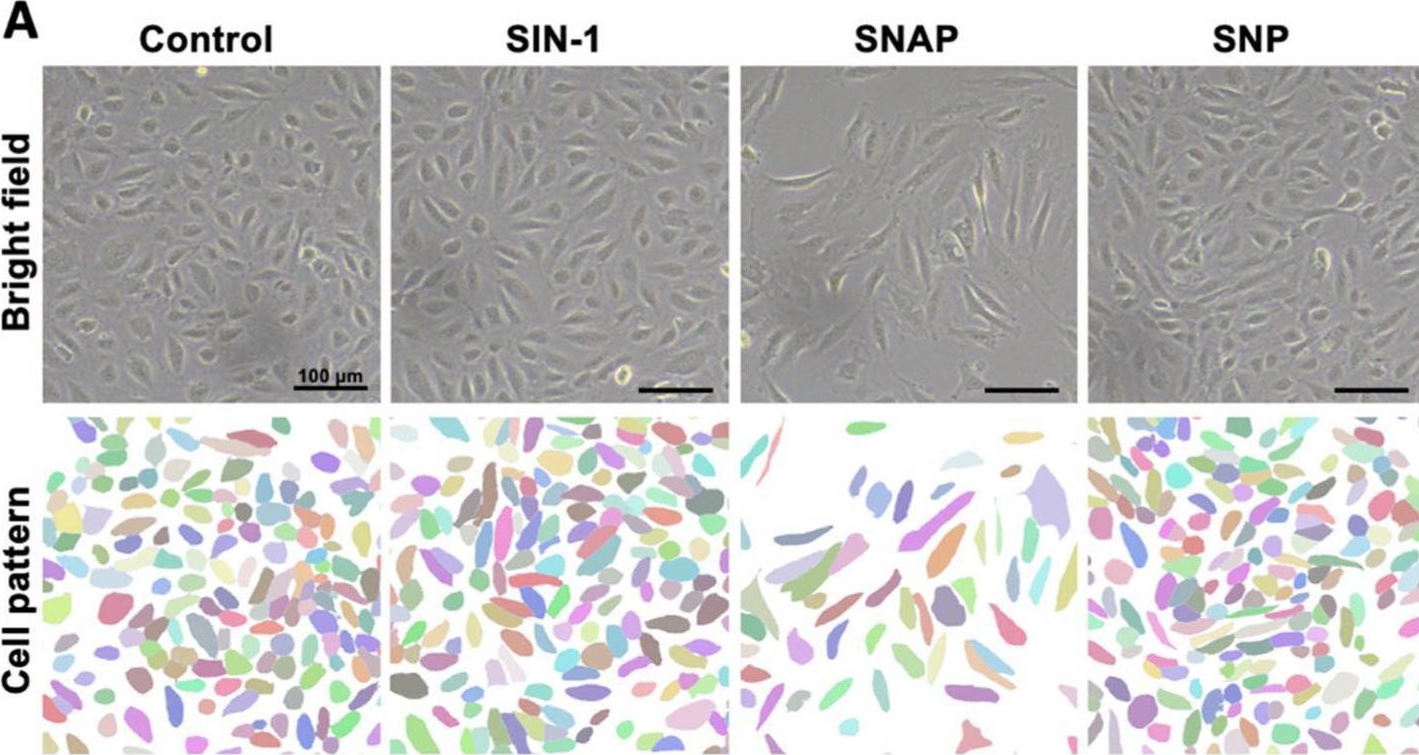
Collected and cropped from International Journal of Molecular Sciences by CiteAb, provided under a CC-BY license
Image 1 of 17
In Int J Mol Sci on 25 July 2025 by Lu, Z., Zhou, X., et al.
Fig.1.C
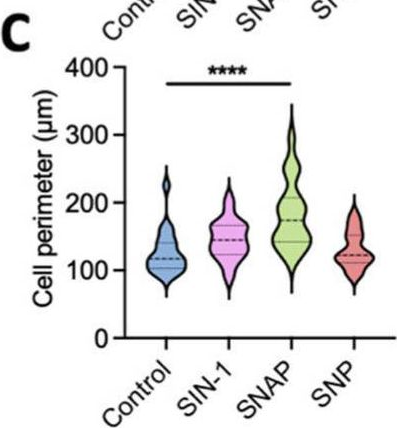
Collected and cropped from International Journal of Molecular Sciences by CiteAb, provided under a CC-BY license
Image 1 of 17
In Int J Mol Sci on 25 July 2025 by Lu, Z., Zhou, X., et al.
Fig.4.A
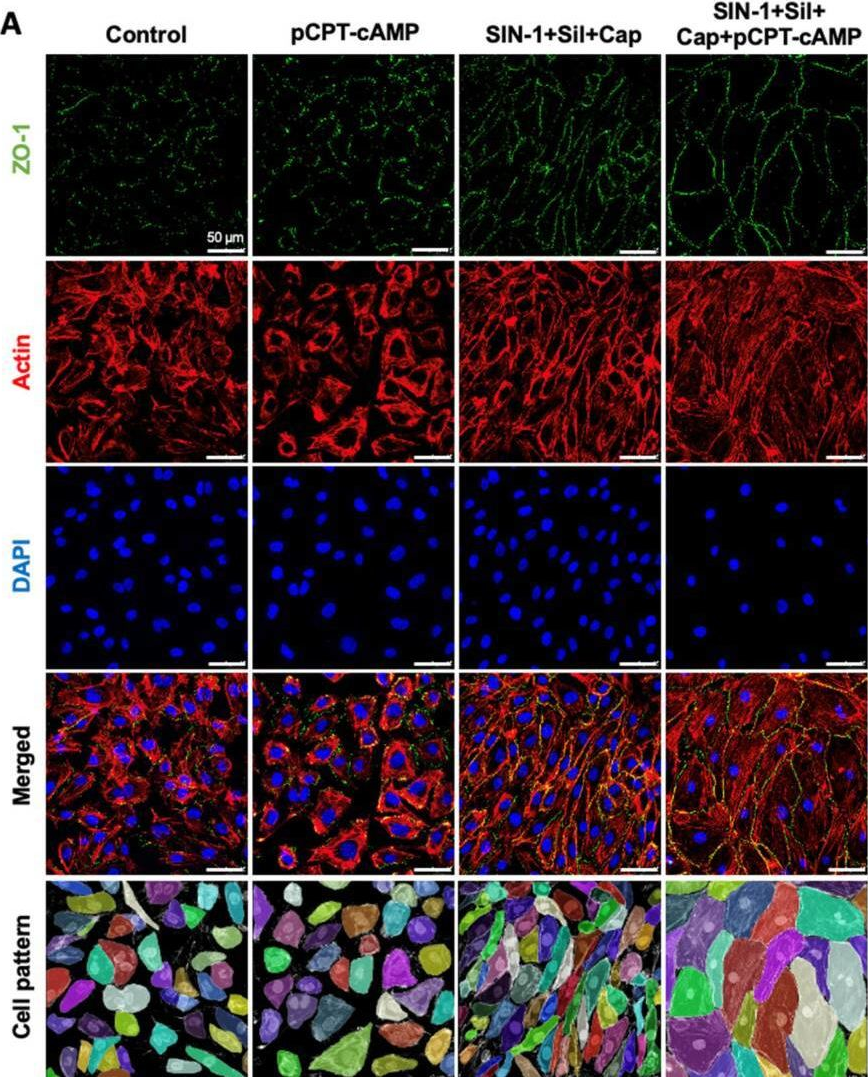
Collected and cropped from International Journal of Molecular Sciences by CiteAb, provided under a CC-BY license
Image 1 of 17
In Int J Mol Sci on 25 July 2025 by Lu, Z., Zhou, X., et al.
Fig.4.C
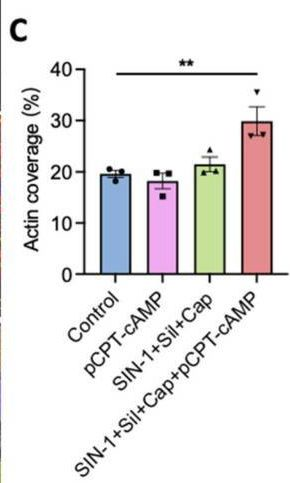
Collected and cropped from International Journal of Molecular Sciences by CiteAb, provided under a CC-BY license
Image 1 of 17
In Int J Mol Sci on 25 July 2025 by Lu, Z., Zhou, X., et al.
Fig.4.B
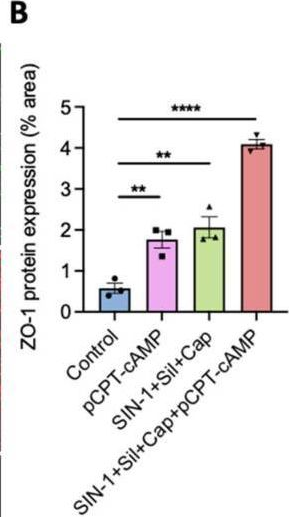
Collected and cropped from International Journal of Molecular Sciences by CiteAb, provided under a CC-BY license
Image 1 of 17
In Int J Mol Sci on 25 July 2025 by Lu, Z., Zhou, X., et al.
Fig.4.E
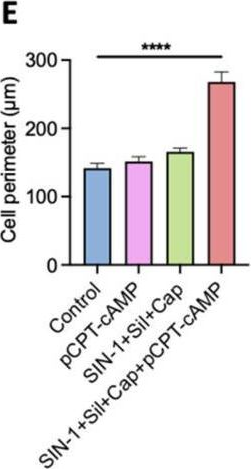
Collected and cropped from International Journal of Molecular Sciences by CiteAb, provided under a CC-BY license
Image 1 of 17
In Int J Mol Sci on 25 July 2025 by Lu, Z., Zhou, X., et al.
Fig.4.F
Collected and cropped from International Journal of Molecular Sciences by CiteAb, provided under a CC-BY license
Image 1 of 17
