Cell transplantation therapy is a promising new treatment for myocardial infarction and heart failure. Developing methods to promote the engraftment of cardiac stem cells into cardiac tissue and their differentiation into cardiac cells is a strategy for improving the efficacy of cell transplantation therapy. Tanshinone VI (TanVI), a component extracted from the roots of Salvia miltiorrhiza Bunge (Labiatae), is a possible pharmacological agent for ischemic heart disease. However, since the effects of TanVI on the differentiation of cardiac stem cells remain unclear, we examined the effects of TanVI on the cardiac differentiation potential of cardiac stem cells. Cardiosphere-derived cells (CDCs), the cardiac stem/progenitor cells, were prepared from adult rat hearts. After CDCs were cultured in the presence of TanVI, cell numbers, expression of cardiomyocyte markers, and intracellular signaling protein levels involved in the differentiation into cardiomyocytes were measured. Treatment of CDCs with TanVI inhibited the increase in cell numbers and induced their expression of cardiomyocyte marker proteins. TanVI treatment also inhibited the Akt/glycogen synthase kinase 3β (GSK3β)/β-catenin and c-Raf/MEK1/2/extracellular signal-regulated kinase 1/2 (ERK1/2) pathways. We found that TanVI promotes cardiac differentiation of CDCs. Our findings also suggest that inhibition of the Akt/GSK3β/β-catenin pathway and the c-Raf/MEK1/2/ERK1/2 pathway may contribute to the differentiation from CDCs to cardiomyocytes. TanVI may play an adjunctive role in cardiac stem cell transplantation therapy.
Applications
Reactivity
Research Area
In Biological Pharmaceutical Bulletin on 28 August 2025 by Kawahara, Y., Marunouchi, T., et al.
-
WB
-
Rattus norvegicus (Rat)
-
Cardiovascular biology
In Molecular Medicine on 7 May 2021 by Shi, Q., Ni, X., et al.
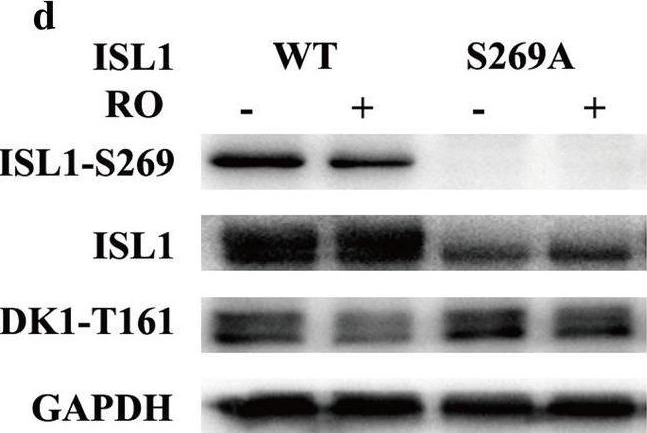
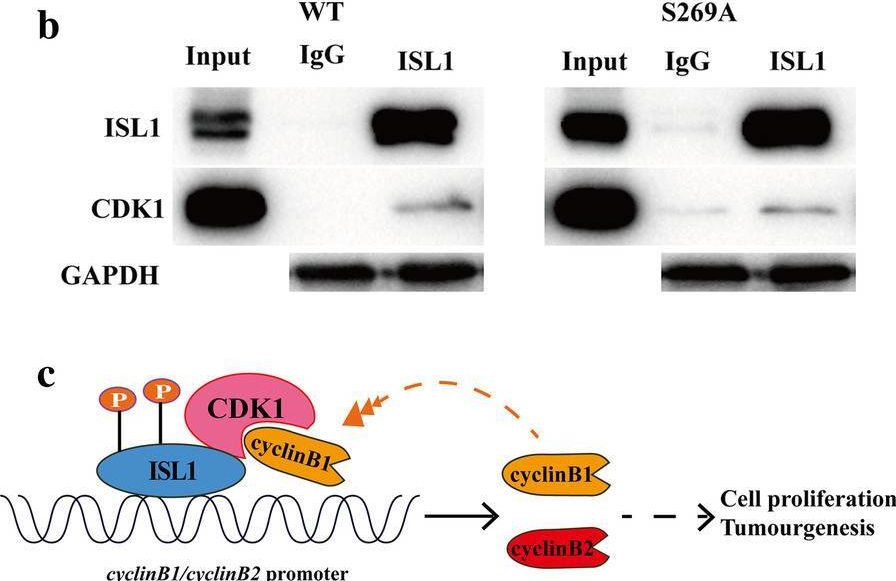
Despite recent advances in diagnostic and therapeutic approaches for gastric cancer (GC), the survival of patients with advanced GC remains very low. Islet-1 (ISL1) is a LIM-homeodomain transcription factor, which is upregulated and promotes cell proliferation in GC. The exact mechanism by which ISL1 influences GC development is unclear.
Co-immunoprecipitation (co-IP) and glutathione S-transferase (GST)-pulldown assays were employed to evaluate the interaction of ISL1 with CDK1. Western blot and immunohistochemistry analyses were performed to evaluate the ability of CDK1 to phosphorylate ISL1 at Ser 269 in GC cell and tissue specimens. Chromatin immunoprecipitation (ChIP), ChIP re-IP, luciferase reporter, and CCK-8 assays were combined with flow cytometry cell cycle analysis to detect the transactivation potency of ISL1-S269-p and its ability to promote cell proliferation. The self-stability and interaction with CDK1 of ISL1-S269-p were also determined.
ISL1 is phosphorylated by CDK1 at serine 269 (S269) in vivo. Phosphorylation of ISL1 by CDK1 on serine 269 strengthened its binding on the cyclin B1 and cyclin B2 promoters and increased its transcriptional activity in GC. Furthermore, CDK1-dependent phosphorylation of ISL1 correlated positively with ISL1 protein self-stability in NIH3T3 cells.
ISL1-S269-p increased ISL1 transcriptional activity and self-stability while binding to the cyclinB1 and cyclinB2 promoters promotes cell proliferation. ISL1-S269-p is therefore crucial for tumorigenesis and potentially a direct therapeutic target for GC.
-
IP
-
WB
-
Homo sapiens (Human)
-
Biochemistry and Molecular biology
-
Cancer Research
Cross-talk of SFRP4, integrin α1β1, and Notch1 inhibits cardiac differentiation of P19CL6 cells.
In Cellular Signalling on 1 November 2016 by Tian, Y., Wang, W., et al.
Signaling pathways play an important role in cardiogenesis. Secreted frizzled-related protein 4 (SFRP4), a member of the Wnt family, contributes to adipogenesis and tumorigenesis. However, how SFRP4 participates in cardiogenesis and the detailed molecular mechanisms involved have not been elucidated. The aim of this work was to determine cross-talk between SFRP4, integrin α1β1, and Notch1 during cardiac differentiation of P19CL6 cells. Using a well-established in vitro P19CL6 cell cardiomyocyte differentiation system, we found that SFRP4 inhibited P19CL6 cell cardiac differentiation via SFRP4 overexpression or knockdown. In addition, the SFRP4 overexpression augmented Notch1 and HES1 production. Further investigation demonstrated that SFRP4 bound to integrin α1β1 to activate the focal adhesion kinase (FAK) pathway and that phosphorylated FAK Y397 (p-FAK Y397) aided Notch intracellular domain 1 (NICD1) nuclear translocation to form a p-FAK Y397-NICD1 complex that activated the Hes1 promoter. Taken together, the cross-talk between SFRP4, integrin α1β1, and Notch1 suppresses the cardiac differentiation of P19CL6 cells.
Copyright © 2016 The Authors. Published by Elsevier Inc. All rights reserved.
-
WB
-
Homo sapiens (Human)
-
Cardiovascular biology
-
Cell Biology
In Frontiers in Genetics on 5 December 2015 by Lizio, M., Ishizu, Y., et al.
Mammals are composed of hundreds of different cell types with specialized functions. Each of these cellular phenotypes are controlled by different combinations of transcription factors. Using a human non islet cell insulinoma cell line (TC-YIK) which expresses insulin and the majority of known pancreatic beta cell specific genes as an example, we describe a general approach to identify key cell-type-specific transcription factors (TFs) and their direct and indirect targets. By ranking all human TFs by their level of enriched expression in TC-YIK relative to a broad collection of samples (FANTOM5), we confirmed known key regulators of pancreatic function and development. Systematic siRNA mediated perturbation of these TFs followed by qRT-PCR revealed their interconnections with NEUROD1 at the top of the regulation hierarchy and its depletion drastically reducing insulin levels. For 15 of the TF knock-downs (KD), we then used Cap Analysis of Gene Expression (CAGE) to identify thousands of their targets genome-wide (KD-CAGE). The data confirm NEUROD1 as a key positive regulator in the transcriptional regulatory network (TRN), and ISL1, and PROX1 as antagonists. As a complimentary approach we used ChIP-seq on four of these factors to identify NEUROD1, LMX1A, PAX6, and RFX6 binding sites in the human genome. Examining the overlap between genes perturbed in the KD-CAGE experiments and genes with a ChIP-seq peak within 50 kb of their promoter, we identified direct transcriptional targets of these TFs. Integration of KD-CAGE and ChIP-seq data shows that both NEUROD1 and LMX1A work as the main transcriptional activators. In the core TRN (i.e., TF-TF only), NEUROD1 directly transcriptionally activates the pancreatic TFs HSF4, INSM1, MLXIPL, MYT1, NKX6-3, ONECUT2, PAX4, PROX1, RFX6, ST18, DACH1, and SHOX2, while LMX1A directly transcriptionally activates DACH1, SHOX2, PAX6, and PDX1. Analysis of these complementary datasets suggests the need for caution in interpreting ChIP-seq datasets. (1) A large fraction of binding sites are at distal enhancer sites and cannot be directly associated to their targets, without chromatin conformation data. (2) Many peaks may be non-functional: even when there is a peak at a promoter, the expression of the gene may not be affected in the matching perturbation experiment.
-
ChIP
-
Homo sapiens (Human)
-
Biochemistry and Molecular biology
-
Genetics
ISL-1 promotes pancreatic islet cell proliferation by forming an ISL-1/Set7/9/PDX-1 complex.
In Cell Cycle on 16 July 2015 by Yang, Z., Zhang, Q., et al.
Islet-1 (ISL-1), a LIM-homeodomain transcription factor, has been recently found to be essential for promoting postnatal pancreatic islet proliferation. However, the detailed mechanism has not yet been elucidated. In the present study, we investigated the mechanism by which ISL-1 promotes β-cell proliferation through regulation of CyclinD1 in HIT-T15 and NIT-1 cells, as well in rat islet mass. Our results provide the evidence that ISL-1 promotes adult pancreatic islet β-cell proliferation by activating CyclinD1 transcription through cooperation with Set7/9 and PDX-1 to form an ISL-1/Set7/9/PDX-1 complex. This complex functions in an ISL-1-dependent manner, with Set7/9 functioning not only as a histone methyltransferase, which increases the histone H3K4 tri-methylation of the CyclinD1 promoter region, but also an adaptor to bridge ISL-1 and PDX-1, while PDX-1 functions as a RNA pol II binding modulator. Furthermore, the formation of the ISL-1/Set7/9/PDX-1 complex is positively associated with insulin-like growth factor-1 treatment in NIT and HIT-T15 cells in vitro, while may be negatively correlated with age in vivo.
-
IP
-
Cell Biology
In Mol Med on 7 May 2021 by Shi, Q., Ni, X., et al.
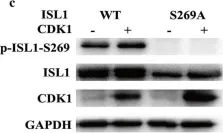
Fig.3.C
-
WB
-
Collected and cropped from Molecular Medicine by CiteAb, provided under a CC-BY license
Image 1 of 5
In Mol Med on 7 May 2021 by Shi, Q., Ni, X., et al.
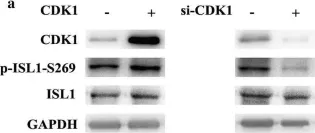
Fig.3.B
-
WB
-
Collected and cropped from Molecular Medicine by CiteAb, provided under a CC-BY license
Image 1 of 5
In Mol Med on 7 May 2021 by Shi, Q., Ni, X., et al.
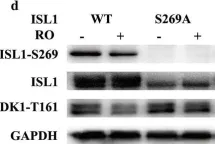
Fig.3.A
-
WB
-
Collected and cropped from Molecular Medicine by CiteAb, provided under a CC-BY license
Image 1 of 5
In Mol Med on 7 May 2021 by Shi, Q., Ni, X., et al.
Fig.3.D
-
WB
-
Collected and cropped from Molecular Medicine by CiteAb, provided under a CC-BY license
Image 1 of 5
In Mol Med on 7 May 2021 by Shi, Q., Ni, X., et al.
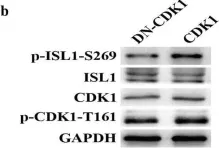
Fig.6.B
-
WB
-
Collected and cropped from Molecular Medicine by CiteAb, provided under a CC-BY license
Image 1 of 5